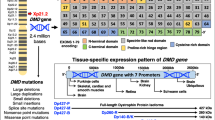
Abstract
Numerous biomedical advances have been made since Carl and Gerty Cori discovered the enzyme phosphorylase in the 1940s and the Scottish physician Brian McArdle reported in 1951 a previously ‘undescribed disorder characterized by a gross failure of the breakdown in muscle of glycogen’. Today we know that this disorder, commonly known as ‘McArdle disease’, is caused by inherited deficiency of the muscle isoform of glycogen phosphorylase (GP). Here we review the main aspects of the ‘pathogenomics’ of this disease including, among others: the spectrum of mutations in the gene (PYGM) encoding muscle GP; the interplay between the different tissue GP isoforms in cellular cultures and in patients; what can we learn from naturally occurring and recently laboratory-generated animal models of the disease; and potential therapies.


Similar content being viewed by others
References
Andersen ST, Vissing J (2008) Carbohydrate- and protein-rich diets in McArdle disease: effects on exercise capacity. J Neurol Neurosurg Psychiatry 79(12):1359–1363
Andersen ST, Haller RG, Vissing J (2008) Effect of oral sucrose shortly before exercise on work capacity in McArdle disease. Arch Neurol 65(6):786–789
Angelos S, Valberg SJ, Smith BP et al (1995) Myophosphorylase deficiency associated with rhabdomyolysis and exercise intolerance in 6 related Charolais cattle. Muscle Nerve 18(7):736–740
Aquaron R, Berge-Lefranc JL, Pellissier JF et al (2007) Molecular characterization of myophosphorylase deficiency (McArdle disease) in 34 patients from Southern France: identification of 10 new mutations. Absence of genotype-phenotype correlation. Neuromuscul Disord 17(3):235–241
Bartram C, Edwards RH, Clague J, Beynon RJ (1993) McArdle’s disease: a nonsense mutation in exon 1 of the muscle glycogen phosphorylase gene explains some but not all cases. Hum Mol Genet 2(8):1291–1293
Bartram C, Edwards RH, Clague J, Beynon RJ (1994) McArdle’s disease: a rare frameshift mutation in exon 1 of the muscle glycogen phosphorylase gene. Biochim Biophys Acta 1226(3):341–343
Bidou L, Hatin I, Perez N, Allamand V, Panthier JJ, Rousset JP (2004) Premature stop codons involved in muscular dystrophies show a broad spectrum of readthrough efficiencies in response to gentamicin treatment. Gene Ther 11(7):619–627
Bidou L, Allamand V, Rousset JP, Namy O (2012) Sense from nonsense: therapies for premature stop codon diseases. Trends Mol Med 18(11):679–688
Birch KE, Quinlivan RM, Morris GE (2013) Cell models for McArdle disease and aminoglycoside-induced read-through of a premature termination codon. Neuromuscul Disord 23(1):43–51
Bresolin N, Miranda AF, Jacobson MP, Lee JH, Capilupi T, Dimauro S (1983) Phosphorylase isoenzymes of human brain. Neurochem Pathol 1(3):171–178
Brooke MH, Patterson VH, Kaiser KK (1983) Hypoxanthine and Mcardle disease: a clue to metabolic stress in the working forearm. Muscle Nerve 6(3):204–206
Bruno C, Cassandrini D, Martinuzzi A et al (2006) McArdle disease: the mutation spectrum of PYGM in a large Italian cohort. Hum Mutat 27(7):718
David ES, Crerar MM (1986) Quantitation of muscle glycogen phosphorylase mRNA and enzyme amounts in adult rat tissues. Biochim Biophys Acta 880(1):78–90
Day TJ, Mastaglia FL (1985) Depot-glucagon in the treatment of McArdle’s disease. Aust N Z J Med 15(6):748–750
den Dunnen JT, Paalman MH (2003) Standardizing mutation nomenclature: why bother? Hum Mutat 22(3):181–182
Deschauer M, Morgenroth A, Joshi PR et al (2007) Analysis of spectrum and frequencies of mutations in McArdle disease. Identification of 13 novel mutations. J Neurol 254(6):797–802
Di Mauro S (2007) Muscle glycogenoses: an overview. Acta Myol 26(1):35–41
DiMauro S, Arnold S, Miranda A, Rowland LP (1977) McArdle disease: the mysterious appearance of phosphorylase activity in cells that ought to lack the genetic program. A fetal isoenzyme? Trans Am Neurol Assoc 102:112–115
DiMauro S, Arnold S, Miranda A, Rowland LP (1978) McArdle disease: the mystery of reappearing phosphorylase activity in muscle culture–a fetal isoenzyme. Ann Neurol 3(1):60–66
el-Schahawi M, Tsujino S, Shanske S, DiMauro S (1996) Diagnosis of McArdle’s disease by molecular genetic analysis of blood. Neurology 47(2):579–580
Fernandez-Cadenas I, Andreu AL, Gamez J et al (2003) Splicing mosaic of the myophosphorylase gene due to a silent mutation in McArdle disease. Neurology 61(10):1432–1434
Garcia-Consuegra I, Rubio JC, Nogales-Gadea G et al (2009) Novel mutations in patients with McArdle disease by analysis of skeletal muscle mRNA. J Med Genet 46(3):198–202
Gomez-Cabrera MC, Pallardo FV, Sastre J, Vina J, Garcia-del-Moral L (2003) Allopurinol and markers of muscle damage among participants in the Tour de France. JAMA 289(19):2503–2504
Gomez-Cabrera MC, Martinez A, Santangelo G, Pallardo FV, Sastre J, Vina J (2006) Oxidative stress in marathon runners: interest of antioxidant supplementation. Br J Nutr 96(Suppl 1):S31–S33
Gomez-Gallego F, Santiago C, Moran M et al (2008) The I allele of the ACE gene is associated with improved exercise capacity in women with McArdle disease. Br J Sports Med 42(2):134–140
Graves DJ, Fischer EH, Krebs EG (1960) Specificity studies on muscle phosphorylase phosphatase. J Biol Chem 235:805–809
Gurgel-Giannetti J, Nogales-Gadea G, van der Linden H Jr et al (2013) Clinical and molecular characterization of McArdle’s disease in Brazilian patients. Neuromol Med 15(3):470–475
Hadjigeorgiou GM, Sadeh M, Musumeci O et al (2002) Molecular genetic study of myophosphorylase deficiency (McArdle’s disease) in two Yemenite-Jewish families. Neuromuscul Disord 12(9):824–827
Haller RG, Clausen T, Vissing J (1998) Reduced levels of skeletal muscle Na + K + −ATPase in McArdle disease. Neurology 50(1):37–40
Haller RG, Wyrick P, Taivassalo T, Vissing J (2006) Aerobic conditioning: an effective therapy in McArdle’s disease. Ann Neurol 59(6):922–928
Howell JM, Quinlinvan R, Sewry C (2008a) Investigation of possible treatment regimes for McArdle’s disease using the sheep model of the disease [Poster:G.P.16.06]. Neuromuscul Disord 18:724–833
Howell JM, Walker KR, Davies L et al (2008b) Adenovirus and adeno-associated virus-mediated delivery of human myophosphorylase cDNA and LacZ cDNA to muscle in the ovine model of McArdle’s disease: expression and re-expression of glycogen phosphorylase. Neuromuscul Disord 18(3):248–258
Isackson PJ, Tarnopolsky M, Vladutiu GD (2005) A novel mutation in the PYGM gene in a family with pseudo-dominant transmission of McArdle disease. Mol Genet Metab 85(3):239–242
Johannessen CU (2000) Mechanisms of action of valproate: a commentatory. Neurochem Int 37(2–3):103–110
Kitaoka Y, Ogborn DI, Nilsson MI, Mocellin NJ, MacNeil LG, Tarnopolsky MA (2013) Oxidative stress and Nrf2 signaling in McArdle disease. Mol Genet Metab 110(3):297–302
Lane RJ, Turnbull DM, Welch JL, Walton J (1986) A double-blind, placebo-controlled, crossover study of verapamil in exertional muscle pain. Muscle Nerve 9(7):635–641
Lebo RV, Gorin F, Fletterick RJ et al (1984) High-resolution chromosome sorting and DNA spot-blot analysis assign McArdle’s syndrome to chromosome 11. Science 225(4657):57–59
Lederer B, Van Hoof F, Van den Berghe G, Hers H (1975) Glycogen phosphorylase and its converter enzymes in haemolysates of normal human subjects and of patients with type VI glycogen-storage disease. A study of phosphorylase kinase deficiency. Biochem J 147(1):23–35
Lucia A, Nogales-Gadea G, Perez M, Martin MA, Andreu AL, Arenas J (2008) McArdle disease: what do neurologists need to know? Nat Clin Pract Neurol 4(10):568–577
Lucia A, Ruiz JR, Santalla A et al (2012) Genotypic and phenotypic features of McArdle disease: insights from the Spanish national registry. J Neurol Neurosurg Psychiatry 83(3):322–328
Lucia A, Quinlivan R, Wakelin A, Martin MA, Andreu AL (2013) The 'McArdle paradox': exercise is a good advice for the exercise intolerant. Br J Sports Med 47(12):728–729
MacLean D, Vissing J, Vissing SF, Haller RG (1998) Oral branched-chain amino acids do not improve exercise capacity in McArdle disease. Neurology 51(5):1456–1459
Martin MA, Rubio JC, Wevers RA et al (2004) Molecular analysis of myophosphorylase deficiency in Dutch patients with McArdle’s disease. Ann Hum Genet 68(Pt 1):17–22
Martinuzzi A, Vergani L, Carrozzo R et al (1993) Expression of muscle-type phosphorylase in innervated and aneural cultured muscle of patients with myophosphorylase deficiency. J Clin Invest 92(4):1774–1780
Martinuzzi A, Schievano G, Nascimbeni A, Fanin M (1999) McArdle’s disease. The unsolved mystery of the reappearing enzyme. Am J Pathol 154(6):1893–1897
Martinuzzi A, Sartori E, Fanin M et al (2003) Phenotype modulators in myophosphorylase deficiency. Ann Neurol 53(4):497–502
Martinuzzi A, Liava A, Trevisi E et al (2008) Randomized, placebo-controlled, double-blind pilot trial of ramipril in McArdle’s disease. Muscle Nerve 37(3):350–357
Mate-Munoz JL, Moran M, Perez M et al (2007) Favorable responses to acute and chronic exercise in McArdle patients. Clin J Sport Med 17(4):297–303
McArdle B (1951) Myopathy due to a defect in muscle glycogen breakdown. Clin Sci 10(1):13–35
Meienhofer MC, Askanas V, Proux-Daegelen D, Dreyfus JC, Engel WK (1977) Muscle-type phosphorylase activity present in muscle cells cultured from three patients with myophosphorylase deficiency. Arch Neurol 34(12):779–781
Mineo I, Kono N, Shimizu T et al (1985) Excess purine degradation in exercising muscles of patients with glycogen storage disease types V and VII. J Clin Invest 76(2):556–560
Mitsumoto H (1979) McArdle disease: phosphorylase activity in regenerating muscle fibers. Neurology 29(2):258–262
Newgard CB, Nakano K, Hwang PK, Fletterick RJ (1986) Sequence analysis of the cDNA encoding human liver glycogen phosphorylase reveals tissue-specific codon usage. Proc Natl Acad Sci U S A 83(21):8132–8136
Newgard CB, Littman DR, van Genderen C, Smith M, Fletterick RJ (1988) Human brain glycogen phosphorylase. Cloning, sequence analysis, chromosomal mapping, tissue expression, and comparison with the human liver and muscle isozymes. J Biol Chem 263(8):3850–3857
Newgard CB, Hwang PK, Fletterick RJ (1989) The family of glycogen phosphorylases: structure and function. Crit Rev Biochem Mol Biol 24(1):69–99
Nogales-Gadea G, Rubio JC, Fernandez-Cadenas I et al (2008) Expression of the muscle glycogen phosphorylase gene in patients with McArdle disease: the role of nonsense-mediated mRNA decay. Hum Mutat 29(2):227–283
Nogales-Gadea G, Mormeneo E, Garcia-Consuegra I et al (2010) Expression of glycogen phosphorylase isoforms in cultured muscle from patients with McArdle’s disease carrying the p.R771PfsX33 PYGM mutation. PLoS One 5(10)
Nogales-Gadea G, Pinos T, Lucia A et al (2012) Knock-in mice for the R50X mutation in the PYGM gene present with McArdle disease. Brain 135(Pt 7):2048–2057
Perez M, Mate-Munoz JL, Foster C et al (2007a) Exercise capacity in a child with McArdle disease. J Child Neurol 22(7):880–882
Perez M, Moran M, Cardona C et al (2007b) Can patients with McArdle’s disease run? Br J Sports Med 41(1):53–54
Perez M, Foster C, Gonzalez-Freire M, Arenas J, Lucia A (2008) One-year follow-up in a child with McArdle disease: exercise is medicine. Pediatr Neurol 38(2):133–136
Pernow B, Saltin B (1971) Availability of substrates and capacity for prolonged heavy exercise in man. J Appl Physiol 31(3):416–422
Phoenix J, Hopkins P, Bartram C, Beynon RJ, Quinlivan RC, Edwards RH (1998) Effect of vitamin B6 supplementation in McArdle’s disease: a strategic case study. Neuromuscul Disord 8(3–4):210–212
Poels PJ, Braakhekke JP, Joosten EM, Stegeman DF (1990) Dantrolene sodium does influence the second-wind phenomenon in McArdle’s disease. Electrophysiological evidence during exercise in a double-blind placebo-controlled, cross-over study in 5 patients. J Neurol Sci 100(1–2):108–112
Proux D, Vibert M, Meienhofer MC, Dreyfus JC (1974) The isozymes of glycogen phosphorylase in human and rabbit tissues. II. Electrofocusing in polyacrylamide gels. Clin Chim Acta 57(3):211–216
Rubio JC, Garcia-Consuegra I, Nogales-Gadea G et al (2007a) A proposed molecular diagnostic flowchart for myophosphorylase deficiency (McArdle disease) in blood samples from Spanish patients. Hum Mutat 28(2):203–204
Rubio JC, Gomez-Gallego F, Santiago C et al (2007b) Genotype modulators of clinical severity in McArdle disease. Neurosci Lett 422(3):217–222
Russo PJ, Phillips JW, Seidler NW (1992) The role of lipid peroxidation in McArdle’s disease: applications for treatment of other myopathies. Med Hypotheses 39(2):147–151
Sanchis-Gomar F, Pareja-Galeano H, Gomez-Cabrera MC et al (2014) Allopurinol prevents cardiac and skeletal muscle damage in professional soccer players. Scand J Med Sci Sports
Sato K, Imai F, Hatayama I, Roelofs RI (1977) Characterization of glycogen phosphorylase isoenzymes present in cultured skeletal muscle from patients with McArdle’s disease. Biochem Biophys Res Commun 78(2):663–668
Sato S, Ohi T, Nishino I, Sugie H (2012) Confirmation of the efficacy of vitamin B6 supplementation for McArdle disease by follow-up muscle biopsy. Muscle Nerve 45(3):436–440
Schroers A, Kley RA, Stachon A et al (2006) Gentamicin treatment in McArdle disease: failure to correct myophosphorylase deficiency. Neurology 66(2):285–286
Shulman RG, Hyder F, Rothman DL (2001) Cerebral energetics and the glycogen shunt: neurochemical basis of functional imaging. Proc Natl Acad Sci U S A 98(11):6417–6422
Simoni RD, Hill RL, Vaughan M (2002) Carbohydrate metabolism: glycogen phosphorylase and the work of Carl F. and Gerty T. Cori. 1928–1943. J Biol Chem 277(29):18e
Sohn EH, Kim HS, Lee AY, Fukuda T, Sugie H, Kim DS (2008) A novel PYGM mutation in a Korean patient with McArdle disease: the role of nonsense-mediated mRNA decay. Neuromuscul Disord 18(11):886–889
Steele IC, Patterson VH, Nicholls DP (1996) A double blind, placebo controlled, crossover trial of D-ribose in McArdle’s disease. J Neurol Sci 136(1–2):174–177
Tan P, Allen JG, Wilton SD, Akkari PA, Huxtable CR, Laing NG (1997) A splice-site mutation causing ovine McArdle’s disease. Neuromuscul Disord 7(5):336–342
Tork S, Hatin I, Rousset JP, Fabret C (2004) The major 5' determinant in stop codon read-through involves two adjacent adenines. Nucleic Acids Res 32(2):415–421
Tsujino S, Shanske S, DiMauro S (1993) Molecular genetic heterogeneity of myophosphorylase deficiency (McArdle’s disease). N Engl J Med 329(4):241–245
Tsujino S, Shanske S, Nonaka I, DiMauro S (1995) The molecular genetic basis of myophosphorylase deficiency (McArdle’s disease). Muscle Nerve Suppl 3:S23–S27
Tsujino S, Shanske S, Valberg SJ, Cardinet GH 3rd, Smith BP, DiMauro S (1996) Cloning of bovine muscle glycogen phosphorylase cDNA and identification of a mutation in cattle with myophosphorylase deficiency, an animal model for McArdle’s disease. Neuromuscul Disord 6(1):19–26
Tunnicliff G (1999) Actions of sodium valproate on the central nervous system. J Physiol Pharmacol 50(3):347–365
Vieitez I, Teijeira S, Fernandez JM et al (2011) Molecular and clinical study of McArdle’s disease in a cohort of 123 European patients. Identification of 20 novel mutations. Neuromuscul Disord 21(12):817–823
Vissing J, Haller RG (2003) The effect of oral sucrose on exercise tolerance in patients with McArdle’s disease. N Engl J Med 349(26):2503–2509
Vorgerd M, Grehl T, Jager M et al (2000) Creatine therapy in myophosphorylase deficiency (McArdle disease): a placebo-controlled crossover trial. Arch Neurol 57(7):956–963
Vorgerd M, Zange J, Kley R et al (2002) Effect of high-dose creatine therapy on symptoms of exercise intolerance in McArdle disease: double-blind, placebo-controlled crossover study. Arch Neurol 59(1):97–101
Welch EM, Barton ER, Zhuo J et al (2007) PTC124 targets genetic disorders caused by nonsense mutations. Nature 447(7140):87–91
Williams AG, Rayson MP, Jubb M et al (2000) The ACE gene and muscle performance. Nature 403(6770):614
Wolfe GI, Baker NS, Haller RG, Burns DK, Barohn RJ (2000) McArdle’s disease presenting with asymmetric, late-onset arm weakness. Muscle Nerve 23(4):641–645
Wu Y, Weber JL, Vladutiu GD, Tarnopolsky MA (2011) Six novel mutations in the myophosphorylase gene in patients with McArdle disease and a family with pseudo-dominant inheritance pattern. Mol Genet Metab 104(4):587–591
Acknowledgments
Research in the field of McArdle disease by Alejandro Lucia, Alfredo Santalla and Tomàs Pinós is funded by the Fondo de Investigaciones Sanitarias (FIS, grant numbers PI12/00914, PI13/00855). Gisela Nogales-Gadea and Noemi de Luna are supported by Sara Borrell contracts of ISCIII CD10/00027 and CD11/00060, respectively. Astrid Brull is supported by a FIS grant of ISCIII FI11/00709.
Compliance with Ethics Guidelines
ᅟ
Conflict of interest
None.
Informed consent and animal rights
This article does not contain any studies with human or animal subjects performed by the any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Bruce A. Barshop
Rights and permissions
About this article
Cite this article
Nogales-Gadea, G., Santalla, A., Brull, A. et al. The pathogenomics of McArdle disease—genes, enzymes, models, and therapeutic implications. J Inherit Metab Dis 38, 221–230 (2015). https://doi.org/10.1007/s10545-014-9743-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-014-9743-2