Abstract
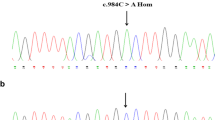
Coagulation factor XII deficiency is a rare autosomal recessive disorder, which could be found in a consanguineous family. We studied a Chinese family in which the activated partial thromboplastin time (APTT) of the proband had clearly prolonged up to 101.7 s, associated with low FXII activity of 3% and FXII antigen < 1%. To analyze the gene mutation in this FXII-deficient patient, we performed FXII mutation screening, and analyzed the DNA sequence of the F12 gene. A ClustalX-2.1-win and four online bioinformatics software services were used to study the conservatism and effects of the mutation. A transient in vitro expression study was performed to elucidate the possible pathological mechanism. Sequence analysis revealed a homozygous c.1681 G > A point mutation in exon 14, causing a novel Gly542Ser mutation in the catalytic domain. The results of the conservatism and bioinformatics analyses both indicated that the mutation likely affects the function of the protein. Additional expression studies in COS-7 cells showed that the antigen level of mutant FXII (FXII-Gly542Ser) was lower than wild type in culture medium, whereas the corresponding level of FXII antigen in cell lysates was equivalent. These results suggest that the Gly542Ser mutation causes FXII deficiency through intracellular degradation.




Similar content being viewed by others
References
Citarella F, Ravon DM, Pascucci B, Felici A, Fantoni A, Hack CE. Structure/function analysis of human factor XII using recombinant deletion mutants. Eur J Biochem. 1996;238:240–9.
Ishii K, Oguchi S, Moriki T, Yatabe Y, Takeshita E, Murata M, et al. Genetic analyses and expression studies identified a novel mutation (W486C) as a molecular basis of congenital coagulation factor XII deficiency. Blood Coagul Fibrin. 2004;15:367–73.
Kaplan AP, Silverberg M. The coagulation-kinin pathway of human plasma. Blood. 1987;70:1–15.
McMullen BA, Fujikawa K. Amino acid sequence of the heavy chain of human a-factor XIIa (activated hageman factor). J Biol Chem. 1985;260:5328–41.
Iijima K, Arakawa Y, Sugahara Y, Matsushita M, Moriguchi Y, Shimohiro H, et al. Factor XII Osaka: abnormal factor XII with partially defective prekallikrein cleavage activity. Thromb Haemost. 2011;105:473–8.
Pauer HU, Burfeind P, Köstering H, Emons G, Hinney B. Factor XII deficiency is strongly associated with primary recurrent abortions. Fertil Steril. 2003;80:590–4.
Mordillo C, Martinez-Marchαn E, Fontcuberta J, Soria JM. Molecular analysis of multiple genetic variants in Spanish FXII-deficient families. Haematologica. 2007;92:1569–72.
Renné T, Schmaier AH, Nickel KF, Blombäck M, Maas C. In vivo roles of factor XII. Blood. 2012;120(22):4296–303.
Singhamatr P, Kanjanapongkul S, Rojnuckarin P. Molecular analysis of factor XII gene in Thai patients with factor XII deficiency. Blood Coagul Fibrinolysis. 2013;24:599–604.
Ishii K, Oguchi S, Moriki T, Yatabe Y, Takeshita E, Murata M, et al. Genetic analyses and expression studies identified a novel mutation (W486C) as a molecular basis of congenital coagulation factor XII deficiency. Blood Coagul Fibrinolysis. 2004;15:367–73.
Suzuki K, Murai K, Suwabe A, Ishida Y. Factor XII Ofunato: Lys346Asn mutation associated with blood coagulation factor XII deficiency causes impaired secretion through a proteasome-mediated degradation. Thromb Res. 2010;125:438–43.
Yang LH, Wang YY, Zhou JP, Cheng XL, Hao XP, Xie HX, et al. Identification of genetic defects underlying FXII deficiency in four unrelated Chinese patients. Acta Haematol. 2016;135:238–40.
Dai LY, Zhang DT, Lin J, Wang YY, Tong Y, Li J, et al. A pedigree analysis of Hereditary coagulation factor XII deficiency caused by a novel homozygous mutation: Leu519Arg. Chin J Lab Med. 2015;38:466–9 (China).
Li M, Xie HX, Wang MS, Ding HX. Molecular characterization of a novel missense mutation (Asp538Asn) in a Chinese patient with factor XII deficiency. Clin Lab. 2015;61:1967–71.
Miyata T, Kawabata S, Iwanaga S, Takahashi I, Alving B, Saito H. Coagulation factor XII (Hageman factor) Washington DC: inactive factor XIIa results from Cys-571-Ser substitution. Proc Natl Acad Sci USA. 1989;86:8319–22.
Kondo S, Tokunaga F, Kawano S, Oono Y, Kumagai S, Koide T. Factor XII Tenri, a novel cross-reacting material negative factor XII deficiency, occurs through a proteasome-mediated degradation. Blood. 1999;93:4300–8.
Matsuki E, Miyakawa Y, Okamoto S. A novel factor XII mutation, FXII R84P, causing factor XII deficiency in a patient with hereditary spastic paraplegia. Blood Coagul Fibrinolysis. 2011;22:227–30.
Yarovaya GA, Blokhina TB, Neshkova EA. Contact system. New concepts on activation mechanisms and bioregulatory functions. Biochemistry. 2002;67:13–24.
Wang XF, Dai J, Wang MS, Ding QL, Wang HL. Analysis of the factor XII gene mutations in two Chinese pedigrees with congenital clotting factor XII deficiency. J Diagn Concepts Pract. 2005;4:447–50 (Shanghai).
Acknowledgements
We thank all the participants in this study.
Funding
This work was supported by The Project Supported by Zhejiang Provincial Natural Science Foundation of China (LY16H080005) and The Project of Wenzhou Public Welfare Science and Technology (Y20150098).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
No potential conflict of interest was reported by the authors.
About this article
Cite this article
Zou, A., Wang, M., Jin, Y. et al. Genetic analysis of a novel missense mutation (Gly542Ser) with factor XII deficiency in a Chinese patient of consanguineous marriage. Int J Hematol 107, 436–441 (2018). https://doi.org/10.1007/s12185-017-2393-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-017-2393-z