Abstract
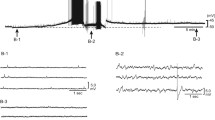
Previous studies in cats using isolated NaClCO2 perfusion of the lower brainstem demonstrated an intrinsic chemosensitivity of sympathoexcitatory bulbospinal neurones within the rostroventrolateral medulla (RVLM). In the present experiments, the effects of inhibitors of enzymatic and cellular systems, known to be involved in pH regulation, were investigated. Isolated perfusion of the lower brainstem with CO2-enriched solutions was performed and preganglionic sympathetic nerve activity (SNA) was recorded. Drugs were locally injected into the left RVLM with glass micropipettes. Perfusion of the RVLM with CO2enriched solutions over a period of 15 s induced a marked increase in SNA. The magnitude of absolute changes in SNA during perfusion depended on the level of basal SNA before perfusion. Microinjections of 4,4′-diisothiocyanatostilbene-2,2′-disulphonic acid (DIDS) and acetazolamide (ACZ) induced a marked rise in basal SNA, whereas diethylpyrocarbonate (DEPC) and ethylisopropylamiloride (EIPA) had no significant effect on basal SNA. After application of DIDS and DEPC, the peak change in SNA due to perfusion of the RVLM with CO2-enriched solutions was slightly diminished. Furthermore, neither ACZ nor EIPA produced any significant influence on the slope, peak change and time course of the increase in SNA compared with control perfusions. We conclude that the enzymatic and cellular carrier systems tested in this study are not or only slightly involved in central sympathetic chemosensitivity.
Similar content being viewed by others
References
Adams JM, Johnson NL (1990) Inhibiting carbonic anhydrase in brain tissue increases the respiratory response to rebreathing CO2. Brain Res 519:23–28
Amendt K, Czachurski J, Dembowsky K, Seller H (1978) Neurones within the “chemosensitive area” of the ventral surface of the brain stem which project to the intermediolateral column. Pflügers Arch 375:289–292
Coates EL, Li A, Nattie EE (1991) Acetazolamide on the ventral medulla of the cat increases phrenic output and delays the ventilatory response to CO2. J Physiol (Lond) 441:433–451
Durbin RP, Heinz E (1958) Electromotive chloride transport and gastric acid secretion in the frog. J Gen Physiol 41:1035–1047
Grinstein S, Rothstein A (1986) Mechanisms of regulation of the Na+/H+ exchanger. J Membr Biol 90:1–12
Hanson MA, Nye PCG, Torrance RW (1981) The exodus of an extracellular bicarbonate theory of chemoreception and the genesis of an intracellular one. In: Belmonte C, Pallot DJ, Acker H, Fidone S (eds) Arterial chemoreceptors. University of Leicester, Leicester, pp 403–416
Hanson MA, Nye PCG, Torrance RW (1981) The location of carbonic anhydrase in relation to the blood-brain barrier at the medullary chemoreceptors of the cat. J Physiol (Lond) 320:113–125
Housley GD, Sinclair JD (1988) Localization by kainic acid lesions of neurones transmitting the carotid chemoreceptor stimulus for respiration in rat. J Physiol (Lond) 406:99–114
Koepchen HP, Thurau K (1959) Über die Entstehungsbedingungen der atemsynchronen Schwankungen des Vagustonus (Respiratorische Arrhythmie). Pflügers Arch 269:10–30
König S, Seller H (1991) Historical development of current concepts on central chemosensitivity. Arch Ital Biol 129:223–237
König S, Offner B, Czachurski J, Seller H (1995) Changes in medullary extracellular pH, sympathetic and phrenic nerve activity during brain stem perfusion with CO2-enriched solution. J Auton Nerv Syst 51:67–75
Lioy F, Trzebski A (1984) Pressor effect of CO2 in the rat: different thresholds of central cardiovascular and respiratory responses to CO2. J Auton Nerv Syst 10:43–54
Loeschcke HH (1982) Central chemosensitivity and reaction therapy. J Physiol (Lond) 332:1–24
McAllen RM (1986) Identification and properties of sub-retrofacial neurones: a descending cardiovascular pathway in the cat. J Auton Nerv Syst 17:151–164
Millhorn DE (1986) Neural and circulatory interaction during chemoreceptor stimulation and cooling of ventral medulla in cats. J Physiol (Lond) 370:217–231
Murer H, Hopfer U, Kinne R (1976) Sodium/protein antiport in brush-border membrane vesicles isolated from rat small intestine and kidney. Biochem J 154:597–604
Nattie EE (1990) The alphastat hypothesis in respiratory control and acid-base balance. J Appl Physiol 69:1201–1207
Ridderstrale Y, Hanson M (1985) Histochemical study of the distribution of carbonic anhydrase in the cat brain. Acta Physiol Scand 124:557–564
Seller H, König St, Czachurski J (1990) Chemosensitivity of sympathoexcitatory neurones in the rostroventrolateral medulla of the cat. Pflügers Arch 416:735–741
Spalding BC, Taber B, Swift JG, Horowicz P (1991) Response of chloride efflux from skeletal muscle in Rana pipiens to changes of temperature and membrane potential and diethylpyrocarbonate treatment. J Membr Biol 123:223–233
Thomas RC (1976) Ionic mechanism of the H+ pump in a snail neurone. Nature 262:54–55
Trzebski A, Kubin L (1981) Is the central inspiratory activity responsible for PCO2 dependent drive of the sympathetic discharge? J Auton Nerv Syst 3:401–420
Weiner IW (1990) Diuretics and other agents employed in the mobilization of edema fluid. In: Goodman S and Gilman A (eds) The pharmacological basis of therapeutics. Pergamon, Oxford, pp 713–731
Wuttke WA, Walz W (1990) Sodium- and bicarbonate-independent regulation of intracellular pH in cultured mouse astrocytes. Neurosci Lett 117:105–110
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
König, S.A., Offner, B., Czachurski, J. et al. Effects of inhibitors of enzymatic and cellular pH-regulating systems on central sympathetic chemosensitivity. Pflugers Arch. 430, 690–696 (1995). https://doi.org/10.1007/BF00386163
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00386163