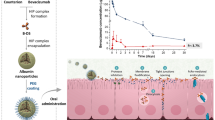
Abstract
Paliperidone (PPD) is the most recent second-generation atypical antipsychotic approved for the treatment of schizophrenia. An immediate release dose causes extrapyramidal side effects. In this work, a novel nanolipomer carrier system for PPD with enhanced intestinal permeability and sustained release properties has been developed and optimized. PPD was successfully encapsulated into a lipomer consisting of a specific combination of biocompatible materials including poly-ε-caprolactone as a polymeric core, Lipoid S75, and Gelucire® 50/13 as a lipid shell and polyvinyl alcohol as a stabilizing agent. The lipomer system was characterized by dynamic light scattering, TEM, DSC, and FTIR. An optimized lipomer formulation possessed a particle size of 168 nm, PDI of 0.2, zeta potential of −23 mV and an encapsulation efficiency of 87.27% ± 0.098. Stability in simulated gastrointestinal fluids investigated in terms of particle size, zeta potential, and encapsulation efficiency measurements ensured the integrity of the nanoparticles upon oral administration. PPD-loaded nanolipomers demonstrated a superior sustained release behavior up to 24 h and better ex vivo intestinal permeation for PPD compared to the corresponding polymeric and solid lipid nanoparticles and drug suspension. The in vitro hemocompatibility test on red blood cells revealed no hemolytic effect of PPD-loaded lipomers which reflects its safety. The elaborated nanohybrid carrier system represents a promising candidate for enhancing the absorption of PPD providing a 2.6-fold increase in the intestinal permeation flux compared to the drug suspension while maintaining a sustained release behavior. It is a convenient alternative to the commercially available dosage form of PPD.









Similar content being viewed by others
REFERENCES
Yang LP. Oral paliperidone. CNS Drugs. 2011;25(6):523–38.
Kumar S, Randhawa JK. Preparation and characterization of paliperidone loaded solid lipid nanoparticles. Colloids Surf, B. 2013;102:562–8.
Canuso CM, Youssef EA, Bossie CA, Turkoz I, Schreiner A, Simpson GM. Paliperidone extended-release tablets in schizophrenia patients previously treated with risperidone. Int Clin Psychopharmacol. 2008;23(4):209–15.
Wang D, Zhao J, Liu X, Sun F, Zhou Y, Teng L, et al. Parenteral thermo-sensitive organogel for schizophrenia therapy, in vitro and in vivo evaluation. Eur J Pharm Sci. 2014;60:40–8.
Patel RB, Patel MR, Bhatt KK, Patel BG. Paliperidone-loaded mucoadhesive microemulsion in treatment of schizophrenia: formulation consideration. J Pharm Innov. 2013;8(3):195–204.
Kanuganti S, Jukanti R, Veerareddy PR, Bandari S. Paliperidone-loaded self-emulsifying drug delivery systems (SEDDS) for improved oral delivery. J Dispersion Sci Technol. 2012;33(4):506–15.
Medina C, Santos‐Martinez M, Radomski A, Corrigan O, Radomski M. Nanoparticles: pharmacological and toxicological significance. Br J Pharmacol. 2007;150(5):552–8.
Hadinoto K, Sundaresan A, Cheow WS. Lipid-polymer hybrid nanoparticles as a new generation therapeutic delivery platform: a review. Eur J Pharm Biopharm. 2013;85(3):427–43.
Raemdonck K, Braeckmans K, Demeester J, De Smedt SC. Merging the best of both worlds: hybrid lipid-enveloped matrix nanocomposites in drug delivery. Chem Soc Rev. 2014;43(1):444–72.
Zhang LI, Zhang L. Lipid-polymer hybrid nanoparticles: synthesis, characterization and applications. Nano LIFE. 2010;01(01n02):163–73.
Rao S, Prestidge CA. Polymer-lipid hybrid systems: merging the benefits of polymeric and lipid-based nanocarriers to improve oral drug delivery. Expert Opin Drug Deliv. 2016;13(5):691–707.
Hallan SS, Kaur P, Kaur V, Mishra N, Vaidya B. Lipid polymer hybrid as emerging tool in nanocarriers for oral drug delivery. Artificial Cells, Nanomedicine, and Biotechnology. 2015:1–16.
Belletti D, Riva G, Tosi G, Forni F, Barozzi P, Luppi M, et al. Novel polymeric/lipidic hybrid systems (PLHs) for effective cidofovir delivery: preparation, characterization and comparative in vitro study with polymeric particles and liposomes. Int J Pharm. 2011;413(1–2):220–8.
Benival DM, Devarajan PV. Lipomer of doxorubicin hydrochloride for enhanced oral bioavailability. Int J Pharm. 2012;423(2):554–61.
Cheow WS, Hadinoto K. Factors affecting drug encapsulation and stability of lipid-polymer hybrid nanoparticles. Colloids Surf, B. 2011;85(2):214–20.
Cheow WS, Chang MW, Hadinoto K. The roles of lipid in anti-biofilm efficacy of lipid-polymer hybrid nanoparticles encapsulating antibiotics. Colloids Surf A:Physicochem Eng Asp. 2011;389(1–3):158–65.
Jain S, Valvi PU, Swarnakar NK, Thanki K. Gelatin coated hybrid lipid nanoparticles for oral delivery of amphotericin B. Mol Pharmaceutics. 2012;9(9):2542–53.
Peter Christoper GV, Vijaya Raghavan C, Siddharth K, Siva Selva Kumar M, Hari Prasad R. Formulation and optimization of coated PLGA - zidovudine nanoparticles using factorial design and in vitro in vivo evaluations to determine brain targeting efficiency. Saudi Pharm J. 2014;22(2):133–40.
Prego C, Torres D, Fernandez-Megia E, Novoa-Carballal R, Quiñoá E, Alonso M. Chitosan-PEG nanocapsules as new carriers for oral peptide delivery: effect of chitosan pegylation degree. J Controlled Release. 2006;111(3):299–308.
Freag MS, Elnaggar YSR, Abdallah OY. Development of novel polymer-stabilized diosmin nanosuspensions: in vitro appraisal and ex vivo permeation. Int J Pharm. 2013;454(1):462–71.
Cattani VB, Fiel LA, Jäger A, Jäger E, Colomé LM, Uchoa F, et al. Lipid-core nanocapsules restrained the indomethacin ethyl ester hydrolysis in the gastrointestinal lumen and wall acting as mucoadhesive reservoirs. Eur J Pharm Sci. 2010;39(1):116–24.
Khurana S, Jain N, Bedi P. Development and characterization of a novel controlled release drug delivery system based on nanostructured lipid carriers gel for meloxicam. Life Sci. 2013;93(21):763–72.
Fang RH, Aryal S, Hu C-MJ, Zhang L. Quick synthesis of lipid−polymer hybrid nanoparticles with low polydispersity using a single-step sonication method. Langmuir. 2010;26(22):16958–62.
Sinha V, Bansal K, Kaushik R, Kumria R, Trehan A. Poly-ϵ-caprolactone microspheres and nanospheres: an overview. Int J Pharm. 2004;278(1):1–23.
Jürgens K, Müller B. A new formulation concept for drugs with poor water solubility for parenteral application. Die Pharmazie-An Int J Pharm Sci. 2005;60(9):665–70.
Valencia PM, Basto PA, Zhang L, Rhee M, Langer R, Farokhzad OC, et al. Single-step assembly of homogenous lipid−polymeric and lipid−quantum dot nanoparticles enabled by microfluidic rapid mixing. ACS Nano. 2010;4(3):1671–9.
Alexis F, Pridgen E, Molnar LK, Farokhzad OC. Factors affecting the clearance and biodistribution of polymeric nanoparticles. Mol Pharmaceutics. 2008;5(4):505–15.
Polymeric nanomaterials. In: Kumar CSSR, editor. Nanomaterials for the Life Sciences: WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim; 2011.
Dash S, Murthy PN, Nath L, Chowdhury P. Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol Pharm. 2010;67:217–23.
Abdelwahed W, Degobert G, Fessi H. A pilot study of freeze drying of poly (epsilon-caprolactone) nanocapsules stabilized by poly (vinyl alcohol): formulation and process optimization. Int J Pharm. 2006;309(1):178–88.
Leimann FV, Biz MH, Kaufmann KC, Maia WJ, Honçalves OH, Cardozo Filho L, et al. Characterization of progesterone loaded biodegradable blend polymeric nanoparticles. Ciência Rural. 2015;45(11):2082–8.
Reis EFD, Campos FS, Lage AP, Leite RC, Heneine LG, Vasconcelos WL, et al. Synthesis and characterization of poly (vinyl alcohol) hydrogels and hybrids for rMPB70 protein adsorption. Mater Res. 2006;9(2):185–91.
Passerini N, Perissutti B, Moneghini M, Voinovich D, Albertini B, Cavallari C, et al. Characterization of carbamazepine-Gelucire 50/13 microparticles prepared by a spray‐congealing process using ultrasounds. J Pharm Sci. 2002;91(3):699–707.
Desai PP, Date AA, Patravale VB. Overcoming poor oral bioavailability using nanoparticle formulations—opportunities and limitations. Drug Discov Today: Technol. 2012;9(2):e87–95.
Balimane PV, Chong S, Morrison RA. Current methodologies used for evaluation of intestinal permeability and absorption. J Pharmacol Toxicol Methods. 2000;44(1):301–12.
Mathiowitz E, Jacob JS, Jong YS, Carino GP, Chickering DE, Chaturvedi P, et al. Biologically erodable microspheres as potential oral drug delivery systems. Nature. 1997;386(6623):410–4.
Aji Alex MR, Chacko AJ, Jose S, Souto EB. Lopinavir loaded solid lipid nanoparticles (SLN) for intestinal lymphatic targeting. Eur J Pharm Sci. 2011;42(1–2):11–8.
Chen C, Cheng YC, Yu CH, Chan SW, Cheung MK, Yu PH. In vitro cytotoxicity, hemolysis assay, and biodegradation behavior of biodegradable poly (3‐hydroxybutyrate)-poly (ethylene glycol)-poly (3‐hydroxybutyrate) nanoparticles as potential drug carriers. J Biomed Mater Res, Part A. 2008;87(2):290–8.
Nafee N, Schneider M, Schaefer UF, Lehr C-M. Relevance of the colloidal stability of chitosan/PLGA nanoparticles on their cytotoxicity profile. Int J Pharm. 2009;381(2):130–9.
Bender EA, Adorne MD, Colomé LM, Abdalla DS, Guterres SS, Pohlmann AR. Hemocompatibility of poly (ɛ-caprolactone) lipid-core nanocapsules stabilized with polysorbate 80-lecithin and uncoated or coated with chitosan. Int J Pharm. 2012;426(1):271–9.
Acknowledgements
The authors thank Gattefossé Company for providing the lipid used in this study and Lipoid Company for providing Lipoid S75.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Helal, H.M., Mortada, S.M. & Sallam, M.A. Paliperidone-Loaded Nanolipomer System for Sustained Delivery and Enhanced Intestinal Permeation: Superiority to Polymeric and Solid Lipid Nanoparticles. AAPS PharmSciTech 18, 1946–1959 (2017). https://doi.org/10.1208/s12249-016-0657-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-016-0657-1