Abstract
Purpose
Spinal cord gliomas are rare, and there is no consensus on the optimal radiotherapy (RT) regimen. Herein, we investigated therapeutic outcomes in spinal cord gliomas to obtain clues for the optimal RT regimen.
Methods
We assessed 45 patients who received RT for primary spinal cord non-ependymoma gliomas between 2005 and 2017: 37 (82%) received postoperative RT, 6 (13%) underwent definitive RT without surgery, and 2 (5%) received salvage RT for recurrent tumors. Craniospinal irradiation (CSI; median, 40 Gy) was administered in 4 patients with seeding at diagnosis; all other patients received local RT only (median, 50.4 Gy).
Results
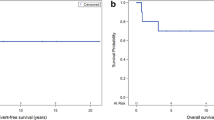
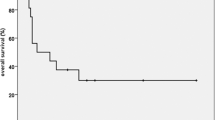
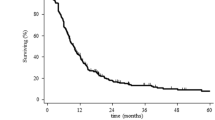
In all 23 failures occurred (20 in patients without initial seeding +3 in patients with initial seeding and CSI; median follow-up, 33 months). The 2‑year overall survival and progression-free survival rates were 74 and 54%, respectively. Overall, 13 (32%) new seeding events outside the local RT field developed either first or subsequently. Tumor grade was significantly associated with survival endpoints (p = 0.009, 0.028) and overall seeding rates (p = 0.042). In grade II tumors, seeding developed in 23%, with a dismal prognosis (median, 10 months after RT). In grade III tumors, seeding developed in 45% with diverse prognosis. In grade IV tumors, seeding developed in 45%. The survival of patients with newly developed seeding was significantly worse than the others (2-year 50%, p < 0.001).
Conclusion
To encompass a considerable rate of progressive disease seeding, aggressive treatment such as pre-emptive application of CSI needs to be considered for high-grade spinal cord gliomas with adverse features. Prophylactic CSI could be an option for survival prolongation and requires prospective validation.
Zusammenfassung
Zweck
Rückenmarksgliome sind selten, bezüglich der bestmöglichen Radiotherapie (RT) besteht kein Konsens. Untersucht wurden Behandlungsresultate im Hinblick auf das optimale RT- Schema.
Methoden
Untersucht wurden 45 Patienten, welche zwischen 2005 und 2017 wegen primärer Non-ependymomaler-Rückenmarksgliome mittels RT behandelte wurden: 37 (82 %) waren postoperativ, 6 (13 %) waren definitiv bestrahlt worden, 2 (5 %) haben aufgrund von Tumorrezidiven eine Salvage-RT erhalten. 4 Patienten mit zum Diagnosezeitpunkt bereits metastasierten Tumoren haben eine kraniospinale Bestrahlung („craniospinal irradiation“) erhalten. Die übrigen Patienten waren allein lokal bestrahlt worden (Median 50,4 Gy).
Ergebnisse
In 23 Fällen kam es zu einem Therapieversagen (bei 20 Patienten ohne Metastasierung zum Diagnosezeitpunkt und bei 3 Patienten mit Metastasen zum Diagnosezeitpunkt welche mit CSI behandelt wurden). Die Zweijahresgesamtüberlebensrate und die krankheitsfreie Überlebensrate betrugen 74 bzw. 54 %. Insgesamt traten zu Beginn oder im Verlauf in 13 Fällen (32 %) neue Metastasen außerhalb des Bestrahlungsfeldsauf. Das Tumorgrading korrelierte signifikant mit den Überlebensendpunkten (p = 0,009; 0,028) und der Gesamtaussaatrate (p = 0,042). Bei G2‑Tumoren entwickelten sich in 23 % Absiedlungen mit sehr ungünstiger Prognose (Median 10 Monate nach RT), bei G3‑Tumoren kam es bei 45 % zu einer Streuung mit unterschiedlicher Prognose. Patienten mit G4‑Tumoren entwickelten in 45 % Metastasen, die Überlebensraten bei neu auftretenden Metastasen waren deutlich geringer (Zweijahresüberlebensrate 50 %, p < 0,001).
Schlussfolgerung
Aufgrund der hohen Metastasierungsrate bei Rückenmarksgliomen mit hohem Grading und ungünstigen Merkmalen sollte eine intensivierte Therapie wie etwa eine prophylaktische CSI erwogen werden. Eine prophylaktische CSI könnte eine Option zur Verlängerung des Überlebens sein und erfordert eine prospektive Validierung.


Similar content being viewed by others
References
Henson JW (2001) Spinal cord gliomas. Curr Opin Neurol 14:679–682
Nabors LB, Portnow J, Ammirati M et al (2017) NCCN guidelines insights: central nervous system cancers, version 1.2017. J Natl Compr Canc Netw 15:1331–1345
Kahn J, Loeffler JS, Niemierko A et al (2011) Long-term outcomes of patients with spinal cord gliomas treated by modern conformal radiation techniques. Int J Radiat Oncol Biol Phys 81:232–238
Corradini S, Hadi I, Hankel V et al (2016) Radiotherapy of spinal cord gliomas: a retrospective mono-institutional analysis. Strahlenther Onkol 192:139–145
Raco A, Esposito V, Lenzi J et al (2005) Long-term follow-up of intramedullary spinal cord tumors: a series of 202 cases. Neurosurgery 56:972–981 (discussion 972–981)
Abdel-Wahab M, Etuk B, Palermo J et al (2006) Spinal cord gliomas: a multi-institutional retrospective analysis. Int J Radiat Oncol Biol Phys 64:1060–1071
Kim MS, Chung CK, Choe G et al (2001) Intramedullary spinal cord astrocytoma in adults: postoperative outcome. J Neurooncol 52:85–94
Isaacson SR (2000) Radiation therapy and the management of intramedullary spinal cord tumors. J Neurooncol 47:231–238
McLaughlin MP, Buatti JM, Marcus RB Jr. et al (1998) Outcome after radiotherapy of primary spinal cord glial tumors. Radiat Oncol Investig 6:276–280
Garcia DM (1985) Primary spinal cord tumors treated with surgery and postoperative irradiation. Int J Radiat Oncol Biol Phys 11:1933–1939
Shaw EG, Evans RG, Scheithauer BW et al (1986) Radiotherapeutic management of adult intraspinal ependymomas. Int J Radiat Oncol Biol Phys 12:323–327
Chang CH, Housepian EM, Herbert C Jr. (1969) An operative staging system and a megavoltage radiotherapeutic technic for cerebellar medulloblastomas. Radiology 93:1351–1359
Schellinger KA, Propp JM, Villano JL et al (2008) Descriptive epidemiology of primary spinal cord tumors. J Neurooncol 87:173–179
McGuire CS, Sainani KL, Fisher PG (2009) Both location and age predict survival in ependymoma: a SEER study. Pediatr Blood Cancer 52:65–69
Schild SE, Nisi K, Scheithauer BW et al (1998) The results of radiotherapy for ependymomas: the Mayo Clinic experience. Int J Radiat Oncol Biol Phys 42:953–958
Abdel-Wahab M, Corn B, Wolfson A et al (1999) Prognostic factors and survival in patients with spinal cord gliomas after radiation therapy. Am J Clin Oncol 22:344–351
Shirato H, Kamada T, Hida K et al (1995) The role of radiotherapy in the management of spinal cord glioma. Int J Radiat Oncol Biol Phys 33:323–328
Hulshof MC, Menten J, Dito JJ et al (1993) Treatment results in primary intraspinal gliomas. Radiother Oncol 29:294–300
Milano MT, Johnson MD, Sul J et al (2010) Primary spinal cord glioma: a surveillance, epidemiology, and end results database study. J Neurooncol 98:83–92
Lee J, Cho J, Chang JH et al (2016) Re-irradiation for recurrent gliomas: treatment outcomes and prognostic factors. Yonsei Med J 57:824–830
Lee J, Ahn SS, Chang JH et al (2018) Hypofractionated re-irradiation after maximal surgical resection for recurrent glioblastoma: therapeutic adequacy and its prognosticators of survival. Yonsei Med J 59:194–201
Shi W, Scannell BM, Gilbert MR et al (2018) Investigating the effect of reirradiation or systemic therapy in patients with glioblastoma after tumor progression: a secondary analysis of NRG oncology/radiation therapy oncology group trial 0525. Int J Radiat Oncol Biol Phys 100:38–44
Arvold ND, Shi DD, Aizer AA et al (2017) Salvage re-irradiation for recurrent high-grade glioma and comparison to bevacizumab alone. J Neurooncol 135:581–591
Tsang DS, Burghen E, Klimo P Jr. et al (2018) Outcomes after reirradiation for recurrent pediatric intracranial ependymoma. Int J Radiat Oncol Biol Phys 100:507–515
Chun HC, Schmidt-Ullrich RK, Wolfson A et al (1990) External beam radiotherapy for primary spinal cord tumors. J Neurooncol 9:211–217
Rodrigues GB, Waldron JN, Wong CS et al (2000) A retrospective analysis of 52 cases of spinal cord glioma managed with radiation therapy. Int J Radiat Oncol Biol Phys 48:837–842
Sandler HM, Papadopoulos SM, Thornton AF Jr. et al (1992) Spinal cord astrocytomas: results of therapy. Neurosurgery 30:490–493
Ryu SJ, Kim JY, Kim KH et al (2016) A retrospective observational study on the treatment outcomes of 26 patients with spinal cord astrocytoma including two cases of malignant transformation. Eur Spine J 25:4067–4079
Benes V 3rd, Barsa P, Benes V Jr. et al (2009) Prognostic factors in intramedullary astrocytomas: a literature review. Eur Spine J 18:1397–1422
Xiao R, Abdullah KG, Miller JA et al (2016) Molecular and clinical prognostic factors for favorable outcome following surgical resection of adult intramedullary spinal cord astrocytomas. Clin Neurol Neurosurg 144:82–87
Innocenzi G, Salvati M, Cervoni L et al (1997) Prognostic factors in intramedullary astrocytomas. Clin Neurol Neurosurg 99:1–5
Roonprapunt C, Houten JK (2006) Spinal cord astrocytomas: presentation, management, and outcome. Neurosurg Clin N Am 17:29–36
Solomon DA, Wood MD, Tihan T et al (2016) Diffuse midline gliomas with Histone H3-K27M mutation: a series of 47 cases assessing the spectrum of morphologic variation and associated genetic alterations. Brain Pathol 26:569–580
Louis DN, Perry A, Reifenberger G et al (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820
Korshunov A, Ryzhova M, Hovestadt V et al (2015) Integrated analysis of pediatric glioblastoma reveals a subset of biologically favorable tumors with associated molecular prognostic markers. Acta Neuropathol 129:669–678
Sturm D, Witt H, Hovestadt V et al (2012) Hotspot mutations in H3F3A and IDH1 define distinct epigenetic and biological subgroups of glioblastoma. Cancer Cell 22:425–437
Khuong-Quang DA, Buczkowicz P, Rakopoulos P et al (2012) K27M mutation in histone H3.3 defines clinically and biologically distinct subgroups of pediatric diffuse intrinsic pontine gliomas. Acta Neuropathol 124:439–447
Aihara K, Mukasa A, Gotoh K et al (2014) H3F3A K27M mutations in thalamic gliomas from young adult patients. Neuro-oncology 16:140–146
Feng J, Hao S, Pan C et al (2015) The H3.3 K27M mutation results in a poorer prognosis in brainstem gliomas than thalamic gliomas in adults. Hum Pathol 46:1626–1632
Acknowledgements
The abstract of this study was accepted for poster viewing at the 37th Annual Meeting of the the European SocieTy for Radiotherapy & Oncology (ESTRO), Barcelona, Spain, April, 2018, and presented at the Annual Meeting of the Korean Society for Neuro-Oncology (KSNO), Seoul, March 2018.
The authors would like to thank Franziska Walter (LMU University Hospital, D-81377, Munich, Germany.) for her help with the German abstract of this manuscript.
Funding
This work was supported by Ministry of Science, Korea through the R&D program of NRF-2017R1C1B2010379.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
S.H. Choi, H.I. Yoon, S. Yi, J.W. Park, J. Cho, D.A. Shin, Y. Ha, D.-S. Kim, S.H. Kim, S.-K. Lee, J.H. Chang and C.-O. Suh declare that they have no competing interests.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Caption Electronic Supplementary Material
66_2018_1366_MOESM1_ESM.docx
Supplementary Material: Supplementary Text: Details about follow-up visits and definitions of failure events; Supplementary Figures: Survival graphs and Univariate/multivariate analyses for survivals
Rights and permissions
About this article
Cite this article
Choi, S.H., Yoon, H.I., Yi, S. et al. Treatment outcomes of radiotherapy for primary spinal cord glioma. Strahlenther Onkol 195, 164–174 (2019). https://doi.org/10.1007/s00066-018-1366-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-018-1366-3