Abstract
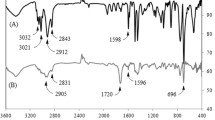
Free radical polymerization of methyl methacrylate (MMA) and styrene (St) has been performed in N,N-dimethylformamide (DMF) at 70 °C in the presence of varying concentration of mangiferin as chain transfer agent using 2,2′-azobisisobutyronitrile (AIBN) as initiator. With increase in mangiferin concentration, molecular weight of the obtained polySt gradually decreases as expected due to the chain transfer phenomenon, but that of polyMMA interestingly increases possibly due to the preferential auto-acceleration phenomenon during MMA polymerization in the presence of higher concentration of mangiferin. The observed chain transfer constant of mangiferin for styrene polymerization is 0.14. The incorporation of mangiferin in the resultant polymers is confirmed by FTIR and UV–Vis studies.
Graphical abstract






Similar content being viewed by others
References
Masibo M, He Q (2008) Major mango polyphenols and their potential significance to human health. Compr Rev Food Sci Food Saf 7:309–319
Chattopadhyay S, Chattopadhyay U, Shukla SP, Ghosal S (1984) Effect of mangiferin, a naturally occurring glycosylxanthone, on reproductory function of rats. Pharm Res 1(6):279–282
Sanchez GM, Re L, Giuliani A, Nunez-Selles AJ, Davison GP, Leon-Fernandez OS (2000) Protective effects of Mangifera indica L. extract, mangiferin and selected antioxidants against TPA-induced biomolecules oxidation and peritoneal macrophage activation in mice. Pharmacol Res 42(6):565–573
Muruganandan S, Gupta S, Kataria M, Lal J, Gupta PK (2002) Mangiferin protects the streptozotocin-induced oxidative damage to cardiac and renal tissues in rats. Toxicology 176(3):165–173
Garrido G, Delgado R, Lemus Y, Rodriguez J, Garcia D, Nunez-Selles AJ (2004) Protection against septic shock and suppression of tumor necrosis factor alpha and nitric oxide production on macrophages and microglia by a standard aqueous extract of Mangifera indica L. (VIMANG). Role of mangiferin isolated from the extract. Pharmacol Res 50(2):165–172
Rodriguez J, Pierro DD, Gioia M, Monaco S, Delgado R, Coletta M et al (2006) Effects of a natural extract from Mangifera indica L, and its active compound, mangiferin, on energy state and lipid peroxidation of red blood cells. Biochim Biophys Acta 1760(9):1333–1342
Andreu GP, Delgado R, Velho JA, Curti C, Vercesi AE (2005) Iron complexing activity of mangiferin, a naturally occurring glucosylxanthone, inhibits mitochondrial lipid peroxidation induced by Fe2+-citrate. Eur J Pharm 513(1–2):47–55
Leiro JM, Alvarez E, Arranz JA, Siso IG, Orallo F (2003) In vitro effects of mangiferin on superoxide concentrations and expression of the inducible nitric oxide synthase, tumour necrosis factor-alpha and transforming growth factor-beta genes. Biochem Pharm 65(8):1361–1371
Guha S, Ghosal S, Chattopadhyay U (1996) Antitumor, immunomodulatory and anti-HIV effect of mangiferin, a naturally occurring glucosylxanthone. Chemotherapy 42(6):443–445
Yoosook C, Bunyapraphatsara N, Boonyakiat Y, Kantasuk C (2000) Anti-herpes simplex virus activities of crude water extracts of Thai medicinal plants. Phytomedicine 6(6):411–419
Zheng MS, Lu ZY (1990) Antiviral effect of mangiferin and isomangiferin on herpes simplex virus. Chin Med J Peking 103(2):160–165
Zhu XM, Song JX, Huang ZZ, Wu YM, Yu MJ (1993) Antiviral activity of mangiferin against herpes simplex virus type 2 in vitro. Acta Pharmacol Sin 14(5):452–454
Stoilova I, Gargova S, Stoyanova A, Ho L (2005) Antimicrobial and antioxidant activity of the polyphenol mangiferin. Herb Polonica 51(1–2):37–43
Garcia D, Leiro J, DelgadoSanmartin RML, Ubeira FM (2003) Mangifera indica L. extract (Vimang) and mangiferin modulate mouse humoral immune responses. Phytother Res 17(10):1182–1187
Leiro J, Arranz JA, Yanez M, Ubeira FM, Sanmartin M, Orallo LF (2004) Expression profiles of genes involved in the mouse nuclear factor-kappa B signal transduction pathway are modulated by mangiferin. Int Immunopharmacol 4(6):763–778
Sarkar A, Sreenivasan Y, Ramesh GT, Manna SK (2004) beta-d-Glucoside suppresses tumor necrosis factor-induced activation of nuclear transcription factor kappaB but potentiates apoptosis. J Biol Chem 279(32):33768–33781
Ichiki H, Miura T, Kubo M, Ishihara E, Komatsu Y, Tanigawa K et al (1998) New antidiabetic compounds, mangiferin and its glucoside. Biol Pharm Bull 21(12):1389–1390
Miura T, Ichiki H, Hashimoto I, Iwamoto N, Kato M, Kubo M et al (2001) Antidiabetic activity of a xanthone compound, mangiferin. Phytomedicine 8(2):85–87
Rajtar G, Zolkowska D, Kleinrok Z, Marona H (1999) Antiplatelets activity of some xanthone derivatives. Acta Pol Pharm 56(4):319–324
Lin CN, Hsieh HK, Liou SJ, Ko HH, Lin HC, Chung MI et al (1996) Synthesis and antithrombotic effect of xanthone derivatives. J Pharm Pharmacol 48(9):887–890
Lin CN, Chung MI, Liou SJ, Lee TH, Wang JP (1996) Synthesis and anti-inflammatory effects of xanthone derivatives. J Pharm Pharmacol 48(5):532–538
Matkar RA, Kyu T (2006) Phase diagrams of binary crystalline–crystalline polymer blends. J Phys Chem B 110(32):16059–16065
Dayal P, Matkar RA, Kyu T (2006) Crystal–liquid crystal binary phase diagrams. J Chem Phys 124(22):224902–224907
Neelakandan C, Kyu T (2009) Hydrogen bonding interactions and miscibility studies of poly(amide)/poly-(vinyl pyrrolidone) blends containing mangiferin. Polymer 50:2885–2892
Neelakandan C, Kyu T (2009) Miscibility characterization in relation to phase morphology of poly(ether sulfone)/poly(vinyl pyrrolidone) blends containing a phytochemical. J Phys Chem B 113(25):8520–8526
Wauthoz N, Balde A, Balde ES, Damme MV, Duez P (2007) Ethnopharmacology of Mangifera indica L. bark and pharmacological studies of its main C glucosylxanthone, mangiferin. Int J Biomed Pharm Sci 1(2):112–119
Dar A, Faizi S, Naqvi S, Roome T, Rehman SZ, Ali M, Firdous S, Moin ST (2005) Analgesic and antioxidant activity of mangiferin and its derivatives: the structure activity relationship. Biol Pharm Bull 28(4):596–600
Stoilova I, Jirovetz L, Stoyanovan A, Krastanov A, Gargova S, Ho L (2008) Antioxidant activity of the polyphenol mangiferin. Electron J Environ Agric Food Chem 7(13):2706–2716
Godsay MP, Harpell GA, Russell KE (1962) The effect of phenols on the polymerization of styrene. J Polym Sci 57(165):641–650
Barton SC, Bird RA, Russell KE (1963) The effect of phenols and aromatic thiols on the polymerization of methyl methacrylate. Can J Chem 41:2737–2742
Parnell RD, Russel KE (2003) Polymerization of vinyl acetate retarded by 4- methyl- 2, 6- di-tert-butylphenol: kinetic and ESR studies. J Polym Sci Part A Polym Chem 12(2):347–356
Fujisawa S, Kadoma Y (1992) Effect of phenolic compounds on the polymerization of methyl methacrylate. Dent Mater 8(5):324–326
Bordwell FG, Zhang XM, Satish AV, Cheng JP (1994) Assessment of the importance of changes in ground-state energies on the bond dissociation enthalpies of the O–H bonds in phenols and the S–H bonds in thiophenols. J Am Chem Soc 116(15):6605–6610
DiLabio GA, Pratt DA, LoFaro AD, Wright JS (1999) Theoretical study of X–H bond energetics (X=C, N, O, S): application to substituent effects, gas phase acidities, and redox potentials. J Phys Chem A 103:1653–1661
Laarhoven LJJ, Mulder P, Wagner DDM (1999) Determination of bond dissociation enthalpies in solution by photoacoustic calorimetry. Acc Chem Res 32:342–349
Foti M, Ingold KU, Lusztyk J (1994) The surprisingly high reactivity of phenoxyl radicals. J Am Chem Soc 116(15):9440–9447
Ito O (1995) Flash photolysis study on reversible addition reactions of thiyl radicals. Res Chem Intermed 21(1):69–93
Wardman P, Von Sonntag C (1995) Kinetic factors that control the fate of thiyl radicals in cells. Methods Enzymol 251:31–41
Valdebenito A, Encinas MV (2008) Chain transfer agents in vinyl polymerizations photo induced by bimolecular photo initiators. J Photochem Photobiol Chem 194(2–3):206–211
Ghosal S, Rao G, Sarvanan V, Misra N, Rana D (1996) A plausible chemical mechanism of the bioactivities of mangiferin. Indian J Chem 35B:561–566
Ray B, Mandal BM (1997) Dispersion polymerization of acrylamide. Langmuir 13:2192–2196
Guha S, Ray B, Mandal BM (2001) Anomalous solubility of polyacrylamide prepared by dispersion (precipitation) polymerization in aqueous tert-butyl alcohol. J Polym Sci Part A Polym Chem 39:3434–3442
Acknowledgments
We acknowledge the financial support of Banaras Hindu University in the form of fellowship. The author would also like to thank the financial support of Council of Scientific and Industrial Research (CSIR) in the form of Senior Research Fellowship (SRF).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jaiswal, S., Ramesh, K., Kapusetti, G. et al. Mangiferin as chain transfer agent: effect on the molecular weight of poly(methyl methacrylate) and polystyrene. Polym. Bull. 72, 1407–1416 (2015). https://doi.org/10.1007/s00289-015-1343-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-015-1343-2