Abstract
Introduction
An important goal in drug regulation is understanding serious safety issues with new drugs as soon as possible. Achieving this goal requires us to understand whether information provided during the Food and Drug Administration (FDA) drug review can predict serious safety issues that are usually identified after the product is approved. However, research on this topic remains understudied. In this paper, we examine whether any pre-marketing drug characteristics are associated with serious post-marketing safety actions.
Methods
We study this question using an internal FDA database containing every new small molecule drug submitted to the FDA’s Center for Drug Evaluation and Research (CDER) on or after November 21, 1997, and approved and commercially launched before December 31, 2009. Serious post-marketing safety actions include whether these drugs ever experienced either a post-marketing boxed warning or a withdrawal from the market due to safety concerns. A random effects logistic regression model was used to test whether any pre-marketing characteristics were associated with either post-marketing safety action.
Results
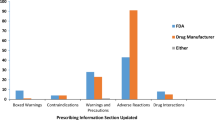
A total of 219 new molecular entities were analyzed. Among these drugs, 11 experienced a safety withdrawal and 30 received boxed warnings by July 31, 2016. Contrary to prevailing hypotheses, we find that neither clinical trial sample sizes nor review time windows are associated with the addition of a post-marketing boxed warning or safety withdrawal. However, we do find that new drugs approved with either a boxed warning or priority review are more likely to experience post-marketing boxed warnings. Furthermore, drugs approved with boxed warnings tend to receive post-marketing boxed warnings resulting from new safety information that are unrelated to the original warning. Drugs approved with a boxed warning are 3.88 times more likely to receive a post-marketing boxed warning, while drugs approved with a priority review are 3.51 times more likely to receive a post-marketing boxed warning.
Conclusion
Although drugs approved with a boxed warning or priority review are more likely to experience serious post-marketing safety events, other information provided during the FDA drug review that is easy to quantify is generally not associated with post-marketing safety events. It appears that these post-marketing events are not discernible during a pre-marketing review and therefore might not be avoidable using current review data.
Similar content being viewed by others
References
Duijnhoven R, Straus S, Raine J, Hoes A, De Bruin M. Number of patients studied prior to approval of new medicines: a database analysis. PLoS Med. 2013;10(3):e1001407.
Reed S, Anstrom K, Sells D, Califf R, Schulman K. Use of larger versus smaller drug-safety databases before regulatory approval: the trade-offs. Health Affairs. 2008;27(5):360–70.
Olson M. Pharmaceutical policy and the safety of new drugs. J Law Econ. 2002;45(2):615–42.
Olson M. Are novel drugs more risky for patients than less novel drugs? J Health Econ. 2004;23(6):1135–58.
Olson M. The risk we bear: the effects of review speed and industry user fees on new drug safety. J Health Econ. 2008;27(2):175–200.
Grabowski H, Wang Y. Do faster Food and Drug Administration drug reviews adversely affect patient safety? An analysis of the 1992 Prescription Drug User Fee Act. J Law Econ. 2008;51(2):377–406.
Berndt E, Gottschalk A, Philipson T, Strobeck M. Industry funding of the FDA: effects of PDUFA on approval times and withdrawal rates. Nat Rev Drug Discov. 2005;4(7):545–54.
Carpenter D, Zucker E, Avorn J. Drug-review deadlines and safety problems. N Engl J Med. 2008;358:1354–61.
Carpenter D, Chattopadhyay J, Moffitt S, Nall C. The complications of controlling agency time discretion: FDA review deadlines and post-market drug safety. Am J Political Sci. 2012;56(1):98–114.
Guidance for Industry Expedited Programs for Serious Conditions—Drugs and Biologics. U.S. Food and Drug Administration Center for Drug Evaluation and Research. http://www.fda.gov/downloads/Drugs/Guidances/UCM358301.pdf. Accessed 31 Jan 2017.
CDER 21st Century Review Process Desk Reference Guide. U.S. Food and Drug Administration Center for Drug Evaluation and Research. http://www.fda.gov/downloads/AboutFDA/CentersOffices/CDER/ManualofPoliciesProcedures/UCM218757.htm. Accessed 5 Oct 2016.
Lester J, Neyarapally G, Lipowski E, Graham C, Hall M, Dal Pan G. Evaluation of FDA safety-related drug label changes in 2010. Pharmacoepidemiol Drug Saf. 2013;22(3):302–5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No sources of funding were used to assist in the preparation of this study.
Conflict of interest
Andreas Schick, Kathleen Miller, Mike Lanthier, Gerald Dal Pan, and Clark Nardinelli are employees of the US Food and Drug Administration. The authors have no conflicting interests to declare. The views expressed in this article are those of the authors and not necessarily those of the US Food and Drug Administration or the Center for Drug Evaluation and Research.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Schick, A., Miller, K.L., Lanthier, M. et al. Evaluation of Pre-marketing Factors to Predict Post-marketing Boxed Warnings and Safety Withdrawals. Drug Saf 40, 497–503 (2017). https://doi.org/10.1007/s40264-017-0526-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40264-017-0526-1