Abstract
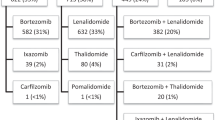
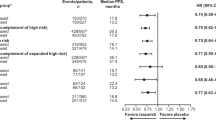
Lenalidomide is an effective therapeutic agent for multiple myeloma (MM). However, its efficacy in the context of chromosomal abnormalities (CA) is poorly understood. We retrospectively analyzed 83 patients with relapsed/refractory (RR) MM, who received lenalidomide plus low-dose dexamethasone (Ld), in the context of CA. The median age and number of prior therapies were 69 and 2, respectively. Three, 11, 45, and 19 patients achieved complete response, very good partial response, partial response, and stable disease, respectively. Median progression-free survival (PFS) and overall survival (OS) were 11.1 and 38.8 months, respectively. Seventy-two patients were evaluated for frequently observed translocations; median PFS was 24.4 months in 20 patients with t(11;14), 13.0 months in 16 patients with t(4;14), and 3.7 months in seven patients with t(14;16). G-banded karyotype analysis detected 11 hypodiploid patients, who had shorter PFS and OS (2.5 and 6.2 months, respectively) compared to others (13.0 and 43.7 months, respectively). Hypodiploid patients showed poor clinical outcome, whereas patients with t(11;14) showed favorable outcome. In summary, the present study presents the clinical impact of chromosomal abnormalities on the outcome of Ld therapy, and contributes to understanding the appropriate choice of lenalidomide-based therapy to achieve effective treatment of RR MM.



Similar content being viewed by others
References
Kumar SK, Rajkumar SV, Dispenzieri A, Lacy MQ, Hayman SR, Buadi FK, et al. Improved survival in multiple myeloma and the impact of novel therapies. Blood. 2008;111:2516–20.
Weber DM, Chen C, Niesvizky R, Wang M, Belch A, Stadtmauer EA, et al. Lenalidomide plus dexamethasone for relapsed multiple myeloma in North America. N Engl J Med. 2007;357:2133–42.
Dimopoulos M, Spencer A, Attal M, Prince HM, Harousseau JL, Dmoszynska A, et al. Lenalidomide plus dexamethasone for relapsed or refractory multiple myeloma. N Engl J Med. 2007;357:2123–32.
Dimopoulos MA, Chen C, Spencer A, Niesvizky R, Attal M, Stadtmauer EA, et al. Long-term follow-up on overall survival from the MM-009 and MM-010 phase III trials of lenalidomide plus dexamethasone in patients with relapsed or refractory multiple myeloma. Leukemia. 2009;23:2147–52.
Avet-Loiseau H, Hulin C, Campion L, Rodon P, Marit G, Attal M, et al. Chromosomal abnormalities are major prognostic factors in elderly patients with multiple myeloma: the intergroupe francophone du myelome experience. J Clin Oncol. 2013;31:2806–9.
Chesi M, Bergsagel PL. Molecular pathogenesis of multiple myeloma: basic and clinical updates. Int J Hematol. 2013;97:313–23.
Munshi NC, Anderson KC, Bergsagel PL, Shaughnessy J, Palumbo A, Durie B, et al. Consensus recommendations for risk stratification in multiple myeloma: report of the International Myeloma Workshop Consensus Panel 2. Blood. 2011;117:4696–700.
Debes-Marun CS, Dewald GW, Bryant S, Picken E, Santana-Davila R, Gonzalez-Paz N, et al. Chromosome abnormalities clustering and its implications for pathogenesis and prognosis in myeloma. Leukemia. 2003;17:427–36.
Fassas AB, Spencer T, Sawyer J, Zangari M, Lee CK, Anaissie E, et al. Both hypodiploidy and deletion of chromosome 13 independently confer poor prognosis in multiple myeloma. Br J Haematol. 2002;118:1041–7.
Smadja NV, Bastard C, Brigaudeau C, Leroux D, Fruchart C. Groupe Francais de Cytogenetique H Hypodiploidy is a major prognostic factor in multiple myeloma. Blood. 2001;98:2229–38.
Fonseca R, Blood E, Rue M, Harrington D, Oken MM, Kyle RA, et al. Clinical and biologic implications of recurrent genomic aberrations in myeloma. Blood. 2003;101:4569–75.
Klein U, Jauch A, Hielscher T, Hillengass J, Raab MS, Seckinger A, et al. Chromosomal aberrations +1q21 and del(17p13) predict survival in patients with recurrent multiple myeloma treated with lenalidomide and dexamethasone. Cancer. 2011;117:2136–44.
Sawyer JR. The prognostic significance of cytogenetics and molecular profiling in multiple myeloma. Cancer Genet. 2011;204:3–12.
Avet-Loiseau H, Soulier J, Fermand JP, Yakoub-Agha I, Attal M, Hulin C, et al. Impact of high-risk cytogenetics and prior therapy on outcomes in patients with advanced relapsed or refractory multiple myeloma treated with lenalidomide plus dexamethasone. Leukemia. 2010;24:623–8.
Chang H, Jiang A, Qi C, Trieu Y, Chen C, Reece D. Impact of genomic aberrations including chromosome 1 abnormalities on the outcome of patients with relapsed or refractory multiple myeloma treated with lenalidomide and dexamethasone. Leuk Lymphoma. 2010;51:2084–91.
Dimopoulos MA, Kastritis E, Christoulas D, Migkou M, Gavriatopoulou M, Gkotzamanidou M, et al. Treatment of patients with relapsed/refractory multiple myeloma with lenalidomide and dexamethasone with or without bortezomib: prospective evaluation of the impact of cytogenetic abnormalities and of previous therapies. Leukemia. 2010;24:1769–78.
Kim K, Kim SJ, Voelter V, Suh C, Yoon SS, Lee JJ, et al. Lenalidomide with dexamethasone treatment for relapsed/refractory myeloma patients in Korea-experience from 110 patients. Ann Hematol. 2014;93:113–21.
Kuroda J, Kobayashi T, Taniwaki M. Prognostic indicators of lenalidomide for multiple myeloma: consensus and controversy. Expert Rev Anticancer Ther. 2015;15:787–804.
Reece D, Song KW, Fu T, Roland B, Chang H, Horsman DE, et al. Influence of cytogenetics in patients with relapsed or refractory multiple myeloma treated with lenalidomide plus dexamethasone: adverse effect of deletion 17p13. Blood. 2009;114:522–5.
Narita T, Inagaki A, Kobayashi T, Kuroda Y, Fukushima T, Nezu M, et al. t(14;16)-positive multiple myeloma shows negativity for CD56 expression and unfavorable outcome even in the era of novel drugs. Blood Cancer J. 2015;5:e285.
Suzuki K, Shinagawa A, Uchida T, Taniwaki M, Hirata H, Ishizawa K, et al. Lenalidomide and low-dose dexamethasone in Japanese patients with newly diagnosed multiple myeloma: a phase II study. Cancer Sci. 2016;107:653–8.
Greipp PR, San Miguel J, Durie BG, Crowley JJ, Barlogie B, Blade J, et al. International staging system for multiple myeloma. J Clin Oncol. 2005;23:3412–20.
Inagaki A, Tajima E, Uranishi M, Totani H, Asao Y, Ogura H, et al. Global real-time quantitative reverse transcription-polymerase chain reaction detecting proto-oncogenes associated with 14q32 chromosomal translocation as a valuable marker for predicting survival in multiple myeloma. Leuk Res. 2013;37:1648–55.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
Avet-Loiseau H, Durie BG, Cavo M, Attal M, Gutierrez N, Haessler J, et al. Combining fluorescent in situ hybridization data with ISS staging improves risk assessment in myeloma: an International Myeloma Working Group collaborative project. Leukemia. 2013;27:711–7.
Usmani SZ, Rodriguez-Otero P, Bhutani M, Mateos MV, Miguel JS. Defining and treating high-risk multiple myeloma. Leukemia. 2015;29:2119–25.
Kobayashi T, Kuroda J, Fuchida S, Kaneko H, Yagi H, Shibayama H, et al. Impact of early use of lenalidomide and low-dose dexamethasone on clinical outcomes in patients with relapsed/refractory multiple myeloma. Int J Hematol. 2015;101:37–45.
Krönke J, Udeshi ND, Narla A, Grauman P, Hurst SN, McConkey M, et al. Lenalidomide causes selective degradation of IKZF1 and IKZF3 in multiple myeloma cells. Science. 2014;343:301–5.
Kriegsmann K, Baertsch MA, Awwad MHS, Merz M, Hose D, Seckinger A, et al. Cereblon-binding proteins expression levels correlate with hyperdiploidy in newly diagnosed multiple myeloma patients. Blood Cancer J. 2019;9:13.
Zambello R, Bonaldi L, Berno T, Martines A, Sechettin E, De March E, et al. Cytogenetic impact on lenalidomide treatment in relapsed/refractory multiple myeloma: a real-life evaluation. Clin Lymphoma Myeloma Leuk. 2015;15:592–8.
Van Wier S, Braggio E, Baker A, Ahmann G, Levy J, Carpten JD, et al. Hypodiploid multiple myeloma is characterized by more aggressive molecular markers than non-hyperdiploid multiple myeloma. Haematologica. 2013;98:1586–92.
Lu J, Lu J, Chen W, Huo Y, Huang X, Hou J, et al. Clinical features and treatment outcome in newly diagnosed Chinese patients with multiple myeloma: results of a multicenter analysis. Blood Cancer J. 2014;4:e239.
Reece D, Song KW, Fu T, Roland B, Chang H, Horsman DE, et al. Influence of cytogenetics in patients with relapsed or refractory multiple myeloma treated with lenalidomide plus dexamethasone: adverse effect of deletion 17p13. Blood. 2009;114:522–5.
Thanendrarajan S, Tian E, Qu P, Mathur P, Schinke C, van Rhee F, et al. The level of deletion 17p and bi-allelic inactivation of TP53 has a significant impact on clinical outcome in multiple myeloma. Haematologica. 2017;102:e364–7.
Acknowledgements
This work was partly supported by a Grant-in-Aids for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology (16K07179 & 16K09855), the National Cancer Center Research and Development Fund (26-A-4), and the Practical Research for Innovative Cancer Control from Japan Agency for Medical Research and development, AMED (15ck0106077h0002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
MR received research funding from Celgene Co., Ltd. SI received research funding and declares Honoraria from Janssen Pharmaceutical K.K., and Celgene Co., Ltd. SI also received research funding from Kyowa Hakko Kirin Co., Ltd., Chugai Pharmaceutical Co., Ltd., Bristol-Myers Squibb, Takeda Yakuhin Co., Ltd., and Ono Pharmaceutical Co., Ltd.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Yoshida, T., Ri, M., Fujinami, H. et al. Impact of chromosomal abnormalities on the efficacy of lenalidomide plus dexamethasone treatment in patients with relapsed/refractory multiple myeloma. Int J Hematol 110, 228–236 (2019). https://doi.org/10.1007/s12185-019-02669-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-019-02669-z