Summary
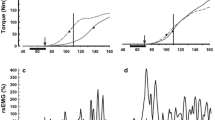
The contractile properties of motor units (MUs) in two multi-tendoned forelimb muscles were investigated. In anesthetized cats single MUs of the extensor carpi ulnaris (ECU) and extensor digitorum communis (EDC) muscles were selectively activated by stimulation of cervical ventral root filaments. MUs were characterized by various tests including single twitches, series of tetanic contractions providing a tension-frequency relation and a fatigue test. They were classified by the parameters contraction time (CT, time-to-peak within unpotentiated single twitches) and fatigue-index (RB, according to Burke). The ECU muscle is composed of 38% type FR MUs (fast, fatigue-sensitive; CT<38 ms; RB<0.5), 35% type FR MUs (CT<38 ms, RB>0.5) and 27% type S MUs (slow; CT>38 ms, RB>0.5). 46% of the EDC MUs were classified as FF (RB≤0.25), 29% as FI (fast, intermediately fatiguable; 0.25<RB<0.75) and 25% as FR/S (fatigue-resistant, fast or slow; RB>-0.75). The latter group was devised since most MUs appeared as fast and the unequivocal presence of slow MUs could neither be demonstrated nor excluded. Normalized tension-frequency relations of fast ECU and EDC MUs were nearly identical and similar to those reported for fast MUs of other muscles. In contrast to this, the tension-frequency relation of slow ECU MUs has a different shape supporting the use of this function to distinguish fast from slow MUs. The distribution of different types of MUs is discussed with regard to the structure and function of the parent muscles and in relation to hindlimb muscles of comparable architecture. As revealed by comparison to EMG data gained in behaving animals (Fritz et al. 1985; Hoffmann et al. 1986, Botterman et al. 1985), the three muscles of the cat distal forelimb investigated so far seem to be adapted to different tasks: the EDC to rapid movements with a high proportion of type FF MUs, flexor carpi radialis to sustained contractions during the body support with a high proportion of fatigue-resistant MUs; the ECU which changes synergism between both muscles has an intermediate composition.
Similar content being viewed by others
References
Botterman BR, Hamm TM, Reinking RM, Stuart DG (1983) Localization of monosynaptic Ia excitatory post-synaptic potentials in the motor nucleus of the cat biceps femoris muscle. J Physiol 338:355–377
Botterman BR, Iwamoto GA, Gonyea WJ (1985) Classification of motor units in flexor carpi radialis muscle of the cat. J Neurophysiol 54:676–690
Brink EE, Jinnai K, Wilson VJ (1981) Pattern of segmental monosynaptic input to cat dorsal neck motoneurons. J Neurophysiol 46:496–505
Burke RE (1981) Motor units: anatomy, physiology and functional organization. In: Handbook of physiology. The nervous system: motor control, vol 1, part 2, ed Brooks, VB. Williams & Wilkins, Bethesda pp 345–422
Burke RE, Tsairis P (1973) Anatomy and innervation ratios in motor units of cat gastrocnemius. J Physiol (Lond) 234:749–765
Burke RE, Levine DN, Tsairis P, Zajac FE (1973) Physiological types and histochemical profiles in motor units of the cat gastrocnemius. J Physiol (Lond) 234:723–748
Burke RE, Rudomin P, Zajac FE (1976) The effect of activation history on tension production by individual muscle units. Brain Res 109:515–529
Crouch JE (1969) Text-atlas of cat anatomy. Lea and Febiger, Philadelphia: pp 117–119
Dum RP, Kennedy TT (1980) Physiological and histochemical characteristics of motor units in cat tibialis anterior and extensor digitorum longus muscles. J Neurophysiol 43:1615–1630
English AW (1978) An electromyographic analysis of forelimb muscles during overground stepping in the cat. J Exp Biol 76:105–122
Fritz N (1981) Ia-Synergismus an der vorderen Extremität der Katze. Thesis, University of München
Fritz N (1987) Contractile properties of motor units in the cat extensor digitorum communis (EDC) muscle. Neuroscience 22:S642
Fritz N, Schimidt Ch (1988) Contractile properties of motor units in the cat extensor carpi ulnaris muscle. Pflügers Arch [Suppl 1] 411:R136
Fritz N, Illert M, Wiedemann E (1985) EMG studies in the distal forelimb of the cat during locomotion and food taking movements. Acta Physiol Scand 123:55A
Fritz N, Illert M, Reeh P (1986a) Location of motoneurones projecting to the cat distal forelimb. II. Median and ulnar motornuclei. J Comp Neurol 244:302–312
Fritz N, Illert M, Saggau P (1986b) Location of motoneurones projecting to the cat distal forelimb. I. Deep radial motornuclei. J Comp Neurol 244:286–301
Fritz N, Illert M, de la Motte S, Reeh P, Saggau P (1989) Pattern of monosynaptic Ia connections in the cat forelimb. J Physiol (Lond) 419:321–351
Fritz N, Schmidt Ch, Yamaguchi T (1991) Biomechanical organization of single motor units within two multi-tendoned muscles of the cat distal forelimb. Exp Brain Res 88:411–421
Goslow GE Jr, Cameron WE, Stuart DG (1977) The fast twitch motor units of cat ankle flexors: 1. Tripartite classification on basis of fatigability. Brain Res 134:35–46
Hamm TM, Koehler W, Stuart DG, Vanden Noven S (1985) Partitioning of monosynaptic Ia excitatory postsynaptic potentials in the motor nucleus of the cat. J Physiol (Lond) 369:397–398
Hoffer JA, Loeb GE, Sugano N, Marks WB, O'Donovan MJ, Pratt CA (1987) Cat hindlimb motoneurons during locomotion. III. Functional segregation in sartorius. J Neurophysiol 57:554–562
Hoffmann P, Illert M, Wiedemann E (1986) EMG pattern of cat forelimb muscles during target reaching and food taking movements. Neurosci Lett [Suppl] 22:S126
Kernell D, Ducati A, Sjöholm H (1975) Properties of motor units in the first deep lumbrical muscle of the cat's foot. Brain Res 98:37–55
Kernell D, Eerbeek O Verhey BA (1983a) Motor unit categorization on basis of contractile properties: an experimental analysis of the composition of the cat's M. Peroneus longus. Exp Brain Res 50:211–219
Kernell D, Eerbeek O, Verhey BA (1983b) Relation between isometric force and stimulus rate in cat's hindlimb motor units of different twitch contraction time. Exp Brain Res 50:220–227
McDonagh JC, Binder MD, Reinking RM, Stuart DG (1980) A commentary on muscle unit properties in cat hindlimb muscles. J Morphol 166:217–230
Norusis MJ (1988) SPSS/PC + Advanced statistics V2.0. SPSS Inc. Chicago
Reinking RM, Stephens JA, Stuart DG (1975) The motor units of cat medial gastrocnemius: problem of their categorisation on the basis of mechanical properties. Exp Brain Res 23:301–313
Reighard J, Jennings HS (1935) Anatomy of the cat. Holt, Rinehart and Winston, New York, pp 203–209
ter Haar Romeny BM, Denier van der Gon JJ, Gielen CCAM (1982) Changes in recruitment order of motor units in the human biceps muscle. Exp Neurol 78:360–368
Thomas JS, Schmidt EM, Hambrecht FT (1978) Facility of motor unit control during tasks defined directly in terms of unit behaviors. Exp Neurol 59:384–395
Vanden Noven S, Hamm TM, Stuart DG (1986) Partitioning of monosynaptic Ia excitatory postsynaptic potentials in the motor nucleus of the cat lateral gastrocnemius muscle. J Neurophysiol 55:569–586
van Zuylen EJ, Denier van der Gon JJ, Gielen CCAM, Jongen HAH (1986) Coordination of human arm muscle in flexion-extension and supination-pronation tasks. Part I: Description of activation patterns. Neurosci Lett [Suppl] 26:S84
Weeks OI, English AW (1985) Compartimentalization of the cat lateral gastrocnemius motor nucleus. J Comp Neurol 235:255–267
Windhorst U, Hamm TM, Stuart DG (1989) On the function of muscle and reflex partitioning. Behav Brain Sci 12:629–681
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fritz, N., Schmidt, C. Contractile properties of single motor units in two multi-tendoned muscles of the cat distal forelimb. Exp Brain Res 88, 401–410 (1992). https://doi.org/10.1007/BF02259115
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02259115