Abstract
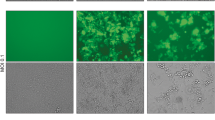
The urokinase receptor (uPAR) plays a critical role in breast cancer (BC) progression and metastases and is a validated target for novel therapies. The current study investigates the effects of MV-uPA, an oncolytic measles virus fully retargeted against uPAR in syngeneic and xenograft BC metastases models. In vitro replication and cytotoxicity of MVs retargeted against human (MV-h-uPA) or mouse (MV-m-uPA) uPAR were assessed in human and murine cancer and non-cancer mammary epithelial cells. The in vivo effects of species-specific uPAR retargeted MVs were assessed in syngeneic and xenograft models of experimental metastases, established by intravenous administration of luciferase expressing 4T1 or MDA-MD-231 cells. Metastases progression was assessed by in vivo bioluminescence imaging. Tumor targeting was evaluated by qRT-PCR of MV-N, rescue of viable viral particles, and immunostaining of MV particles in lungs from tumor bearing mice. In vitro, MV-h-uPA and MV-m-uPA selectively infected, replicated, and induced cytotoxicity in cancer compared to non-cancer cells in a species-specific manner. In vivo, MV-m-uPA delayed 4T1 lung metastases progression and prolonged survival. These effects were associated with identification of viable viral particles, viral RNA, and detection of MV-N by immunostaining from lung tissues in treated mice. In the human MDA-MB-231 metastases model, intravenous administration of MV-h-uPA markedly inhibited metastases progression and significantly improved survival, compared to controls. No significant treatment-related toxicity was observed in treated mice. The above preclinical findings strongly suggest that uPAR retargeted measles virotherapy is a novel and feasible systemic therapy strategy against metastatic breast cancer.





Similar content being viewed by others
References
Siegel R, Ma J, Zou Z, Jemal A (2014) Cancer statistics, 2014. CA Cancer J Clin 64(1):9–29
Chia SK, Speers CH, D’Yachkova Y, Kang A, Malfair-Taylor S, Barnett J, Coldman A, Gelmon KA, O’Reilly SE, Olivotto IA (2007) The impact of new chemotherapeutic and hormone agents on survival in a population-based cohort of women with metastatic breast cancer. Cancer 110(5):973–979
Tang L, Han X (2013) The urokinase plasminogen activator system in breast cancer invasion and metastasis. Biomed Pharmacother 67(2):179–182
Foekens JA, Peters HA, Look MP, Portengen H, Schmitt M, Kramer MD, Brunner N, Janicke F, Meijer-van Gelder ME, Henzen-Logmans SC et al (2000) The urokinase system of plasminogen activation and prognosis in 2780 breast cancer patients. Cancer Res 60(3):636–643
Harbeck N, Kates RE, Schmitt M (2002) Clinical relevance of invasion factors urokinase-type plasminogen activator and plasminogen activator inhibitor type 1 for individualized therapy decisions in primary breast cancer is greatest when used in combination. J Clin Oncol 20(4):1000–1007
Janicke F, Prechtl A, Thomssen C, Harbeck N, Meisner C, Untch M, Sweep CG, Selbmann HK, Graeff H, Schmitt M (2001) Randomized adjuvant chemotherapy trial in high-risk, lymph node-negative breast cancer patients identified by urokinase-type plasminogen activator and plasminogen activator inhibitor type 1. J Natl Cancer Inst 93(12):913–920
Look MP, van Putten WL, Duffy MJ, Harbeck N, Christensen IJ, Thomssen C, Kates R, Spyratos F, Ferno M, Eppenberger-Castori S et al (2002) Pooled analysis of prognostic impact of urokinase-type plasminogen activator and its inhibitor PAI-1 in 8377 breast cancer patients. J Natl Cancer Inst 94(2):116–128
Blasi F, Carmeliet P (2002) uPAR: a versatile signalling orchestrator. Nat Rev Mol Cell Biol 3(12):932–943
Jo M, Lester RD, Montel V, Eastman B, Takimoto S, Gonias SL (2009) Reversibility of epithelial-mesenchymal transition (EMT) induced in breast cancer cells by activation of urokinase receptor-dependent cell signaling. J Biol Chem 284(34):22825–22833
Romer J, Nielsen BS, Ploug M (2004) The urokinase receptor as a potential target in cancer therapy. Curr Pharm Des 10(19):2359–2376
Bianchi E, Cohen RL, Thor AT, Todd RF, Mizukami IF, Lawrence DA, Ljung BM, Shuman MA, Smith HS (1994) The urokinase receptor is expressed in invasive breast cancer but not in normal breast tissue. Cancer Res 54(4):861–866
Boyd D, Florent G, Kim P, Brattain M (1988) Determination of the levels of urokinase and its receptor in human colon carcinoma cell lines. Cancer Res 48(11):3112–3116
Dass K, Ahmad A, Azmi AS, Sarkar SH, Sarkar FH (2008) Evolving role of uPA/uPAR system in human cancers. Cancer Treat Rev 34(2):122–136
Lester RD, Jo M, Montel V, Takimoto S, Gonias SL (2007) uPAR induces epithelial-mesenchymal transition in hypoxic breast cancer cells. J Cell Biol 178(3):425–436
Ohba K, Miyata Y, Kanda S, Koga S, Hayashi T, Kanetake H (2005) Expression of urokinase-type plasminogen activator, urokinase-type plasminogen activator receptor and plasminogen activator inhibitors in patients with renal cell carcinoma: correlation with tumor associated macrophage and prognosis. J Urol 174(2):461–465
Rabbani SA, Xing RH (1998) Role of urokinase (uPA) and its receptor (uPAR) in invasion and metastasis of hormone-dependent malignancies. Int J Oncol 12(4):911–920
Smith HW, Marshall CJ (2010) Regulation of cell signalling by uPAR. Nat Rev Mol Cell Biol 11(1):23–36
Patel MR, Kratzke RA (2013) Oncolytic virus therapy for cancer: the first wave of translational clinical trials. Trans Res 161:355–364
Nakamura T, Russell SJ (2004) Oncolytic measles viruses for cancer therapy. Expert Opin Biol Ther 4(10):1685–1692
Russell SJ (2002) RNA viruses as virotherapy agents. Cancer Gene Ther 9(12):961–966
Galanis E, Hartmann LC, Cliby WA, Long HJ, Peethambaram PP, Barrette BA, Kaur JS, Haluska PJ Jr, Aderca I, Zollman PJ et al (2010) Phase I trial of intraperitoneal administration of an oncolytic measles virus strain engineered to express carcinoembryonic antigen for recurrent ovarian cancer. Cancer Res 70(3):875–882
Galanis E, Atherton PJ, Maurer MJ, Knutson KL, Dowdy SC, Cliby WA, Halulska P Jr., Long HJ, Oberg A, Aderca I et al. (2014) Oncolytic measles virus expressing the sodium iodide symporter to treat drug-resistant ovarian cancer. Cancer Res
Russell SJ, Federspiel MJ, Peng KW, Tong C, Dingli D, Morice WG, Lowe V, O’Connor MK, Kyle RA, Leung N et al (2014) Remission of disseminated cancer after systemic oncolytic virotherapy. Mayo Clin Proc 89(7):926–933
Jing Y, Tong C, Zhang J, Nakamura T, Iankov I, Russell SJ, Merchan JR (2009) Tumor and vascular targeting of a novel oncolytic measles virus retargeted against the urokinase receptor. Cancer Res 69(4):1459–1468
Sliutz G, Eder H, Koelbl H, Tempfer C, Auerbach L, Schneeberger C, Kainz C, Zeillinger R (1996) Quantification of uPA receptor expression in human breast cancer cell lines by cRT-PCR. Breast Cancer Res Treat 40(3):257–263
Nguyen DH, Hussaini IM, Gonias SL (1998) Binding of urokinase-type plasminogen activator to its receptor in MCF-7 cells activates extracellular signal-regulated kinase 1 and 2 which is required for increased cellular motility. J Biol Chem 273(14):8502–8507
Rabbani SA, Gladu J (2002) Urokinase receptor antibody can reduce tumor volume and detect the presence of occult tumor metastases in vivo. Cancer Res 62(8):2390–2397
Mazar AP, Henkin J, Goldfarb RH (1999) The urokinase plasminogen activator system in cancer: implications for tumor angiogenesis and metastasis. Angiogenesis 3(1):15–32
Li Y, Cozzi PJ (2007) Targeting uPA/uPAR in prostate cancer. Cancer Treat Rev 33(6):521–527
Jo M, Takimoto S, Montel V, Gonias SL (2009) The urokinase receptor promotes cancer metastasis independently of urokinase-type plasminogen activator in mice. Am J Pathol 175(1):190–200
Grondahl-Hansen J, Peters HA, van Putten WL, Look MP, Pappot H, Ronne E, Dano K, Klijn JG, Brunner N, Foekens JA (1995) Prognostic significance of the receptor for urokinase plasminogen activator in breast cancer. Clin Cancer Res 1(10):1079–1087
Yang L, Peng XH, Wang YA, Wang X, Cao Z, Ni C, Karna P, Zhang X, Wood WC, Gao X et al (2009) Receptor-targeted nanoparticles for in vivo imaging of breast cancer. Clin Cancer Res 15(14):4722–4732
D’Alessio S, Margheri F, Pucci M, Del Rosso A, Monia BP, Bologna M, Leonetti C, Scarsella M, Zupi G, Fibbi G et al (2004) Antisense oligodeoxynucleotides for urokinase-plasminogen activator receptor have anti-invasive and anti-proliferative effects in vitro and inhibit spontaneous metastases of human melanoma in mice. Int J Cancer 110(1):125–133
LeBeau AM, Duriseti S, Murphy ST, Pepin F, Hann B, Gray JW, VanBrocklin HF, Craik CS (2013) Targeting uPAR with antagonistic recombinant human antibodies in aggressive breast cancer. Cancer Res 73(7):2070–2081
Jing Y, Zaias J, Duncan R, Russell SJ, Merchan JR (2014) In vivo safety, biodistribution and antitumor effects of uPAR retargeted oncolytic measles virus in syngeneic cancer models. Gene Ther 21(3):289–297
Nakamura T, Peng KW, Harvey M, Greiner S, Lorimer IA, James CD, Russell SJ (2005) Rescue and propagation of fully retargeted oncolytic measles viruses. Nat Biotechnol 23(2):209–214
Liu C, Hasegawa K, Russell SJ, Sadelain M, Peng KW (2009) Prostate-specific membrane antigen retargeted measles virotherapy for the treatment of prostate cancer. Prostate 69(10):1128–1141
Hasegawa K, Nakamura T, Harvey M, Ikeda Y, Oberg A, Figini M, Canevari S, Hartmann LC, Peng KW (2006) The use of a tropism-modified measles virus in folate receptor-targeted virotherapy of ovarian cancer. Clin Cancer Res 12(20 Pt 1):6170–6178
Allen C, Vongpunsawad S, Nakamura T, James CD, Schroeder M, Cattaneo R, Giannini C, Krempski J, Peng KW, Goble JM et al (2006) Retargeted oncolytic measles strains entering via the EGFRvIII receptor maintain significant antitumor activity against gliomas with increased tumor specificity. Cancer Res 66(24):11840–11850
Allen C, Paraskevakou G, Iankov I, Giannini C, Schroeder M, Sarkaria J, Puri RK, Russell SJ, Galanis E (2008) Interleukin-13 displaying retargeted oncolytic measles virus strains have significant activity against gliomas with improved specificity. Mol Ther 16(9):1556–1564
Peng KW, Frenzke M, Myers R, Soeffker D, Harvey M, Greiner S, Galanis E, Cattaneo R, Federspiel MJ, Russell SJ (2003) Biodistribution of oncolytic measles virus after intraperitoneal administration into Ifnar-CD46Ge transgenic mice. Hum Gene Ther 14(16):1565–1577
Acknowledgments
This work was supported by a research grant from the National Cancer Institute (1R01CA149659-01 to JRM, JZ), and by the Sylvester Comprehensive Cancer Center (JRM).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jing, Y., Bejarano, M.T., Zaias, J. et al. In vivo anti-metastatic effects of uPAR retargeted measles virus in syngeneic and xenograft models of mammary cancer. Breast Cancer Res Treat 149, 99–108 (2015). https://doi.org/10.1007/s10549-014-3236-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-014-3236-8