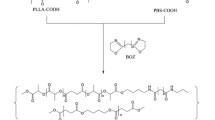
Abstract
High molecular weight poly(l-lactic acid) has been prepared from the corresponding functionally terminated oligomers employing a polyesterification method at room temperature using diisopropylcarbodiimide and 4-(dimethylamino) pyridinium-p-toluene sulfonate. Self-condensation of hydroxyl and carboxylic acid-terminated poly(l-lactic acid) oligomers (M n : ~1,000) resulted in polymers with high molecular weights (M n > 45,000) under mild conditions. End-group analysis by MALDI-TOF provided evidence for N-acylurea formation in the product under the reaction conditions employed.




Similar content being viewed by others
References
Auras RA, Lim L-T, Selke SEM, Tsuji H (2010) Poly(lactic acid): synthesis, structures, properties, processing, and applications. Wiley, USA
Slomkowski S, Penczek S, Duda A (2014) Polylactides an overview. Polym Adv Technol. doi:10.1002/Pat.3281
Mehta R, Kumar V, Bhunia H, Upadhyay SN (2006) Synthesis of poly(lactic acid). J Macromol Sci Part C: Polym Rev 45:325–349
Smith R (2005) Biodegradable polymers for industrial applications. Woodhead Publishing Limited, Cambridge
Mathieu J-LT, Emilie B, Pierre H, Christophe MT (2012) Synthesis of biodegradable polymers from renewable resources. Polym Chem 3:836–851
Moon SI, Lee CW, Miyamoto M, Kimura T (2000) Melt polycondensation of l-lactic acid with Sn(II) catalysts activated by various proton acids: a direct manufacturing route to high molecular weight poly(l-lactic acid). J Polym Sci Polym Chem 38:1673
Maharana T, Mohanty B, Negi YS (2009) Melt condensation of lactic acid and its degradability. Prog Polym Sci 34:99–124
Rachel HP, Linda MH, Charlotte KW (2008) Biocompatible initiators for lactide polymerization. Polym Rev 48:11–63
Dubois P, Coulembier O, Raquez J-M (2009) Handbook of ring-opening polymerization. Wiley, USA
Stridsberg KM, Ryner M, Albertsson A-C (2002) Synthesis and characterization of star shaped poly(d,l-lactide). Adv Polym Sci 157:42–65
Inkinen H, Albertsson A-C, Sodergard A (2011) From lactic acid to poly(lactic acid) (PLA): characterization and analysis of PLA processing and applications. Biomacromolecules 12:523–532
Gupta AP, Kumar V (2007) New emerging trends in synthetic biodegradable polymers—polylactide: A. Crit Eur Polym J 43:4053–4074
Moon SI, Kimura Y (2003) Melt polycondensation of l-lactic acid to poly(l-lactic acid) with Sn(II) catalysts combined with various metal alkoxides. Polym Int 52:299–303
Sedlarik V, Kucharczyk P, Kasparkova V, Drbohlav J, Salakova A, Saka P (2010) Optimization of the reaction conditions and characterization of l-lactic acid direct polycondensation products catalyzed by a non-metal-based compound. J Appl Polym Sci 116:1597–1602
Chen GX, Kim HS, Kim ES, Yoon JS (2006) Synthesis of high-molecular-weight poly(l-lactic acid) through the direct condensation polymerization of l-lactic acid in bulk state. Eur Polym J 42:468–472
Hiltunen K, Seppala J, Haerkoenen M (2007) Effect of catalyst and polymerization conditions on the preparation of low molecular weight lactic acid polymers. Macromolecules 30:373–379
Shyamroy S, Garnaik B, Sivaram S (2005) Structure of poly(l-lactic acid)s prepared by the dehydropolycondensation of l-lactic acid with organotin catalysts. J Polym Sci Part A Polym Chem 43:2164–2177
Datta R, Henry M (2006) Lactic acid: recent advances in products, processes and technologies—a review. J Chem Technol Biotechnol 81:1119–1129
Katiyar V, Nanavati H (2011) In situ synthesis of high molecular weight poly(l-lactic acid) clay nanocomposites. Polym Eng Sci 51:2066–2077
Moore JS, Stupp SI (1990) Room temperature polyesterification. Macromolecules 23:65–70
Panayotov IM, Belcheva N, Tavetanov C (1987) Polyesters prepared by polycondensation in the presence of carbodiimides, 1. Preparation of polyesters from aliphatic dicarboxylic acids and diols under mild conditions. Macromol Chem Phys 188:2821–2830
Trollsas M, Atthoff B, Claesson H, Hedrick JL (1998) Hyperbranched poly(ε-caprolactone)s. Macromolecules 31:3439–3445
Trollsas M, Kelly MA, Claesson H, Siemens R, Hedrick JL (1999) Highly branched block copolymers: design, synthesis and morphology. Macromolecules 32:4917–4924
Fischer AM, Wolf FK, Frey H (2012) Long-chain branched poly(lactide)s based on polycondensation of AB2-type macromonomers. Macromol Chem Phys 213:1349–1358
Akutsu F, Inoki M, Uei H, Sueyoshi M, Kasahima Y, Naruchi K, Yamaguchi Y, Sunahara M (1998) Synthesis of poly(lactic acid) by direct polycondensation of lactic acid using 1,1′-carbonyldiimidazole, N,N,N′,N′-tetramethylchloroformamidinium chloride, and N,N′-dicyclohexylcarbodiimide as condensing agents. Polym J (Tokyo) 30:421–423
(2005) Final report of the safety assessment of urea. Int J Toxicol 24 Suppl 3:1–56
Hiltunen K, Harkonen M, Seppala JV, Vaananen T (1996) Synthesis and characterization of lactic acid based telechelic prepolymers. Macromolecules 29:8677–8682
Mikolajczyk M, Kielbasinski P (1981) Recent developments in the carbodiimide chemistry. Tetrahedron 37:233–284
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shyamroy, S., Garnaik, B. & Sivaram, S. High molecular weight poly(l-lactic acid)s by polyesterification using diisopropylcarbodiimide (DIPC) and 4-(dimethylamino) pyridinium-p-toluene sulfonate (DPTS). Polym. Bull. 72, 405–415 (2015). https://doi.org/10.1007/s00289-014-1285-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-014-1285-0