Abstract
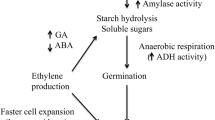
Direct-seeding cultivation of rice is increasing in Asia instead of transplanting system, because of its lower cost and operational simplicity. Low-temperature germinability (LTG) and anaerobic germinability (AG) are important characters for breeding of varieties for wide-spread adaptation of direct-seeding cultivation in rice. This study was performed to evaluate LTG and AG of seven rice varieties and identify varieties with strong germinability on both low-temperature and anaerobic conditions. The mean germination rate and germination vigor of seven varieties were 51.7% and 6.0 under low-temperature condition, respectively. Among these varieties, Cheongcheongjinmi and Hwanggeumnodeul had the highest germination rate of 80%, indicating that Cheongcheongjinmi and Hwanggeumnodeul have a good LTG. In anaerobic conditions, the germination rate and coleoptile length for all varieties were 47.6% and 3.2 cm, respectively. Of them, the highest germination rate and coleoptile length were observed in Subo and Hopum, respectively, suggesting that these two varieties are tolerant to anaerobic during germination stage. Molecular evaluation by SDS-PAGE revealed that the protein patterns differed at 50 kDa, 40 kDa, and 22 kDa between low-temperature and anaerobic conditions. Varieties identified as good LTG or AG in this study may be used for developing new direct-seeding rice cultivars through pyramiding these traits in the breeding program.
Similar content being viewed by others
References
Adachi Y, Sugiyama M, Sakagami JI, Fukuda A, Ohe M, Watanabe H. 2015. Seed germination and coleoptile growth of new rice lines adapted to hypoxic conditions. Plant Prod. Sci. 18: 471–475
Alpi A, Beevers H. 1983. Effects of O2 concentration on rice seedlings. Plant Physiol. 71: 30–34
Angaji S, Septiningsih EM, Mackill DJ, Ismail AM. 2010. QTLs associated with tolerance of flooding during germination in rice (Oryza sativa L.). Euphytica 172: 159–168
Baek MK, Shin WC, Ko JC, Kim BK, Nam JK, Kim KY, et al. 2013. A new mid-late maturing rice variety ⌈Subo⌋ with a good grain quality and for direct seeding. The Korean Society of Breeding Science Symposium, pp. 44–44
Bailey-Serres J, Chang R. 2005. Sensing and signaling in response to oxygen deprivation in plants and other organisms. Ann. Bot. 96: 507–518
Bradford MM. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254
Fujino K, Matsuda Y. 2010. Genome-wide analysis of genes targeted by qLTG3-1 controlling low-temperature germinability in rice. Plant Mol. Biol. 72: 137–152
Fujino K, Sekiguchi H, Matsuda Y, Sugimoto K, Ono K, Yano M. 2008. Molecular identification of a major quantitative trait locus, qLTG3-1, controlling low-temperature germinability in rice. Proc. Natl. Acad. Sci. USA. 105: 12623–12628
Fujino K, Sekiguchi H. 2011. Origins of functional nucleotide polymorphisms in a major quantitative trait locus, qLTG-3-1, controlling low-temperature germinability in rice. Plant Mol. Biol. 75: 1–10
Fukuda A, Kataoka T, Shiratsuchi H, Fukushima A, Yamaguchi H, Mochida H, Ogiwara H. 2014. QTLs for seedling growth of direct seeded rice under submerged and low temperature conditions. Plant Prod. Sci. 17: 41–46
Guglielminetti L, Busilacchi Ham Alpi A. 2000. Effect of anoxia on α-amylase induction in maize caryopsis. J. Plant Res. 112: 353–359
Hwang YS, Thomas BR, Rodriguez RL. 1999. Differential expression of rice α-amylase genes during seedling development under anoxia. Plant Mol. Biol. 40: 911–920
Hyun DY, Choi YM, Lee S, Lee MC, Oh S, Tai TH. 2015a. Selection of tolerant rice germplasm through phenotypic and genotypic evaluation for germination under low temperatures. Plant Breed. Biotech. 3: 253–263
Hyun DY, Lee GA, Kang MJ, Burkart-Waco D, Kim SI, Kim JY, Lee MC, Gwag JG, Kim YG, Tai TH. 2015b. Development of low-temperature germinability markers for evaluation of rice (Oryza sativa L.) germplasm. Mol. Breed. 35: 104
Ismail AM, Ella ES, Vergara GV, Mackill DJ. 2009. Mechanisms associated with tolerance to flooding during germination and early seedling growth in rice (Oryza sativa L.). Ann. Bot. 103: 197–209
Jiang L, Hou M, Wang C, Wan J. 2004. Quantitative trait loci and epistatic analysis of seed anoxia germinability in rice (Oryza sativa). Rice Sci. 11: 238–244
Jiang L, Liu S, Hou M, Tang J, Chen L, Zhai H, Wan J. 2006. Analysis of QTLs for seed low temperature germinability and anoxia germinability in rice (Oryza sativa L.). Field Crops Res. 98: 68–75
Kazemitabar SK, Tomsett AB, Collin HA, Wilkinson MC, Jones MG. 2003. Effect of short term cold stress on rice seedlings. Euphytica 129: 193–200
Ko JC, Baek MK, Kim WJ, Shon JY, Ha KY, Kang HJ, Shin MS, Ko JK. 2011. Analysis of low temperature germination ratio in rice cultivars for breeding of direct seeding variety. Korean J. Intl. Agric. 23: 89–97
Ko JC, Kim BK, Nam JK, Baek MG, Ha KY, Kim KY, et al. 2008. A medium-late maturing new rice cultivar with high grain quality, multi-disease resistance, adaptability to direct seeding and transplanting cultivation, “Hopum”. Korean J. Breed. Sci. 40: 533–536
Komatsu S, Kuji R, Nanjo Y, Hiraga S, Furukawa K. 2012. Comprehensive analysis of endoplasmic reticulum-enriched fraction in root tips of soybean under flooding stress using proteomics techniques. J. Proteome Res. 77: 531–560
Lee J, Lee W, Kwon SW. 2015. A quantitative shotgun proteomics analysis of germinated rice embryos and coleoptiles under low-temperature conditions. Proteome Sci. 13: 27. doi:10.1186/s12953-015-0082-5
Manangkil OE, Vu HTT, Yoshida S, Mori N, Nakamura C. 2008. A simple, rapid and reliable bioassay for evaluating seedling vigor under submergence in indica and japonica rice (Oryza sativa L.). Euphytica 163: 267–274
Nanjo Y, Skultety L, Uvackova L, Klubicova K, Hajduch M, Komatsu S. 2012. Mass spectrometry-based analysis of proteomic changes in the root tips of flooded soybean seedlings. J. Proteome Res. 11: 372–385
Osada A. 1982. Differences in germination, early growth and respiration between japonica and indica rice under low oxygen conditions. Japan. J. Trop. Agr. 26: 93–100
Perata P, Alpi A. 1993. Plant responses to anaerobiosis. Plant Sci. 93: 1–17
Perveen S, Shinwari KI, Jan M, Malook I, Rehman S, Khan M, Jamil M. 2013. Low temperature stress induced in biochemical parameters, protein banding pattern and expression of Zat12 and Myb genes in rice seedling. J. Stress Physiol. Biochem. 9: 193–206
R Development Core Team. 2012. R: a language and environment for statistical computing. R Foundation for Statistical Compuing, Vienna, Austria. http://www.R-project.org/
Rao AN, Johnson DE, Sivaprasad B, Ladha JK, Mortimer AM. 2007. Weed management in direct-seeded rice. In: LS Donalded, Adv. Agron. Academic Press, USA, vol. 93, pp. 153–255
RDA. 2012. Standard protocol of research method and analysis. Rural Development Administration, Suwon, p. 322
Redoña ED, Mackill DJ. 1996. Genetic variation for seedlingvigour traits in rice. Crop Sci. 36: 285–290
Satoh T, Tezuka K, Kawamoto T, Matsumoto S, Satoh-Nagasawa N, Ueda K, Sakurai K, Watanabe A, Takahashi H, Akagi H. 2016. Identification of QTLs controlling low-temperature germination of the East European rice (Oryza sativa L.) variety Maratteli. Euphytica 207: 245–254
Septiningsih EM, Sanchez DL, Singh N, Sendon PMD, Pamplona AM, Heuer S, Mackill DJ. 2012. Identifying novel QTLs for submergence tolerance in rice cultivars IR72 and Madabaru. Theor. Appl. Genet. 124: 867–874
Seshu DV, Krishnasamy V, Siddique SB. 1988. Seed vigor in rice. In: Rice seed health. International Rice Research Institute, Manila, pp. 315–329
Vriezen WH, Zhou Z, VD Straeten D. 2003. Regulation of submergence-induced enhanced shoot elongation in Oryza sativa L. Ann. Bot. 91: 263–270
Xu K, Xia X, Fukao T, Canlas P, Maghirang-Rodriguez R, Heueer S, Ismail AM, Bailey-Serres J, Ronald PC, Mackill DJ. 2006. Sub1A is an ethylene response factor-like gene that confers submergence tolerance to rice. Nature 442: 705–708
Yamauchi M, Aguilar AM, Vaughan DA, Seshu DV. 1993. Rice germplasm suitable for direct sowing under flooded soil surface. Euphytica 67: 177–184
Yamauchi M, Herradura PS, Aguilar AM. 1994. Genotype difference in rice postgermination growth under hypoxia. Plant Sci. 100: 105–113
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hyun, D.Y., Oh, M., Choi, YM. et al. Morphological and molecular evaluation for germinability in rice varieties under low-temperature and anaerobic conditions. J. Crop Sci. Biotechnol. 20, 21–27 (2017). https://doi.org/10.1007/s12892-016-0083-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12892-016-0083-1