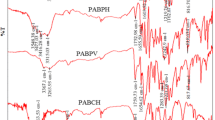
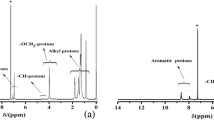
Abstract
The cholesteric polysiloxanes (P series) were obtained by reacting cholesteric monomer and phenolic hydroxyl monomer in different ratios with polysiloxanes. And then the chiral azo-containing polysiloxanes (AP series) were synthesized by esterifying P series members with the acryl acid of azo acid catalyzed by DMAP. The chemical structures and liquid crystal (LC) properties of the monomers and polymers were characterized by use of various experimental techniques such as FTIR, 1H-NMR, POM, DSC, TGA, XRD and ultraviolet-visible. Experimental results proved that obtained polymers were in accordance with the molecular design. The transition temperatures of the polymers exhibited a decreasing trend as the content of the cholesteric units increased and became higher by introducing the azo mesogenic core. The temperatures at which 5 % weight loss occurred are higher than 280 °C. P2–P6 showed blue Grandjean textures and exhibited selective reflection in the visible light region. AP series also possessed Grandjean textures, and the colors exhibited red shift with increasing content of azo moiety. On the heating cycles, when appropriate mechanical pressure was imposed on the polymers, AP2–AP6 selectively reflect visible light; however, there are no reflection peaks in the UV-Vis spectrum without the stimulation of mechanical pressure. All polymers exhibit left-handed optical activity due to having the same cholesteric group. The optical rotation direction of AP changes from left to right when increasing the UV irradiation time, and the photoresponsive behaviors of AP series are also investigated.









Similar content being viewed by others
References
Natansohn A, Rochon P (2002) Photoinduced motions in azo-containing polymers. Chem Rev 102(11):4139–4175
Archut A, Azzellini GC, Balzani V, et al. (1998) Toward photoswitchable dendritic hosts. Interaction between azobenzene-functionalized dendrimers and eosin. J Am Chem So 120(47):12187–12191
Jiang DL, Aida T (1997) Photoisomerization in dendrimers by harvesting of low-energy photons. Nature 388(6641):454–456
Lin PC, Cong YH, Sun C, Zhang BY (2016) Non-covalent modification of reduced graphene oxide by a chiral liquid crystalline surfactant. Nanoscale 8(4):2403–2411
Marin L, Zabulica A, Sava M (2011) New symmetric azomethinic dimer: the influence of structural heterogeneity on the liquid crystalline behaviour. Liq Cryst 38(4):433–440
Andruzzi L, D’Apollo F, Galli G, et al. (2001) Synthesis and structure characterization of liquid crystalline polyacrylates with unconventional fluoroalkylphenyl mesogens. Macromolecules 34(22):7707–7714
Bobrovsky A, Shibaev V, Hamplova V, et al. (2016) Photo-optical properties of amorphous and crystalline films of azobenzene-containing photochromes with bent-shaped molecular structure. Photoch Photobio A 316:75–87
Bobrovsky A, Shibaev V, Bubnov A, et al. (2013) Effect of molecular structure on chiro-optical and photo-optical properties of smart liquid crystalline polyacrylates. Macromolecules 46(11):4276–4284
Ryabchun A, Bobrovsky A, Stumpe J, et al. (2015) Rotatable diffraction gratings based on cholesteric liquid crystals with phototunable helix pitch. Adv Opt Mater 3(9):1273–1279
Meng FB, He XZ, Zhang XD, et al. (2011) Effect of terminal perfluorocarbon chain containing mesogens on phase behaviors of chiral comb-like liquid crystalline polymers. Colloid Polym Sci 289(8):955–965
Han DH, Tong X, Zhao Y, et al. (2010) Cyclic azobenzene-containing side-chain liquid crystalline polymers: synthesis and topological effect on mesophase transition, order, and photoinduced birefringence. Macromolecules 43(8):3664–3671
Gimenez R, Millaruelo M, Pinol M, et al. (2005) Synthesis, thermal and optical properties of liquid crystalline terpolymers containing azobenzene and dye moieties. Polymer 46(22):9230–9242
Hattori H, Uryu T (2000) Synthesis and properties of photochromic liquid-crystalline copolymers containing both spironaphthoxazine and cholesteryl groups. J Polym Sci Pol Chem 38(5):887–894
Statman D, Basore V, Sulai Y, et al. (2008) Photoinduced gliding of the surface director in azo-dye doped nematic liquid crystals. Liq Cryst 35(1):33–38
Singh U, Davis F, Mohan S, Mitchell G (2013) Electro-active nanofibres electrospun from blends of poly-vinyl cinnamate and a cholesteric liquid crystalline silicone polymer. J Mater Sci 48(21):7613–7619
Han M, Morino S, Ichimura K (2000) Factors affecting in-plane and out-of-plane photo orientation of azobenzene side chains attached to liquid crystalline polymers induced by irradiation with linearly polarized light. Macromolecules 33(17):6360–6371
Hvilsted S, Andruzzi F, Kulinna C, et al. (1995) Novel side-chain liquid-crystalline polyester architecture for reversible optical storage. Macromolecules 28(7):2172–2183
Apreutesei D, Mehl GH, Scutaru D (2007) Ferrocene-containing liquid crystals bearing a cholesteryl unit. Liq Cryst 34(7):819–831
Lin PC, Cong YH, Zhang BY (2015) Dispersing carbon nanotubes by chiral network surfactants. ACS Appl Mater Interfaces 7(12):6724–6732
Agnieszka I, Boharewicz B, Tazbir I, et al. (2015) Laser beam induced current technique of polymer solar cells based on new poly(azomethine) or poly(3-hexylthiophene. Solid State Electron 104:53–63
Abrakhi S, Peralta S, Cantin S, et al. (2012) Synthesis and characterization of photosensitive cinnamate-modified cellulose acetate butyrate spin-coated or network derivatives. Colloid Polym Sci 290(5):423–434
Tejedor RM, Oriol L, Serrano J, et al. (2007) Photoinduced chiral nematic organization in an achiral glassy nematic azopolymer. Adv Funct Mater 17:3486–3492
Bobrovsky A, Shibaev V (2006) A study of photooptical processes in photosensitive cholesteric azobenzene-containing polymer mixture under an action of the polarized and nonpolarized light. Polymer 47(12):4310–4317
Andruzzi L, Altomare A, Ciardelli F, et al. (1999) Holographic gratings in azobenzene side-chain polymethacrylates. Macromolecules 32:448–454
Viswanathan NK, Kim DY, Bian S, et al. (1999) Surface relief structures on azo polymer films. J Mater Chem 9(9):1941–1955
Karim MR, Sheikh MRK, Yahya R, et al. (2015) Synthesis of polyme-rizable liquid crystalline monomers and their side chain liquid crystalline polymers bearing azo-ester linked benzothiazole mesogen. Colloid Polym Sci 293:1923–1935
Kanazawa A, Hirano S, Shishido A, et al. (1997) Photochemical phase transition behaviour of polymer azobenzene liquid crystals with flexible siloxane units as a side-chain spacer. Liq Cryst 23(2):293–298
Jui-Hsiang L, Yang P-C, Wang Y-K, et al. (2006) Optical behaviour of cholesteric liquid crystal cells with novel photoisomerizable chiral dopants. Liq Cryst 33(3):237–248
Ho MS, Natansohn A, Rochon P (1996) Synthesis and optical properties of poly{(4-nitrophenyl)-[3-[N-[2-(methacryloyloxy)ethyl]-carbazolyol]]diazene}. Macromolecules 29(1):44–49
Zettsu N, Ogasawara T, Mizoshita N, et al. (2008) Photo-triggered surface relief grating formation in supramolecular liquid crystalline polymer systems with detachable azobenzene units. Adv Mater 20(3):516–521
Forcen P, Oriol L, Sanchez C, et al. (2007) Synthesis, characterization and photoinduction of optical anisotropy, in liquid crystalline diblock azo-copolymers. J Polym Sci Pol Chem 45(10):1899–1910
Bobrovsky A, Shibaev V, Hamplova V, et al. (2010) Gel formation and photoactive properties of azobenzene-containing polymer in liquid crystal mixture. Colloid Polym Sci 288:1375–1384
Srinivasan MV, Kannan P, Roy A (2013) Photo and electrically switchable behavior of azobenzene containing pendant bent-core liquid crystalline polymers. J Polym Sci Pol Chem 51:936–946
Yang ZQ, Herd GA, Clarke SM, et al. (2006) Thermal and UV shape shifting of surface topography. J Am Chem Soc 128:1074–1075
Delaire J, Nakatani K (2000) Linear and nonlinear optical properties of photochromic molecules and materials. Chem Rev 100:1817–1846
Mahimwalla Z, Yager KG, Jun-ichi, et al. (2012) Azobenzene photomechanics: prospects and potential applications. Polym Bull 69:967–1006
Ana-Maria R, Luiza E, Nicolae H (2010) Surface properties, thermal behaviorand molecular simulation of azo-polysiloxanes under light stimuli. Insight into the relaxation. Macromol Res 18:721–729
Zhou QL, Yan SK, Han CC, et al. (2008) Promising functional materials based on ladder polysiloxanes. Adv Mater 20:2970–2976
Kaspar M, Bubnov A, Hamplova V, et al. (2004) New ferroelectric liquid crystalline materials with an azo group in the molecular core. Liq Crtyst 31(6):821–830
Cigl M, Fodor-Csorba K, et al. (2014) Functional photochromic methylhydrosiloxane -based side-chain liquid crystalline polymer. Macromol Chem Phys 215:742–752
Abe J, Hasegawa M, Matsushina H, et al. (1995) Investigation of dipolar alignment of mesogenic chromophores in side chain liquid crystalline polysiloxane using electric field induced second harmonic generation. Macromolecules 28(8):2938–2943
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by Fundamental Research Funds for the Central Universities (N130205001), The National Natural Science Foundation (51273035), and the Scientific and Technical Bureau Foundation of Shen Yang City (F16-205-1-03).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
He, XZ., Gao, YF., Zheng, JJ. et al. Chiral photosensitive side-chain liquid crystalline polymers—synthesis and characterization. Colloid Polym Sci 294, 1823–1832 (2016). https://doi.org/10.1007/s00396-016-3939-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-016-3939-y