Abstract
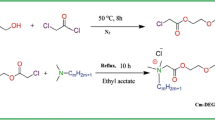
The critical micelle concentration (CMC) of 1,2-bis[N-methyl-N-(3-sulfopropyl)-alkylammonium]-ethane betaine (GCS12) was measured using a tensiometric method in the presence of inorganic salts. Inorganic salt has a little impact on the surface tension and CMC of zwitterionic gemini surfactant. The CMC value of GCS12 is 0.07 mmol/L in distilled water, while all CMC values are around 0.04–0.05 mmol/L in the presence of 0.5 % NaCl, 2 % NaCl, and 2 % NaCl + 0.05 % CaCl2. The interactions between GCS12 and non-ionic surfactant lauric acid diethanolamide (CDA) were investigated by measuring the CMC of their mixtures at different molar ratios. CDA and GCS12 form mixed micelles and exhibit synergism when the mole fraction of CDA is higher than 0.25. Both the steric effect of the head group and GCS12 charge affect the formation and stability of the mixed micelles. Small amounts of GCS12 with a lower CMC penetrate into the micelle of nonionic surfactant with a higher CMC and reduce its degree of hydration inducing an attractive interaction between the two surfactants.






Similar content being viewed by others
References
Rosen MJ, Wang H, Shen P, Zhu Y (2005) Ultralow interfacial tension for enhanced oil recovery at very low surfactant concentration. Langmuir 21:3479
Al-Ashraf T, Ahmed A, Suttar EA (2002) Producing ultralow interfacial tension at the oil-water interface. Pet Sci Technol 20:773
Gogoi SB (2011) Adsorption–desorption of surfactant for enhanced oil recovery. Transp Porous Med 90:589–604
Shaddel S, Tabatabae-Nejad SA (2015) Alkali/surfactant improved low-salinity water flooding. Transp Porous Med 106:621–642
Alam MS, Siddiq AM, Kamely N (2015) Micellization behavior of a cationic gemini surfactant, pentanediyl-1,5-bis(dimethylcetylammonium bromide): effect of asparagine and temperature. J Disp Sci Technol 36:1134–1139
Rosen MJ, Tracy DJ (1998) Gemini surfactants. J Surfactants Deterg 1:547–554
Zana R, Xia J (eds) (2003) Gemini surfactants: synthesis, interfacial and solution-phase behavior, and applications. Marcel Dekker, New York
Liu GY, Gu DM, Liu HY (2012) Thermodynamic properties of micellization of sulfobetaine-type zwitterionic gemini surfactants in aqueous solutions––a free energy perturbation study. J Colloid Interface Sci 375:148–153
Yang QS, Wang DM, Zheng YC (2014) Development of alkaline water and surfactant composite flooding systems in Linnan Oilfield. Oilfield Chem 31:568–571
Alargova RG, Kochijashky II, Sierra ML (2001) Mixed micellization of dimeric (gemini) surfactants and conventional surfactants. II. CMC and micelle aggregation numbers for various mixtures. J Colloid Interface Sci 235:119–129
Zana R, Levy H, Kwetkat K (1998) Mixed micellization of dimeric (gemini) surfactants and conventional surfactants. I. Mixtures of an anionic dimeric surfactant and of the nonionic surfactants C12E5 and C12E8. J Colloid Interface Sci 197:370–376
Du X, Lu Y, Li L, Kou J, Yang Z (2007) Mixed micellization of alkylbenzene sulfonate gemini surfactant Ia and nonionic surfactant C10E6 in aqueous solution. Acta Phys Chim Sin 23(2):173–176
Khan IA, Mohammad R, Alam MS (2010) Surface properties and mixed micellization of cationic gemini surfactants with ethyleneamines. J Chem Eng Data 55:370–380
Lakra J, Tikariha D, Yadav T (2014) Mixed micellization of gemini and cationic surfactants: physicochemical properties and solubilization of polycyclic aromatic hydrocarbons. Colloids Surf A 451:56–65
Singh K, Marangoni DG (2007) Synergistic interactions in the mixed micelles of cationic gemini with zwitterionic surfactants: the pH and spacer effect. J Colloid Interface Sci 315:620–626
Ghosh S, Khatua D, Dey J (2011) Interaction between zwitterionic and anionic surfactants: spontaneous formation of zwitterionic vesicles. Langmuir 27:5184–5192
Xi Z, Feng Y (2010) Synthesis and properties of alkylbetaine zwitterionic gemini surfactants. J Surfact Deterg 13:51–57
Yoshimura T, Ichinokawa T, Kaji M et al (2006) Synthesis and surface-active properties of sulfobetaine-type zwitterionic gemini surfactants. Colloid Surf A 273:208–212
Rais F, Baati R, Dama NK (2008) The use of a eutectic mixture of olive pomace oil fatty amides to easily prepare sulfated amides applied as lime soap dispersants. J Am Oil Chem Soc 85:869–877
Rubingh DN (1979) Solution chemistry of surfactants. Plenum Press, New York
Clint JH (1975) Micellization of mixed nonionic surface active agents. J Chem Soc Faraday Trans 1 71:1327–1334
Holland PM, Rubingh DN (1983) Nonideal multicomponent mixed micelle model. J Phys Chem 87:1984–1990
Huang L, Somasundaran P (1997) Theoretical model and phase behavior for binary surfactant mixtures. Langmuir 13:6683–6688
Rodenas E, Valiente M, Villafruela MS (1999) Different theoretical approaches for the study of the mixed tetraethylene glycol mono-n-dodecyl etherhexadecyltrimethylammonium bromide micelles. J Phys Chem 87:4549–4554
Ren ZH, Luo Y, Zheng YC, Shi DP, Mei P, Li FS (2014) Interacting behavior between amino sulfonate surfactant and octylphenol polyoxyethylene ether (10) in aqueous solution. J Solut Chem 43:853–869
Motomura K, Yamanaka M, Aratono M (1984) Thermodynamic consideration of the mixed micelle of surfactants. Colloid Polym Sci 26(12):945–948
Motomura K, Aratono M (1993) Mixed surfactant systems. Marcel Decker, New York
Rosen MJ (1994) Predicting synergism in binary mixtures of surfactants. Progr Colloid Polym Sci 95:39–47
Acknowledgments
We gratefully acknowledge financial support by the National Natural Science Foundation of China (51474035).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Zheng, Y., Ren, Z., Mei, P. et al. Interactions Between a Sulfobetaine-Type Zwitterionic Gemini Surfactant and Fatty Acid Alkanolamide in Aqueous Micellar Solution. J Surfact Deterg 19, 283–288 (2016). https://doi.org/10.1007/s11743-016-1786-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11743-016-1786-y