Abstract
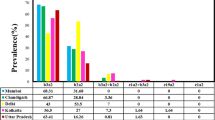
The BCR–ABL1 fusion gene is the driver mutation of Philadelphia chromosome-positive chronic myeloid leukemia (CML). Its expression level in CML patients is monitored by a real-time quantitative polymerase chain reaction defined by the International Scale (qPCRIS). BCR–ABL1 has also been found in asymptomatic normal individuals using a non-qPCRIS method. In the present study, we examined the prevalence of BCR–ABL1 in a normal population in southern Sarawak by performing qPCRIS for BCR–ABL1 with ABL1 as an internal control on total white blood cells, using an unbiased sampling method. While 146 of 190 (76.8%) or 102 of 190 (53.7%) samples showed sufficient amplification of the ABL1 gene at > 20,000 or > 100,000 copy numbers, respectively, in qPCRIS, one of the 190 samples showed amplification of BCR–ABL1 with positive qPCRIS of 0.0023% and 0.0032% in two independent experiments, the sequence of which was the BCR–ABL1 e13a2 transcript. Thus, we herein demonstrated that the BCR–ABL1 fusion gene is expected to be present in approximately 0.5–1% of normal individuals in southern Sarawak.


Similar content being viewed by others
References
Chandra HS, Heisterkamp NC, Hungerford A, Morrissette JJ, Nowell PC, Rowley JD, et al. Philadelphia chromosome symposium: Commemoration of the 50th anniversary of the discovery of the Ph chromosome. Cancer Genet. 2011;204(4):171–9.
Nowell PC, Hungerford DA. Chromosome studies on normal and leukemic human leukocytes. J Natl Cancer Inst. 1960;25:85–109.
Rowley JD. Citation classic. Curr Content/Life Sci. 1988;1988:16.
Druker BJ, Tamura S, Buchdunger E, Ohno S, Segal GM, Fanning S, et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr–Abl positive cells. Nat Med. 1996;2(5):561–6.
Kantarjian H, O’Brien S, Jabbour E, Garcia-Manero G, Quintas-Cardama A, Shan J, et al. Improved survival in chronic myeloid leukemia since the introduction of imatinib therapy: a single-institution historical experience. Blood. 2012;119(9):1981–7.
Baccarani M, Saglio G, Goldman J, Hochhaus A, Simonsson B, Appelbaum F, et al. Evolving concepts in the management of chronic myeloid leukemia: recommendations from an expert panel on behalf of the European LeukemiaNet. Blood. 2006;108(6):1809–20.
Hughes T, Deininger M, Hochhaus A, Branford S, Radich J, Kaeda J, et al. Monitoring CML patients responding to treatment with tyrosine kinase inhibitors: review and recommendations for harmonizing current methodology for detecting BCR–ABL transcripts and kinase domain mutations and for expressing results. Blood. 2006;108(1):28–37.
Vardiman JW, Melo JV, Baccarani M, Thiele J. Chronic myeloid leukemia, BCR–ABL1-positive. In: Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri AS, et al., editors. WHO classification of tumors of hematopoietic and lymphoid tissues. 4th ed. Lyon: IARC Press; 2017. p. 32–7.
Kuan JW, Su AT, Leong CF, Osato M, Sashida G. Systematic review of pre-clinical chronic myeloid leukaemia. Int J Hematol. 2018;108(5):465–84.
Kuan JW, Su AT, Leong CF, Osato M, Sashida G. Systematic review of normal subjects harbouring BCR–ABL1 fusion gene. Acta Haematol. 2019;9:1–16.
Cross NC, White HE, Colomer D, Ehrencrona H, Foroni L, Gottardi E, et al. Laboratory recommendations for scoring deep molecular responses following treatment for chronic myeloid leukemia. Leukemia. 2015;29(5):999–1003.
Department of Statistics. Population distribution and basic demographic characteristics 2010. Population and housing census of Malaysia. Kuala Lumpur: Department of Statistics, Malaysia; 2011.
Hochhaus A, Saglio G, Hughes TP, Larson RA, Kim DW, Issaragrisil S, et al. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia. 2016;30(5):1044–54.
Kuan JW, Melaine Michael S. The epidemiology of chronic myeloid leukaemia in southern Sarawak, Borneo Island. Med J Malaysia. 2018;73(2):78–85.
Steensma DP, Bejar R, Jaiswal S, Lindsley RC, Sekeres MA, Hasserjian RP, et al. Clonal hematopoiesis of indeterminate potential and its distinction from myelodysplastic syndromes. Blood. 2015;126(1):9–16.
Bose S, Deininger M, Gora-Tybor J, Goldman JM, Melo JV. The presence of typical and atypical BCR–ABL fusion genes in leukocytes of normal individuals: biologic significance and implications for the assessment of minimal residual disease. Blood. 1998;92(9):3362–7.
Ismail SI, Naffa RG, Yousef AMF, Ghanim MT. Incidence of bcr–abl fusion transcripts in healthy individuals. Mol Med Rep. 2014;9(4):1271–6.
Greenberg PL, Tuechler H, Schanz J, Sanz G, Garcia-Manero G, Solé F, et al. Revised international prognostic scoring system for myelodysplastic syndromes. Blood. 2012;120(12):2454–65.
Steensma DP. Clinical consequences of clonal hematopoiesis of indeterminate potential. Hematol Am Soc Hematol Educ Program. 2018;2018(1):264–9.
Takahashi K, Wang F, Kantarjian H, Song X, Patel K, Neelapu S, et al. Copy number alterations detected as clonal hematopoiesis of indeterminate potential. Blood Adv. 2017;1(15):1031–6.
Genovese G, Kahler AK, Handsaker RE, Lindberg J, Rose SA, Bakhoum SF, et al. Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. N Engl J Med. 2014;371(26):2477–87.
Jaiswal S, Fontanillas P, Flannick J, Manning A, Grauman PV, Mar BG, et al. Age-related clonal hematopoiesis associated with adverse outcomes. N Engl J Med. 2014;371(26):2488–98.
Sun J, Ramos A, Chapman B, Johnnidis JB, Le L, Ho YJ, et al. Clonal dynamics of native haematopoiesis. Nature. 2014;514(7522):322–7.
Boultwood J, Perry J, Zaman R, Fernandez-Santamaria C, Littlewood T, Kusec R, et al. High-density single nucleotide polymorphism array analysis and ASXL1 gene mutation screening in chronic myeloid leukemia during disease progression. Leukemia. 2010;24(6):1139–45.
Kim T, Tyndel MS, Kim HJ, Ahn JS, Choi SH, Park HJ, et al. Spectrum of somatic mutation dynamics in chronic myeloid leukemia following tyrosine kinase inhibitor therapy. Blood. 2017;129(1):38–47.
Roche-Lestienne C, Marceau A, Labis E, Nibourel O, Coiteux V, Guilhot J, et al. Mutation analysis of TET2, IDH1, IDH2 and ASXL1 in chronic myeloid leukemia. Leukemia. 2011;25(10):1661–4.
Makishima H, Jankowska AM, McDevitt MA, O’Keefe C, Dujardin S, Cazzolli H, et al. CBL, CBLB, TET2, ASXL1, and IDH1/2 mutations and additional chromosomal aberrations constitute molecular events in chronic myelogenous leukemia. Blood. 2011;117(21):e198–206.
Kim T, Tyndel MS, Zhang Z, Ahn J, Choi S, Szardenings M, et al. Exome sequencing reveals DNMT3A and ASXL1 variants associate with progression of chronic myeloid leukemia after tyrosine kinase inhibitor therapy. Leuk Res. 2017;59:142–8.
Saussele S, Richter J, Guilhot J, Gruber FX, Hjorth-Hansen H, Almeida A, et al. Discontinuation of tyrosine kinase inhibitor therapy in chronic myeloid leukaemia (EURO-SKI): a prespecified interim analysis of a prospective, multicentre, non-randomised, trial. Lancet Oncol. 2018;19(6):747–57.
Acknowledgments
We would like to thank Dr. Sai Peng Sim for allowing the usage of the Medical Molecular Genetic Laboratory, UNIMAS and supporting the study, Dr. Yuwana Podin for allowing the storage of RNA samples at the Institute of Health and Community Medicine, UNIMAS, staff from IRCMS, Kumamoto University and FMHS, UNIMAS for assisting this study. We also would like to thank Dr. Jameela Sathar and Phan Chin Lee from Ampang Hospital, Malaysia for assessing the RT-PCR data by NRL. This study was supported by the Japan Society for the Promotion of Science (JSPS) RONPAKU (Dissertation PhD) Program FY2017, UNIMAS Special Grant Scheme no: F05/SpGS/1401/16/2, Fundamental Research Grant Scheme no: FRGS/SKK01(01)/1290/2015(7) and FMHS, UNIMAS under Research no: FPSK/FPI/(F09)/2017/(14).
Author information
Authors and Affiliations
Contributions
JWK conceived, designed, planned, and conducted the entire research, collected, analyzed, and interpreted the data, and wrote the manuscript. ATS designed, planned, and conducted samplings and provided statistical consultation. SPT conducted two of the samplings and performed FBC and smears of PB films. IFL supervised/supported blood lysis and RNA extraction. SK provided technical consultation on qPCR and performed validation of the positive subject. LS supervised and supported the research. MO co-supervised the research and provided technical support. GS supervised the research, provided technical support and consultation, and wrote the manuscript. All authors participated in revising the manuscript and its final approval.
Corresponding authors
Ethics declarations
Conflict of interest
All authors declared no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Kuan, J.W., Su, A.T., Tay, S.P. et al. Low prevalence of the BCR–ABL1 fusion gene in a normal population in southern Sarawak. Int J Hematol 111, 217–224 (2020). https://doi.org/10.1007/s12185-019-02768-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-019-02768-x