Abstract
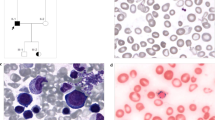
X-linked sideroblastic anemia (XLSA) is a rare hereditary disorder that typically manifests in males as microcytic anemia. Here, we report a family with XLSA that affects females and manifests as macrocytic anemia. The proband was a Japanese woman harboring a heterozygous mutation c.679C>T in the ALAS2 gene. This mutation causes the amino acid substitution R227C, which disrupts the enzymatic activity of erythroid-specific δ-aminolevulinic acid synthase. The mutation was not detected in the ALAS2 complementary DNA from peripheral blood red blood cells of the proband, indicating that the cells were mostly derived from erythroblasts expressing wild-type ALAS2. The proband’s mother, who had been diagnosed with myelodysplastic syndrome, also had XLSA with the same mutation. Clinicians should be aware that XLSA can occur not only in males but also in females, in whom it manifests as macrocytic anemia.




Similar content being viewed by others
References
Yoshida K, Sanada M, Shiraishi Y, Nowak D, Nagata Y, Yamamoto R, et al. Frequent pathway mutations of splicing machinery in myelodysplasia. Nature. 2011;478:64–9.
Ohba R, Furuyama K, Yoshida K, Fujiwara T, Fukuhara N, Onishi Y, et al. Clinical and genetic characteristics of congenital sideroblastic anemia: comparison with myelodysplastic syndrome with ring sideroblast (MDS-RS). Ann Hematol. 2013;92:1–9.
Fujiwara T, Harigae H. Pathophysiology and genetic mutations in congenital sideroblastic anemia. Pediatr Int. 2013;55:675–9.
Bottomley SS, Fleming MD. Sideroblastic anemia: diagnosis and management. Hematol Oncol Clin North Am. 2014;28:653–70.
Cooley TB. A severe type of hereditary anemia with elliptocytosis: interesting sequence of splenectomy. Am J Med Sci. 1945;209:561–8.
Aoki Y, Urata G, Takaku F. Aminolevulinic acid synthetase activity in erythroblasts of patients with primary sideroblastic anemia. Nihon Ketsueki Gakkai Zasshi. 1973;36:74–7.
Yamamoto M, Nakajima O. Animal models for X-linked sideroblastic anemia. Int J Hematol. 2000;72:157–64.
Harigae H, Furuyama K. Hereditary sideroblastic anemia: pathophysiology and gene mutations. Int J Hematol. 2010;92:425–31.
Aivado M, Gattermann N, Rong A, Giagounidis AA, Prall WC, Czibere A, et al. X-linked sideroblastic anemia associated with a novel ALAS2 mutation and unfortunate skewed X-chromosome inactivation patterns. Blood Cells Mol Dis. 2006;37:40–5.
Gong J, Ferreira GC. Aminolevulinate synthase: functionally important residues at a glycine loop, a putative pyridoxal phosphate cofactor-binding site. Biochemistry. 1995;34:1678–85.
Astner I, Schulze JO, van den Heuvel J, Jahn D, Schubert WD, Heinz DW. Crystal structure of 5-aminolevulinate synthase, the first enzyme of heme biosynthesis, and its link to XLSA in humans. EMBO J. 2005;24:3166–77.
Sankaran VG, Ulirsch JC, Tchaikovskii V, Ludwig LS, Wakabayashi A, Kadirvel S, et al. X-linked macrocytic dyserythropoietic anemia in females with an ALAS2 mutation. J Clin Invest. 2015;125:1665–9.
Acknowledgments
DNA sequencing analysis was performed at the Medical Research Support Center, Graduate School of Medicine, Kyoto University. This work was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (26461400, received by HK) and the National Research Group on Idiopathic Bone Marrow Failure Syndromes, granted by the Ministry of Health, Labor and Welfare, Japan (H26-Nanchi-Ippan-062).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Katsurada, T., Kawabata, H., Kawabata, D. et al. A Japanese family with X-linked sideroblastic anemia affecting females and manifesting as macrocytic anemia. Int J Hematol 103, 713–717 (2016). https://doi.org/10.1007/s12185-016-1949-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-016-1949-7