Abstract
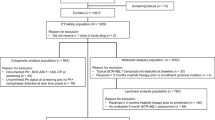
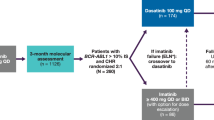
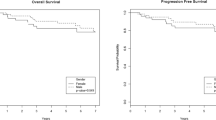
We evaluated the efficacy and safety of switching to nilotinib in CML-CP patients who had achieved MMR with continuous detectable BCR-ABL1 transcript levels after long-term imatinib treatment. Patients who had achieved MMR, but not deep molecular response (DMR), after > 18 months from the initiation of imatinib received nilotinib 400 mg twice daily for up to 24 months. BCR-ABL1 transcript levels were assessed every 3 months. Thirty-eight patients with a median age of 57.5 years (range 22–76 years) were evaluated. Twenty-seven patients completed 24 months of nilotinib treatment; 11 discontinued nilotinib due to retraction of consent (three patients), loss of MMR (1), intolerance (3) or AEs (5). Twenty patients [52.6%, (90% CI 38.2–66.7%)] achieved DMR. The cumulative incidence of achieving DMR by the time of 3, 6, 9, 12, 15, 18, and 21 months was 22.9, 37.7, 47.0, 53.7, 53.7, 53.7, and 53.7%, respectively. Adverse events were consistent with those reported in other nilotinib studies. Patients experienced each of the following cardiovascular complications: atrial fibrillation (G2), chest tightness and dyspnea (G1), myocardial infarction (G2) and heart failure (G3) (n = 1 for each complication). This study indicates nilotinib achieves strong, rapid induction of DMR for patients who achieved MMR after long-term imatinib therapy.

Similar content being viewed by others
References
O’Brien SG, Guilhot F, Larson RA, Gathmann I, Baccarani M, Cervantes F, et al. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2003;348:994–1004.
Druker BJ, Guilhot F, O’Brien SG, Gathmann I, Kantarjian H, Gattermann N, et al. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med. 2006;355:2408–17.
Hochhaus A, O’Brien SG, Guilhot F, Druker BJ, Branford S, Foroni L, et al. Six-year follow-up of patients receiving imatinib for the first-line treatment of chronic myeloid leukemia. Leukemia. 2009;23:1054–61.
Baccarani M, Cortes J, Pane F, Niederwieser D, Saglio G, Apperley J, et al. Chronic myeloid leukemia: an update of concepts and management recommendations of European LeukemiaNet. J Clin Oncol. 2009;27:6041–51.
Baccarani M, Deininger MW, Rosti G, Hochhaus A, Soverini S, Apperley JF, et al. European LeukemiaNet recommendations for the management of chronic myeloid leukemia: 2013. Blood. 2013;122:872–84.
Branford S, Seymour JF, Grigg A, Arthur C, Rudzki Z, Lynch K, et al. BCR-ABL messenger RNA levels continue to decline in patients with chronic phase chronic myeloid leukemia treated with imatinib for more than 5 years and approximately half of all first-line treated patients have stable undetectable BCR-ABL using strict sensitivity criteria. Clin Cancer Res. 2007;13:7080–5.
Branford S, Yeung DT, Ross DM, Prime JA, Field CR, Altamura HK, et al. Early molecular response and female sex strongly predict stable undetectable BCR-ABL1, the criteria for imatinib discontinuation in patients with CML. Blood. 2013;121:3818–24.
Etienne G, Dulucq S, Nicolini FE, Morisset S, Fort MP, Schmitt A, et al. Achieving deeper molecular response is associated with a better clinical outcome in chronic myeloid leukemia patients on imatinib front-line therapy. Haematologica. 2014;99:458–64.
Hehlmann R, Müller MC, Lauseker M, Hanfstein B, Fabarius A, Schreiber A, et al. Deep molecular response is reached by the majority of patients treated with imatinib, predicts survival, and is achieved more quickly by optimized high-dose imatinib: results from the randomized CML-study IV. J Clin Oncol. 2014;32:415–23.
Mahon FX, Réa D, Guilhot J, Guilhot F, Huguet F, Nicolini F, et al. Discontinuation of imatinib in patients with chronic myeloid leukaemia who have maintained complete molecular remission for at least 2 years: the prospective, multicentre Stop Imatinib (STIM) trial. Lancet Oncol. 2010;11:1029–35.
Ross DM, Branford S, Seymour JF, Schwarer AP, Arthur C, Yeung DT, et al. Safety and efficacy of imatinib cessation for CML patients with stable undetectable minimal residual disease: results from the TWISTER study. Blood. 2013;122:515–22.
Imagawa J, Tanaka H, Okada M, Nakamae H, Hino M, Murai K, et al. Discontinuation of dasatinib in patients with chronic myeloid leukaemia who have maintained deep molecular response for longer than 1 year (DADI trial): a multicentre phase 2 trial. Lancet Haematol. 2015;2:e528–35.
Saglio G, Kim DW, Issaragrisil S, le Coutre P, Etienne G, Lobo C, et al. Nilotinib versus imatinib for newly diagnosed chronic myeloid leukemia. N Engl J Med. 2010;362:2251–9.
Hochhaus A, Saglio G, Hughes TP, Larson RA, Kim DW, Issaragrisil S, et al. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia. 2016;30:1044–54.
Nakamae H, Yoshida C, Miyata Y, Hidaka M, Uike N, Koga D, et al. A new diagnostic kit, ODK-1201, for the quantitation of low major BCR-ABL mRNA level in chronic myeloid leukemia: correlation of quantitation with major BCR-ABL mRNA kits. Int J Hematol. 2015;102:304–11.
Hughes TP, Lipton JH, Spector N, Cervantes F, Pasquini R, Clementino NC, et al. Deep molecular responses achieved in patients with CML-CP who are switched to nilotinib after long-term imatinib. Blood. 2014;124:729–36.
Falchi L, Kantarjian HM, Wang X, Verma D, Quintás-Cardama A, O’Brien S, et al. Significance of deeper molecular responses in patients with chronic myeloid leukemia in early chronic phase treated with tyrosine kinase inhibitors. Am J Hematol. 2013;88:1024–9.
Huang X, Cortes J, Kantarjian H. Estimations of the increasing prevalence and plateau prevalence of chronic myeloid leukemia in the era of tyrosine kinase inhibitor therapy. Cancer. 2012;118:3123–7.
Dulucq S, Mahon FX. Deep molecular responses for treatment-free remission in chronic myeloid leukemia. Cancer Med. 2016;5:2398–411.
Hughes TP, Ross DM. Moving treatment-free remission into mainstream clinical practice in CML. Blood. 2016;128:17–23.
Acknowledgements
This study was supported and funded by Novartis Pharmaceutical Corporation. We would like to thank all of the participated patients and their families. We are indebted to the physicians, all other co-medical staff and Independent Data Monitoring Committee (Masahiro Kizaki, Kazuma Ohyashiki and Noriko Usui) who contributed to this study. We also thank the stuffs at the Clinical Research Support Center Kyushu (CReS Kyushu) for their excellent collection and management of data, secretarial assistance, and any other supports.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
I. M. acted as a consultant for Otsuka and received honoraria for Otsuka, Novartis, Bristol-Myers Squibb. J. K. received research funding from Celgene, Bristol-Myers Squibb and Astra Zeneca and honoraria for Celgene, Bristol-Myers Squibb and Janssen. H. N. acted as a consultant and received research funding and honoraria from Novartis. H. S. received research funding and speaker bureau from Novartis. M. H. received research funding and honoraria from Novartis. K. A. acted as a consultant for Sumitomo Dainippon and Kyowa Hakko Kirin and received research funding from Celgene, Kyowa Hakko Kirin, Astellas, Shionogi, Asahi Kasei, Chugai, Bristol-Myers Squibb. Y. K. received research funding from Kyowa Hakko Kirin, Shionogi, Chugai, Pfizer, Eisai, Astellas, Nippon Shinyaku, Alexionpharma, Bristol-Myers Squibb, Toyama Chemical, Fujimotoseiyaku. The remaining authors declare no competing financial interests.
About this article
Cite this article
Ishikawa, J., Matsumura, I., Kawaguchi, T. et al. Efficacy and safety of switching to nilotinib in patients with CML-CP in major molecular response to imatinib: results of a multicenter phase II trial (NILSw trial). Int J Hematol 107, 535–540 (2018). https://doi.org/10.1007/s12185-018-2401-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-018-2401-y