Abstract
Purpose
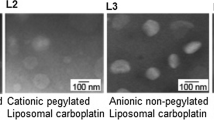
Free doxorubicin (DXR) is not currently used to treat brain tumors because (i) the blood–brain barrier limits the drug deposition into the brain (ii) lethal toxic effects occur when combined with radiation therapy. Since encapsulation of DXR within liposomal carriers could overcome these drawbacks, the present study aimed at evaluating the radiosensitizing properties of non-pegylated (NPL-DXR) and pegylated (PL-DXR) liposomal doxorubicin on orthotopic high-grade glioma xenografts (U87).
Methods
DXR accumulation in brain tissues was assessed by a high-performance liquid chromatography method and antitumor efficacy was evaluated by mice survival determination.
Results
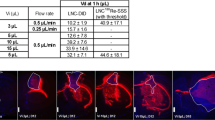
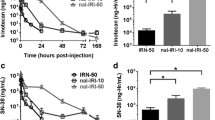
We showed that encapsulation of DXR ensured a preferential deposition of DXR in tumoral tissue in comparison with normal brain tissue: the best AUC tumor tissue/AUC normal tissue ratio depended greatly on the schedule. Overall, thanks to the optimization of the delivery schedule, we demonstrated a radiosensitizing effect for both liposomal DXR without toxicity of this combination on the U87 human malignant glioma orthotopic xenografts.
Conclusion
This study shows that the use of nanocarriers, allowing targeting of intracerebral tumor, renders relevant the combination of anthracyclin with radiation therapy to treat brain tumors, opening a new field of therapeutic applications. However, our results point out that, for each new delivery system, the administration schedules need to be rigorously optimized.



Similar content being viewed by others
Abbreviations
- AUC:
-
Area under the curve
- BBB:
-
Blood–brain barrier
- BTB:
-
Blood–tumor barrier
- CNS:
-
Central nervous system
- DXR:
-
Doxorubicin
- MTD:
-
maximal tolerated dose
- NPL-DXR:
-
Non-pegylated liposomal doxorubicin
- NT:
-
Non-tumor brain hemisphere
- PL-DXR:
-
Pegylated liposomal doxorubicin
- RT:
-
Radiotherapy
- TT:
-
Tumor brain hemisphere
References
Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009;10(5):459–66.
Kreuter J. Drug delivery to the central nervous system by polymeric nanoparticles: what do we know? Rev: Adv. Drug. Deliv; 2013. doi:10.1016/j.addr.2013.08.008.
Wolff JE, Trilling T, Molenkamp G, Egeler RM, Jurgens H. Chemosensitivity of glioma cells in vitro: a meta-analysis. J Cancer Res Clin Oncol. 1999;125(8–9):481–6.
Veringa SJ, Biesmans D, van Vuurden DG, Jansen MH, Wedekind LE, Horsman I, et al. In vitro drug response and efflux transporters associated with drug resistance in pediatric high grade glioma and diffuse intrinsic pontine glioma. PLoS One. 2013;8(4):e61512.
Lesniak M, Upadhyay U, Goodwin R, Tyler B, Brem H. Local delivery of doxorubicin for the treatment of malignant brain tumors in rats. Anticancer Res. 2005;25(6B):3825–31.
Voulgaris S, Partheni M, Karamouzis M, Dimopoulos P, Papadakis N, Kalofonos HP. Intratumoral doxorubicin in patients with malignant brain gliomas. Am J Clin Oncol. 2002;25(1):60–4.
Siegal T, Horowitz A, Gabizon A. Doxorubicin encapsulated in sterically stabilized liposomes for the treatment of a brain tumor model: biodistribution and therapeutic efficacy. J Neurosurg. 1995;83(6):1029–37.
Anders CK, Adamo B, Karginova O, Deal AM, Rawal S, Darr D, et al. Pharmacokinetics and efficacy of PEGylated liposomal doxorubicin in an intracranial model of breast cancer. PLoS One. 2013;8(5).
Fabel K, Dietrich J, Hau P, Wismeth C, Winner B, Przywara S, et al. Long-term stabilization in patients with malignant glioma after treatment with liposomal doxorubicin. Cancer. 2001;92(7):1936–42.
Hau P, Fabel K, Baumgart U, Rümmele P, Grauer O, Bock A, et al. Pegylated liposomal doxorubicin efficacy in patients with recurrent high-grade glioma. Cancer. 2004;100(6):1199–207.
Harrington KJ, Rowlinson-Busza G, Syrigos KN, Vile RG, Uster PS, Peters AM, et al. Pegylated liposome-encapsulated doxorubicin and cisplatin enhance the effect of radiotherapy in a tumor xenograft model. Clin Cancer Res. 2000;6(12):4939–49.
Davies CL, Lundstrom LM, Frengen J, Eikenes L, Bruland SOS, Kaalhus O, et al. Radiation improves the distribution and uptake of liposomal doxorubicin (Caelyx) in human osteosarcoma xenografts. Cancer Res. 2004;64(2):547–53.
Hagtvet E, Røe K, Olsen DR. Liposomal doxorubicin improves radiotherapy response in hypoxic prostate cancer xenografts. Radiat Oncol. 2011;6:135–45.
Labussière M, Aarnink A, Pinel S, Taillandier L, Escanyé JM, Barberi-Heyob M, et al. Interest of liposomal doxorubicin as a radiosensitizer in malignant glioma xenografts. Anticancer Drugs. 2008;19(10):991–8.
Pinel S, Barberi-Heyob M, Cohen-Jonathan E, Merlin JL, Delmas C, Plenat F, et al. Erythropoietin-induced reduction of hypoxia before and during fractionated irradiation contributes to improvement of radioresponse in human glioma xenografts. Int J Radiat Oncol Biol Phys. 2004;59(1):250–9.
Meco D, Colombo T, Ubezio P, Zucchetti M, Zaffaroni M, Riccardi A, et al. Effective combination of ET-743 and doxorubicin in sarcoma: preclinical studies. Cancer Chemother Pharmacol. 2003;52(2):131–8.
Pinel S, Mriouah J, Vandamme M, Chateau A, Plénat F, Guérin E, et al. Synergistic antitumor effect between gefitinib and fractionated irradiation in anaplastic oligodendrogliomas cannot be predicted by the EGFR signaling activity. PLoS One. 2013;8:e68333.
Hong RL, Huang CJ, Tseng YL, Pang VF, Chen ST, Liu JJ, et al. Direct comparison of liposomal doxorubicin with or without polyethylene glycol coating in C-26 tumor-bearing mice: is surface coating with polyethylene glycol beneficial? Clin Cancer Res. 1999;5(11):3645–52.
Pramanik D, Campbell NR, Das S, Gupta S, Chenna V, Bisht S, et al. A composite polymer nanoparticle overcomes multidrug resistance and ameliorates doxorubicin-associated cardiomyopathy. Oncotarget. 2012;3(6):640–50.
Golla K, Cherukuvada B, Ahmed F, Kondapi AK. Efficacy, safety and anticancer activity of protein nanoparticle-based delivery of doxorubicin through intravenous administration in rats. PLoS One. 2012;7(12):e51960.
Tacar O, Sriamornsak P, Dass CR. Doxorubicin: an update on anticancer molecular action, toxicity and novel drug delivery systems. J Pharm Pharmacol. 2013;65(2):157–70.
Sharma US, Sharma A, Chau RI, Straubinger RM. Liposome-mediated therapy of intracranial brain tumors in a rat model. Pharm Res. 1997;14(8):992–8.
Aryal M, Vykhodtseva N, Zhang YZ, Park J, McDannold N. Multiple treatments with liposomal doxorubicin and ultrasound-induced disruption of blood-tumor and blood–brain barriers improve outcomes in a rat glioma model. J Control Release. 2013;169(1–2):103–11.
M. Aryal, C. D. Arvanitis, P. M. Alexander, N. McDannold. Ultrasound-mediated blood–brain barrier disruption for targeted drug delivery in the central nervous system. Adv. Drug Deliv. Rev. (2014).
Koukourakis MI, Koukouraki S, Fezoulidis I, Kelekis N, Kyrias G, Archimandritis S, et al. High intratumoural accumulation of stealth liposomal doxorubicin (Caelyx) in glioblastomas and in metastatic brain tumours. Brit J Cancer. 2000;83(10):1281–6.
Chastagner P, Labussiere M, Aarnink A, Pinel S, Fouyssac F, Bernier V. Comparison of doxorubicin and a liposomal form, doxorubicin chlorhydrate, as radiosensitizer in high grade glioma and rhabdomyosarcoma xenografts. Int J Radiat Oncol Biol Phys. 2006;66:S579.
Parr MJ, Masin D, Cullis PR, Bally MB. Accumulation of liposomal lipid and encapsulated doxorubicin in murine Lewis lung carcinoma: the lack of beneficial effects by coating liposomes with poly(ethylene glycol). J Pharmacol Exp Ther. 1997;280(3):1319–27.
Hendriks BS, Reynolds JG, Klinz SG, Geretti E, Lee H, Leonard SC, et al. Multiscale kinetic modeling of liposomal doxorubicin delivery quantifies the role of tumor and drug-specific parameters in local delivery to tumors. Pharmacometrics Syst Pharmacol. 2012;1:e15.
Charrois GJ, Allen TM. Multiple injections of pegylated liposomal doxorubicin: pharmacokinetics and therapeutic activity. J Pharmacol Exp Ther. 2003;306(3):1058–67.
Charrois GJ, Allen TM. Rate of biodistribution of STEALTH liposomes to tumor and skin: influence of liposome diameter and implications for toxicity and therapeutic activity. Biochim Biophys Acta. 2003;1609(1):102–8.
Allen TM, Cullis PR. Liposomal drug delivery systems: from concept to clinical applications. Adv Drug Deliv Rev. 2013;65(1):36–48.
Duggan ST, Keating GM. Pegylated liposomal doxorubicin: a review of its use in metastatic breast cancer, ovarian cancer, multiple myeloma and AIDS-related Kaposi’s sarcoma. Drugs. 2011;71(18):2531–58.
Arnold RD, Mager DE, Slack JE, Straubinger RM. Effect of repetitive administration of Doxorubicin-containing liposomes on plasma pharmacokinetics and drug biodistribution in a rat brain tumor model. Clin Cancer Res. 2005;11(24 Pt 1):8856–65.
Tsuji A. P-glycoprotein-mediated efflux transport of anticancer drugs at the blood–brain barrier. Ther Drug Monit. 1998;20(5):588–90.
Wohlfart S, Gelperina S, Kreuter J. Transport of drugs across the blood–brain barrier by nanoparticles. J Control Release. 2012;161(2):264–73.
Acknowledgments and Disclosures
We are very grateful to the French “Ligue Contre le Cancer, Comités Lorrains” for financial supports. We thank S. Leclerc and J.M. Escanyé (Service Commun de RMN, Institut Jean Barriol, Université de Lorraine) for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chastagner, P., Sudour, H., Mriouah, J. et al. Preclinical Studies of Pegylated- and Non-Pegylated Liposomal Forms of Doxorubicin as Radiosensitizer on Orthotopic High-Grade Glioma Xenografts. Pharm Res 32, 158–166 (2015). https://doi.org/10.1007/s11095-014-1452-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-014-1452-x