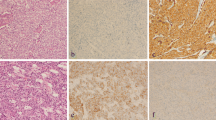
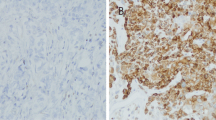
Abstract
Background
Basal-like breast cancer (BLBC) has a poor prognosis and is often identified by the triple-negative phenotype (TNP) and/or basal cytokeratins (CKs). Overexpression of mRNA for forkhead box C1 (FOXC1) transcription factor was recently identified as a pivotal prognostic biomarker of BLBC. We investigated the prognostic value of FOXC1 protein expression in invasive breast cancer and compared its prognostic significance to that of TNP and basal CKs.
Methods
Archived TNP specimens of primary invasive ductal breast cancer from 759 patients were examined by immunohistochemical staining for FOXC1, CK5/6, and CK14; prognostic significance was assessed using multivariate analyses. In addition, the impact of adding FOXC1 versus basal CKs to TNP-based BLBC assessment was assessed.
Results
FOXC1 protein expression was a significant predictor of overall survival on univariate (hazard ratio [HR] 3.364 95% confidence interval [CI] 1.758–6.438, P = 0.0002) and multivariate (HR 3.389 95% CI 1.928–7.645, P = 0.0001) analyses, despite its correlation with younger age (P = 0.0003). Interestingly, nodal status was not significant on multivariate analysis when FOXC1 expression status was included in the analysis. BLBC defined by TNP plus FOXC1 demonstrated superior prognostic relevance compared to BLBC defined by TNP or TNP plus basal CKs.
Conclusions
Immunohistochemical detection of FOXC1 expression in TNP invasive breast cancer is an independent prognostic indicator that is superior to conventional immunohistochemical surrogates of BLBC. Prospective validation is warranted to further define the diagnostic, prognostic, and predictive utility of FOXC1 in breast cancer management and clinical trial design.



Similar content being viewed by others
References
Perou CM, Sorlie T, Eisen MB, et al. Molecular portraits of human breast tumours. Nature. 2000;406(6797):747–52.
Dairkee SH, Puett L, Hackett AJ. Expression of basal and luminal epithelium-specific keratins in normal, benign, and malignant breast tissue. J Natl Cancer Inst. 1988;80:691–5.
Wetzels RHW, Holland R, Haelst UJGMv, et al. Detection of basement membrane components and basal cell keratin 14 in noninvasive and invasive carcinomas of the breast. Am J Pathol. 1989;134:571–9.
Bidard FC, Conforti R, Boulet T, et al. Does triple-negative phenotype accurately identify basal-like tumour? An immunohistochemical analysis based on 143 “triple-negative” breast cancers. Ann Oncol. 2007;18:1285–6.
Rakha EA, Tan DS, Foulkes WD, et al. Are triple-negative tumours and basal-like breast cancer synonymous? Breast Cancer Res. 2007;9:404.
Rakha E, Ellis I, Reis-Filho J. Are triple-negative and basal-like breast cancer synonymous? Clin Cancer Res. 2008;14:618.
Bertucci F, Finetti P, Cervera N, et al. How basal are triple-negative breast cancers? Int J Cancer. 2008;123:236–40.
Carey LA, Perou CM, Livasy CA, et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006;295:2492–502.
Ihemelandu CU, Leffall LD Jr, Dewitty RL, et al. Molecular breast cancer subtypes in premenopausal and postmenopausal African-American women: age-specific prevalence and survival. J Surg Res. 2007;143:109–18.
Ihemelandu CU, Naab TJ, Mezghebe HM, et al. Treatment and survival outcome for molecular breast cancer subtypes in black women. Ann Surg. 2008;247:463–9.
Dent R, Trudeau M, Pritchard KI, et al. Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res. 2007;13(15 Pt 1):4429–34.
Smid M, Wang Y, Zhang Y, et al. Subtypes of breast cancer show preferential site of relapse. Cancer Res. 2008;68:3108–14.
Rakha EA, Elsheikh SE, Aleskandarany MA, et al. Triple-negative breast cancer: distinguishing between basal and nonbasal subtypes. Clin Cancer Res. 2009;15:2302–10.
Nielsen TO, Hsu FD, Jensen K, et al. Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res. 2004;10:5367–74.
Cheang MC, Voduc D, Bajdik C, et al. Basal-like breast cancer defined by five biomarkers has superior prognostic value than triple-negative phenotype. Clin Cancer Res. 2008;14:1368–76.
Elsheikh SE, Green AR, Rakha EA, et al. Caveolin 1 and caveolin 2 are associated with breast cancer basal-like and triple-negative immunophenotype. Br J Cancer. 2008;99:327–34.
Rakha EA, Reis-Filho JS, Ellis IO. Basal-like breast cancer: a critical review. J Clin Oncol. 2008;26:2568–81.
Sorlie T, Perou CM, Tibshirani R, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA. 2001;98:10869–74.
Ray PS, Wang J, Qu Y, et al. FOXC1 is a potential prognostic biomarker with functional significance in basal-like breast cancer. Cancer Res. 2010;70:3870–6.
McShane LM, Altman DG, Sauerbrei W, et al. REporting recommendations for tumour MARKer prognostic studies (REMARK). Br J Cancer. 2005;93:387–91.
Yaziji H, Goldstein LC, Barry TS, et al. HER-2 testing in breast cancer using parallel tissue-based methods. JAMA. 2004;291:1972–7.
Laakso M, Tanner M, Nilsson J, et al. Basoluminal carcinoma: a new biologically and prognostically distinct entity between basal and luminal breast cancer. Clin Cancer Res. 2006;12(14 Pt 1):4185–91.
Bland JM, Altman DG. Survival probabilities (the Kaplan-Meier method). BMJ. 1998;317(7172):1572.
Bland JM, Altman DG. The logrank test. BMJ. 2004;328(7447):1073.
Akaike H. A new look at the statistical model identification. IEEE Trans Automatic Control. 1974;19:716–23.
Dejeux E, Ronneberg JA, Solvang H, et al. DNA methylation profiling in doxorubicin treated primary locally advanced breast tumours identifies novel genes associated with survival and treatment response. Mol Cancer. 2010;9:68.
Acknowledgment
P.S.R., S.P.B., J.W., and X.C. are named inventors on patent applications filed relevant to the role of FOXC1 in cancer. This work was supported by funding from QVC and the Fashion Footwear Association of New York Charitable Foundation, the Associates for Breast and Prostate Cancer Studies, the John Wayne Cancer Foundation, and the Avon Foundation. We thank Drs. John Martens and Silvana Martino for critical reading of the article.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Partha S. Ray and Sanjay P. Bagaria contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10434_2011_1657_MOESM2_ESM.eps
Flow diagram of patient identification, sample collection and tissue processing for immunohistochemical assessment of CK5/6, CK14 and FOXC1. Supplementary material 2 (EPS 681 kb)
10434_2011_1657_MOESM3_ESM.eps
Kaplan–Meier 10-year survival curves for breast cancer patients grouped according to molecular subtypes as assessed by standard immunohistochemistry. Three different cutoff scores of FOXC1 protein staining used to define BLBC are shown. Positive expression of FOXC1 was a significant predictor of overall survival, independent of cutoff scores and other standard clinicopathologic factors. 4NP denotes ER-PR-HER2-FOXC1-patients. Supplementary material 3 (EPS 785 kb)
10434_2011_1657_MOESM4_ESM.eps
Kaplan–Meier 10-year survival curves for breast cancer patients grouped according to 3-biomarker, 5-biomarker or 4-biomarker models, each defining BLBC with a different combination of biomarkers. The prognostic impact of immunohistochemical biomarker definition of BLBC is shown in red. 4NP denotes ER-PR-HER2-FOXC1-patients. 5NP denotes ER-PR-HER2-CK5/6-CK14-patients. Supplementary material 4 (EPS 778 kb)
Rights and permissions
About this article
Cite this article
Ray, P.S., Bagaria, S.P., Wang, J. et al. Basal-Like Breast Cancer Defined by FOXC1 Expression Offers Superior Prognostic Value: A Retrospective Immunohistochemical Study. Ann Surg Oncol 18, 3839–3847 (2011). https://doi.org/10.1245/s10434-011-1657-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-011-1657-8