Abstract
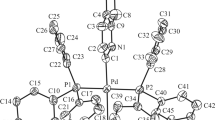
The title complex [Pd(Me2bqb)] (1), [Me2bqb2– = 1,2-bis(quinoline-2-carboxamide)-4,5-dimethyl-benzene dianion], has been synthesized and characterized by elemental analyses and spectroscopic methods, and the crystal and molecular structure of [Pd(Me2bqb)] has been determined by X-ray crystallography. The complex exhibits distorted square-planar PdN4 coordination geometry with two short and two long Pd–N bonds (Pd–N ~1.957 and ~2.095 Å, respectively). In addition to the molecular geometry from X-ray experiment, theoretical studies have been carried out on the structure of the complex at the density functional theory (DFT-B3LYP) level in conjunction with effective core potential basis set (LANL2DZ) for Pd atom and 6-311++G(d,p) basis set for N, O, C and H atoms. Electrochemical studies in CH2Cl2 solution revealed A reversible redox process corresponding to the PdII/PdIII couple with E 1/2 at 0.924 V (vs. SCE).







Similar content being viewed by others
References
O. Belda, C. Moberg, Coord. Chem. Rev. 249, 727 (2005)
R. Del Litto, V. Benessere, F. Ruffo, C. Moberg, Eur. J. Org. Chem. 1352 (2009)
S.H. Lee, J.H. Han, H. Kwak, S.J. Lee, E.Y. Lee, H.J. Kim, J.H. Lee, C. Bae, S.N. Lee, Y. Kim, C. Kim, Chem. Eur. J. 13, 9393 (2007)
J. Singh, G. Hundal, R. Gupta, Eur. J. Inorg. Chem. 2052 (2008)
D.-H. Lee, J.H. Lee, B.K. Park, E.Y. Kim, Y. Kim, C. Kim, I.-M. Lee, Inorg. Chim. Acta 362, 5097 (2009)
P. Srinivas, P.R. Likhar, H. Maheswaran, R. Sridhar, K. Ravikumar, M.L. Kantam, Chem. Eur. J. 15, 1578 (2009)
S.L. Jain, P. Bhattacharyya, H.L. Milton, A.M.Z. Slawin, J.A. Crayston, J.D. Woollins, Dalton Trans. 862 (2004)
R. Lescouëzec, J. Vaissermann, M. Toma, R. Carrasco, F. Lloret, M. Julve, Inorg. Chem. 43, 2234 (2004)
Z.-H. Ni, H.-Z. Kou, L.-F. Zhang, W.-W. Ni, Y.-B. Jiang, A.-L. Cui, J. Ribas, O. Sato, Inorg. Chem. 44, 9631 (2005)
J.I. Kim, H.S. Yoo, E.K. Koh, H.C. Kim, C.S. Hong, Inorg. Chem. 46, 8481 (2007a)
J.I. Kim, H.S. Yoo, E.K. Koh, C.S. Hong, Inorg. Chem. 46, 10461 (2007b)
N. Alizadeh, H. Teymourian, M. Aghamohammadi, S. Meghdadi, M. Amirnasr, IEEE Sens J 7, 1727 (2007)
H.A. Zamani, M.R. Ganjali, P. Norouzi, S. Meghdadi, J. Appl. Electrochem. 37, 853 (2007)
B. Rezaei, S. Meghdadi, V. Nafisi, Sens. Actuators B121, 600 (2007)
B. Rezaei, S. Meghdadi, R. Fazel Zarandi, J. Hazard. Mater. 153, 179 (2008)
A.K. Patra, M.J. Rose, K.A. Murphy, M.M. Olmstead, P.K. Mascharak, Inorg. Chem. 43, 4487 (2004)
S.R. Collinson, J.H.R. Tucker, T. Gelbrich, M.B. Hursthouse, Chem. Commun. 555 (2001)
I. Huc, M.J. Krische, D.P. Funeriu, J-M. Lehn, Eur. J. Inorg. Chem. 1415 (1999)
M.J. Chmielewski, J. Jurczak, Chem. Eur. J. 11, 6080 (2005)
J. Zhang, Q. Liu, C. Duan, Y. Shao, J. Ding, Z. Miao, X.-Z. You, Z. Guo, J. Chem. Soc. Dalton Trans. 4, 591 (2002)
W. Jacob, R. Mukherjee, Inorg. Chim. Acta 361, 1231 (2008)
D.H. Lee, J.Y. Lee, J.Y. Ryu, Y. Kim, C. Kim, I.M. Lee, Bull. Korean Chem. Soc. 27, 1031 (2006a)
D.H. Lee, C. Kim, I.M. Lee, Stud. Surf. Sci. Catal. 161, 59 (2006b)
S. Meghdadi, H.R. Khavasi, S. Nalchigar, Acta Crystallogr. E 62, o5492 (2006a)
S. Meghdadi, M. Amirnasr, V. Langer, A. Zamanpoor, Can. J. Chem. 84, 971 (2006b)
S. Meghdadi, M. Amirnasr, M.H. Habibi, A. Amiri, V. Ghodsi, A. Rohani, R.W. Harrington, W. Clegg, Polyhedron 27, 2771 (2008)
A. Amiri, M. Amirnasr, S. Meghdadi, K. Mereiter, V. Ghodsi, A. Gholami, Inorg. Chim. Acta 362, 3934 (2009)
S. Meghdadi, M. Amirnasr, K. Mereiter, A. Amiri, V. Ghodsi, Inorg. Chim. Acta 363, 1587 (2010a)
S. Meghdadi, K. Mereiter, A. Amiri, N.S. Mohammadi, F. Zamani, M. Amirnasr, Polyhedron 29, 2225 (2010b)
N.G. Connelly, W.E. Geiger, Chem. Rev. 96, 877 (1996)
Siemens Analytical X-ray Instruments Inc. SMART and SAINT [computer programs] (Siemens Analytical X-ray Instruments Inc., Madison, 1995)
G.M. Sheldrick. SADABS. Version 2.10 [computer program]. University of Göttingen, Göttingen, Germany, 2003
G.M. Sheldrick, Bruker Analytical X-ray Systems. SHELXTL. Version 6.14 [computer program]. Acta Crystallogr. A 64, 112 (2008)
A. Bondi, J. Phys. Chem. 68, 441 (1964)
M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery, Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, N.J. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, Ö. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski, D.J. Fox, Gaussian Development Version, Revision B.01 (Gaussian, Inc., Wallingford, 2009)
H. Nakatsuji, Chem. Phys. Lett. 59, 362 (1978)
S. Huzinaga, J. Andz elm, M. Klobukowski, E. Radz io-Andz elm, Y. Sakai, H. Tatewaki, Gaussian Basis Sets for Molecular Calculations (Elsevier, New York, 1984)
S.K. Dutta, U. Beckmann, E. Bill, T. Weyhermuller, K. Wieghardt, Inorg. Chem. 39, 3355 (2000)
I.O. Koshevoy, P. Lahuerta, M. Sanaú, M.A. Ubeda, A. Doménech, Dalton Trans. 5536 (2006)
N.R. Deprez, M.S. Sanford, J. Am. Chem. Soc. 131, 11234 (2009)
Acknowledgments
Partial support of this work by the Isfahan University of Technology Research Council is gratefully acknowledged. Special thanks are also due to Professor Peter C. Ford for providing the SM and MA with the necessary instruments and CHN facilities through the National Science Foundation grant (NSF-CHE-0749524).
Author information
Authors and Affiliations
Corresponding author
Additional information
M. Amirnasr is on sabbatical leave from the Department of Chemistry, Isfahan University of Technology, Isfahan, Iran.
Electronic supplementary material
Below is the link to the electronic supplementary material.
13738_2011_8_MOESM1_ESM.txt
Supplementary crystallographic data for 1 can be obtained free of charge via http://www.ccdc.cam.ac.uk/conts/retrieving.html, or from the Cambridge Crystallographic Data Centre, 12 Union Road, Cambridge CB2 1EZ, UK; fax: (+44) 1223-336-033; or e-mail: deposit@ccdc.cam.ac.uk (TXT 22 kb)
Rights and permissions
About this article
Cite this article
Meghdadi, S., Langer, V., Farrokhpour, H. et al. Synthesis, X-ray structure, electrochemistry, and theoretical studies of palladium(II) complex with a tetradentate bis(quinoline-2-carboxamide) ligand. J IRAN CHEM SOC 9, 85–92 (2012). https://doi.org/10.1007/s13738-011-0008-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-011-0008-9