Summary
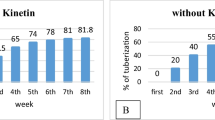
Jasmonic acid (JA) effects on in vitro tuberization of potato nodal explants cvs. Sangre and Russet Burbank were tested under liquid and solid media conditions and 0,8, and 16h photoperiod. Explants taken from stock plants grown on 2.5μM JA-supplemented medium tuberized first, particularly in darkness. The most pronounced benefits of the JA pretreatment were recorded under 16h photoperiod, which is known to inhibit tuberization. Cultivar Sangre benefited from the JA preconditioning of stock plants more than Russet Burbank. Russet Burbank required the JA supplement in tuberization media to reach the same degree of stimulation. Overall, microtubers produced either from JA preconditioned stock plants or on the JA-containing tuberization media were more uniform and larger than from other treatments. Eight hours photoperiod was by far the best treatment for the production of high-quality uniform microtubers. JA conditioning of stock plants prior to taking explants for tuberization is being proposed as a treatment enhancing the quality of microtubers.
Similar content being viewed by others
References
Coleman, W. K.; Coleman, S. E. Modification of potato microtuber dormancy during induction and growth in vitro and ex vitro. Am. J. Potato Res. 77:103–110; 2000.
Coltman, R.; Kostichka, C. Performance of minitubers produced from microshoots vs. microtubers. Am. Potato J. 70:803–804 (abstract); 1993.
Crowde, R. M. J.; Hand, D. J. Analysis of repeated measures, New York: Chapman and Hall; 1990.
Dobránszki, J.; Mandi, M. Induction of in vitro tuberization by short day period and dark treatment of potato shoots grown on hormone-free medium. Acta Biol. Hung. 44:411–420; 1993.
Estrada, R.; Tovar, P.; Dodds, J. H. Induction of in vitro tubers in a broad range of potato genotypes. Plant Cell Tiss. Organ Cult. 7:3–10; 1986.
Gopal, J.; Minocha, J. L.; Sidhu, J. S. Comparative performance of potato crops raised from microtubers induced in the dark versus microtubers induced in light. Potato Res. 40:407–412; 1997.
Gregory, L. E. Some factors for tuberization in the potato plant. Am. J. Bot. 43:281–288; 1956.
Hussey, G.; Stacey, N. J. In vitro propagation of potato (Solanum tuberosum L.). Ann. Bot. 48:787–796; 1981.
Hussey, G.; Stacey, N. J. Factors affecting the formation of in vitro tubers of potato (Solanum tuberosum L.). Ann. Bot. 53:565–578; 1984.
Jackson, S. D. Multiple signalling pathways control tuber induction in potato. Plant Physiol. 119:1–8; 1999.
Koda, Y. Possible involvement of jasmonates in various morphogenic events. Physiol. Plant. 98:407–412; 1997.
Koda, Y.; Kikuta, Y.; Tazaki, H.; Tsuhino, Y.; Sakamura, S.; Yoshihara, T. Potato tuber-inducing activities of jasmonic acid and related compounds. Phytochemistry 30:1435–1438; 1991.
Kreft, S.; Ravinkar, M.; Mesko, P.; Pungercar, J.; Umek, A.; Kregar, I.; Strukelj, B. Jasmonic acid inducible aspartic proteinase inhibitors from potato. Phytochemistry 44:1001–1006; 1997.
Lê, C. L. In vitro microtuberization: an evaluation of culture conditions for the production of virus-free potatoes. Potato Res. 42:489–498; 1999.
Leclerc, Y.; Donnelly, D. J.; Coleman, W. K.; King, R. R. Microtuber dormancy in three potato cultivars. Am. Potato J. 72:215–223; 1995.
Lillo, C. A simple two-phase system for efficient in vitro tuberization in potato. Norwegian J. Agric. Sci. 3:23–27; 1989.
Lommen, W. J. M. Basic studies on the production and performance of potato minitubers. PhD thesis. Agricultural University of Wageningen, The Netherlands; 1995:181.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–497; 1962.
Pelacho, A. M.; Mingo-Castel, A. M. Jasmonic acid induces tuberization of potato stolons cultured in vitro. Plant Physiol. 97:1253–1255; 1991.
Pruski, K. Micropropagation technology in early phases of commercial seed potato production. PhD thesis, Agricultural University of Wageningen. The Netherlands; 2001:166.
Pruski, K.; Nowak, J.; Lewis, T. Jasmonates and photoperiod effect on microtuber production in two potato cultivars. In Vitro Cell Dev. Biol. Plant 29:69 (abstract); 1993.
Ranalli, P. Innovative propagation methods in seed tuber multiplication programmes. Potato Res. 40:439–453; 1997.
Ravnikar, M.; Vilhar, B.; Gogala, N. Stimulatory effects of jasmonic acid on potato node and protoplast culture. J. Plant Growth Reg. 11:29–33; 1992.
SAS Institute Inc.: SAS/STAT software: changes and enhancements through release 6.11. Cary, NC: SAS Institute Inc.; 1996.
Struik, P. C.; Boon, E. J.; Vreugdenhil, D. Effects of extracellular extracts from leaves on the tuberization of cuttings of potato (Solanum tuberosum L.). Plant Physiol. 84:214–217; 1987.
Struik, P. C.; Lommen, W. J. M. Production, storage and use of micro- and minitubers. Proceedings of the 11th Triennial Conference of the European Association for Potato Research (EAPR), Edinburgh, UK; 1990:122–133.
Struik, P. C.; Wiersema, S. C. Seed potato technology. The Netherlands: Wageningen Pers; 1999:383.
Tábori, K. M.; Dobránszki, J.; Ferenczy, A. Some sprouting characteristics of microtubers. Potato Res. 42:611–617; 1999.
Teisson, C.; Alvard, D. In vitro production of microtubers in liquid medium using temporary immersion. Potato Res. 42:499–504; 1999.
Van den Berg, J. H.; Ewing, E. E. Jasmonates and their role in plant growth and development with special reference to the control of potato tuberization: a review. Am. Potato J. 68:781–794; 1991.
Xu, X.; Vreugdenhil, D.; van Lammeren, A. A. M. Cell division and cell enlargement during potato tuber formation: a comparison of in vitro and in vivo tuber development. J. Exp. Bot. 49:573–582; 1998.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pruski, K., Astatkie, T. & Nowak, J. Jasmonate effects on in vitro tuberization and tuber bulking in two potato cultivars (Solanum tuberosum L.) under different media and photoperiod conditions. In Vitro Cell.Dev.Biol.-Plant 38, 203–209 (2002). https://doi.org/10.1079/IVPIVP2001265
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1079/IVPIVP2001265