Abstract
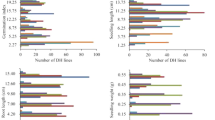
Seed longevity is a crucial issue for germplasm conservation and seed marketing. This trait is determined not only by environmental conditions, but also by genetic factors. Molecular mapping of responsible loci has been performed with several crops, but not with tobacco (Nicotiana tabacum L.). In the present study, we investigate 122 recombinant inbred lines derived from a cross between the cultivars Florida 301 and Hicks. Four germination-related traits were studied by examining seeds either untreated or after controlled deterioration (CD): total germination (TG, %), normal germination (%), time to reach 50 % of TG (h), and the area under the curve after 200 h of germination. Whereas Hicks exhibited high germination percentage and speed in untreated (fresh) seeds, Florida 301 seems to withstand the CD treatment better, having increased seed longevity. In total, four genomic regions located on four different linkage groups (LGs) were identified to be associated with the selected traits. Positive alleles for the individual traits were contributed by both parents. A major quantitative trait locus (QTL) for high percentage TG located on LG 8/18 appeared in both control and deteriorated seeds and was contributed by Hicks. In contrast, Florida 301 donated a favorable allele for germination speed on LG seven after CD only. The position of this locus compared well with a QTL detected in the same population, in a former study examining resistance against the black shank disease caused by Phytophthora nicotianae). The effects of environmental growing conditions of the mother plants on seed longevity are discussed.



Similar content being viewed by others
References
Agacka M, Depta A, Börner M, Doroszewska T, Hay FR, Börner A (2013) Viability of Nicotiana spp. seeds stored under ambient temperature. Seed Sci Technol 41:474–478
Agacka M, Laskowska D, Doroszewska T, Hay FR, Börner A (2014) Longevity of Nicotiana seeds conserved at low temperatures in ex situ genebanks. Seed Sci Technol 42:355–362
Bentsink L, Alonso-Blanco C, Vreugdenhil D, Tesnier K, Groot SPC, Koornneef M (2000) Genetic analysis of seed-soluble oligosaccharides in relation to seed storability of Arabidopsis. Plant Physiol 124:1595–1604
Bindler G, Plieske J, Bakaher N, Gunduz I, Ivanov N, van der Hoeven R, Ganal M, Donini P (2011) A high density genetic map of tobacco (Nicotiana tabacum L.) obtained from large scale microsatellite marker development. Theor Appl Genet 123:219–230
Burgess JL (1938) Report on project to determine the percentage and duration of viability of different varieties of soybeans grown in North Carolina. Proc Assoc Off Seed Anal 23:69
Clerkx EJM, El-Lithy ME, Vierling E, Ruys GJ, Blankestijin-De Vries H, Groot SPC, Vreugdenhil D, Koornneef M (2004) Analysis of natural allelic variation of Arabidopsis seed germination and seed longevity traits between the accessions Landsberg erecta and Shakdara, using a new recombinant inbred line population. Plant Physiol 135:432–443
Haberlandt F (1873) Die Keimfähigkeit unserer Getreidekörner, ihre Dauer und die Mittel ihrer Erhaltung (The germinating capacity of our cereals, their longevity and the means of preserving it). Wien Landw Ztg 22:126
Hampton JG, TeKrony DM (1995) Handbook of vigour test methods. International Seed Testing Association, Zürich, p 117
Hay FR, Adams J, Manger K, Probert R (2008) The use of non-saturated lithium chloride solutions for experimental control of seed water content. Seed Sci Technol 36:737–746
ISTA (2014) International rules for seed testing. International Seed Testing Association, Bassersdorf
Johnson J, Murwin HF, Ogden WB (1930) The germination of tobacco seed. Wis Agric Exp Station Res Bull 104:15
Jones JW (1926) Germination of rice seed as affected by temperature, fungicides, and age. Agron J 18:576–592
Joosen RVL, Kodde J, Willems LAJ, Ligterink W, van der Plas LHW, Hilhorst HWM (2010) Germinator: a software package for high-throughput scoring and curve fitting of Arabidopsis seed germination. Plant J 62:148–159
Kingcaid RR (1943) Effect of storage conditions on the viability of tobacco seed. J Agric Res 67:407–410
Landjeva S, Lohwasser U, Börner A (2010) Genetic mapping within the wheat D genome reveals QTLs for germination, seed vigour and longevity, and early seedling growth. Euphytica 171:129–143
Miura K, Lyn SY, Yano M, Nagamine T (2002) Mapping quantitative trait loci controlling seed longevity in rice (Oryza sativa L.). Theor Appl Genet 104:981–986
Nagel M, Börner A (2010) The longevity of crop seeds stored under ambient conditions. Seed Sci Res 20:1–12
Nagel M, Vogel H, Landjeva S, Buck-Sorlin G, Lohwasser G, Scholz U, Börner A (2009) Seed conservation in ex situ genebanks—genetic studies on longevity in barley. Euphytica 170:5–14
Nagel M, Rehman-Arif MA, Rosenhauer M, Börner A (2010) Longevity of seeds—intraspecific differences in the Gatersleben genebank collections. In: Proc 60th Tagung der Vereinigung der Pflanzenzüchter und Saatgutkaufleute Österreichs, Gumpenstein, Österreich, 24–26 Nov 2009, pp 179–181
Nagel M, Rosenhauer M, Willner E, Snowdon RJ, Friedt W, Börner A (2011) Seed longevity in oilseed rape (Brassica napus L.)—genetic variation and QTL mapping. Plant Genet Resour 9:260–263
Nagel M, Kranner I, Neumann K, Rolletschek H, Seal C, Colville L, Fernández-Marín B, Börner A (2014) Genome-wide association mapping and biochemical markers reveal that seed ageing and longevity are intricately affected by genetic background, developmental and environmental conditions in barley. Plant Cell Environ. doi:10.1111/pce.12474
Priestley DA, Cullinan VI, Wolfe J (1985) Differences in seed longevity at the species level. Plant Cell Environ 8:557–562
Rehman Arif MA, Nagel M, Neumann K, Kobiljski B, Lohwasser U, Börner A (2012) Genetic studies of seed longevity in hexaploid wheat using segregation and association mapping approaches. Euphytica 186:1–13
Revilla P, Butrón A, Rodríguez VM, Malvar RA, Ordás A (2009) Identification of genes related to germination in aged maize seed by screening natural variability. J Exp Bot 60:4151–4157
Schwember AR, Bradford KJ (2010) Quantitative trait loci associated with longevity of lettuce seeds under conventional and controlled deterioration storage conditions. J Exp Bot 61:4423–4436
Shamel AD, Cobey WW (1907) Tobacco Breeding. US Dept Agric Bur Plant Ind Bull 96:71
Singh RK, Raipuria RK, Bhatia VS, Rani A, Pushpendra, Husain SM, Chauhan D, Chauhan GS, Mohapatra T (2008) SSR markers associated with seed longevity in soybean. Seed Sci Technol 36:162–167
Vontimitta V, Lewis RS (2012) Mapping of quantitative trait loci affecting resistance to Phytophthora nicotianae in tobacco (Nicotiana tabacum L.) line Beinhart-1000. Mol Breed 29:89–98
Walters C, Wheeler LM, Grotenhuis JM (2005) Longevity of seeds stored in a genebank: species characteristics. Seed Sci Res 15:1–20
Xiao B, Drake K, Vontimitta V, Tong Z, Zhang X, Li M, Leng X, Li Y, Lewis RS (2013) Location of genomic regions contributing to Phytophthora nicotianae resistance in tobacco cultivar Florida 301. Crop Sci 53:473–481
Xue Y, Zhang SQ, Yao QH, Peng RH, Xiong AS, Li X, Zhu WM, Zhu YY, Zha DS (2008) Identification of quantitative trait loci for seed storability in rice (Oryza sativa L.). Euphytica 164:739–744
Zeng ZB (1994) Precision mapping of quantitative trait loci. Genetics 136:1457–1468
Zeng DL, Guo LB, Xu YB, Yasukumi K, Zhu LH, Qian Q (2006) QTL analysis of seed storability in rice. Plant Breed 125:57–60
Acknowledgments
We thank Sibylle Pistrick and Gabriele Matzig for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agacka-Mołdoch, M., Nagel, M., Doroszewska, T. et al. Mapping quantitative trait loci determining seed longevity in tobacco (Nicotiana tabacum L.). Euphytica 202, 479–486 (2015). https://doi.org/10.1007/s10681-015-1355-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-015-1355-x