Abstract
Purpose
To investigate the effects of therapeutically relevant dose levels of bleomycin, etoposide and cisplatin (BEP) on testicular steroidogenic enzymes, and possible protective effects of an antioxidant cocktail (AC).
Methods
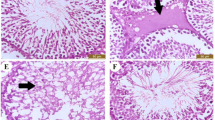
Adult Sprague–Dawley rats received BEP with or without the AC (α-tocopherol, l-ascorbic acid, selenium and zinc) for either (a) 4 days (short term; 1.5, 15 and 3 mg/kg), or (b) three cycles of 21 days each (0.75, 7.5 and 1.5 mg/kg), or (c) the three cycles with a 63-day recovery period. The expression of steroidogenic enzymes were measured in the testes by Western blotting and immunofluorescent labeling.
Results
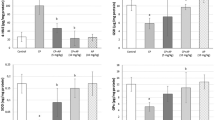
The short-term BEP exposure resulted in a decrease in scavenger receptor class-B1 and an increase in luteinizing hormone receptor (LHR). The AC with or without BEP has increased the levels of LHR, 3β-hydroxysteroid dehydrogenase (3β-HSD) and 17β-HSD, but without significant changes in testosterone levels. The three cycles of BEP up-regulated the expression of steroidogenic acute regulatory protein (StAR) and down-regulated that of cholesterol side chain cleavage enzyme (P450scc), cytochrome p450 17A1 (Cyp17A1, recovered by the AC) and 17β-HSD, associated with significant reduction in testosterone levels. The three cycles with the recovery time led to decreases in LHR, StAR, P450scc and Cyp17A1 and increases in 3β-HSD and 17β-HSD. The AC did not enhance the recovery of the enzyme levels.
Conclusion
The three cycles of BEP treatment inhibit the testosterone synthesis pathway even after the recovery time. The AC recovers the effects of BEP chemotherapy on a few steroidogenic enzymes.








Similar content being viewed by others
Abbreviations
- 17β-HSD:
-
17-Beta-hydroxysteroid dehydrogenase
- 3β-HSD:
-
3-Beta-hydroxysteroid dehydrogenase
- Cyp17A1:
-
Cytochrome p450 17A1
- Cyp19A1:
-
Cytochrome P450 19A1
- LHR:
-
Luteinizing hormone receptor
- P450scc:
-
Cholesterol side chain cleavage enzyme
- SRB1:
-
Scavenger receptor type B class 1
- StAR:
-
Steroidogenic acute regulatory protein
References
Tsang RY, Al-Fayea T, Au HJ (2009) Cisplatin overdose: toxicities and management. Drug Saf 32(12):1109–1122. doi:10.2165/11316640-000000000-00000
Altun Z, Olgun Y, Ercetin P, Aktas S, Kirkim G, Serbetcioglu B, Olgun N, Guneri EA (2014) Protective effect of acetyl-l-carnitine against cisplatin ototoxicity: role of apoptosis-related genes and pro-inflammatory cytokines. Cell Prolif 47(1):72–80. doi:10.1111/cpr.12080
Kilarkaje N, Mousa AM, Al-Bader MM, Khan KM (2013) Antioxidants enhance the recovery of three cycles of bleomycin, etoposide, and cisplatin-induced testicular dysfunction, pituitary-testicular axis, and fertility in rats. Fertil Steril 100(4):1151–1159. doi:10.1016/j.fertnstert.2013.06.019
Narayana K, Al-Bader M, Mousa A, Khan KM (2012) Molecular effects of chemotherapeutic drugs and their modulation by antioxidants in the testis. Eur J Pharmacol 674(2–3):207–216. doi:10.1016/j.ejphar.2011.11.025
Howell SJ, Shalet SM (2001) Testicular function following chemotherapy. Hum Reprod Update 7(4):363–369
Poorman-Allen P, Backer LC, Adler ID, Westbrook-Collins B, Moses MJ, Allen JW (1990) Bleomycin effects on mouse meiotic chromosomes. Mutagenesis 5(6):573–581
Howell SJ, Shalet SM (2002) Fertility preservation and management of gonadal failure associated with lymphoma therapy. Curr Oncol Rep 4(5):443–452
Marcon L, Hales BF, Robaire B (2008) Reversibility of the effects of subchronic exposure to the cancer chemotherapeutics bleomycin, etoposide, and cisplatin on spermatogenesis, fertility, and progeny outcome in the male rat. J Androl 29(4):408–417. doi:10.2164/jandrol.107.004218
Narayana K, Verghese S, Jacob SS (2009) l-ascorbic acid partially protects two cycles of cisplatin chemotherapy-induced testis damage and oligo-astheno-teratospermia in a mouse model. Exp Toxicol Pathol 61(6):553–563. doi:10.1016/j.etp.2008.12.001
Travis LB, Beard C, Allan JM, Dahl AA, Feldman DR, Oldenburg J, Daugaard G, Kelly JL, Dolan ME, Hannigan R, Constine LS, Oeffinger KC, Okunieff P, Armstrong G, Wiljer D, Miller RC, Gietema JA, van Leeuwen FE, Williams JP, Nichols CR, Einhorn LH, Fossa SD (2010) Testicular cancer survivorship: research strategies and recommendations. J Natl Cancer Inst 102(15):1114–1130. doi:10.1093/jnci/djq216
Bray F, Richiardi L, Ekbom A, Pukkala E, Cuninkova M, Moller H (2006) Trends in testicular cancer incidence and mortality in 22 European countries: continuing increases in incidence and declines in mortality. Int J Cancer 118(12):3099–3111. doi:10.1002/ijc.21747
Fung C, Vaughn DJ (2011) Complications associated with chemotherapy in testicular cancer management. Nat Rev Urol 8(4):213–222. doi:10.1038/nrurol.2011.26
Azouri H, Bidart JM, Bohuon C (1989) In vivo toxicity of cisplatin and carboplatin on the leydig cell function and effect of the human choriogonadotropin. Biochem Pharmacol 38(4):567–571. doi:10.1016/0006-2952(89)90200-1
Maines MD, Sluss PM, Iscan M (1990) Cis-platinum-mediated decrease in serum testosterone is associated with depression of luteinizing hormone receptors and cytochrome P-450scc in rat testis. Endocrinology 126(5):2398–2406. doi:10.1210/endo-126-5-2398
Rezvanfar MA, Shahverdi AR, Ahmadi A, Baeeri M, Mohammadirad A, Abdollahi M (2013) Protection of cisplatin-induced spermatotoxicity, DNA damage and chromatin abnormality by selenium nano-particles. Toxicol Appl Pharmacol 266(3):356–365. doi:10.1016/j.taap.2012.11.025
Tomita E, Kondo T, Nakazawa H, Ito F, Hashimoto Y, Tanabe K (2007) Successful testis preservation for bilateral testicular tumors with a new chemotherapy-based protocol: initial results of three cases. Int J Urol 14(9):879–882. doi:10.1111/j.1442-2042.2007.01842.x
Puhse G, Secker A, Kemper S, Hertle L, Kliesch S (2011) Testosterone deficiency in testicular germ-cell cancer patients is not influenced by oncological treatment. Int J Androl 34(5 Pt 2):e351–e357. doi:10.1111/j.1365-2605.2010.01123.x
Howell SJ, Shalet SM (2005) Spermatogenesis after cancer treatment: damage and recovery. J Natl Cancer Inst Monogr 34:12–17. doi:10.1093/jncimonographs/lgi003
Brydoy M, Fossa SD, Klepp O, Bremnes RM, Wist EA, Wentzel-Larsen T, Dahl O (2010) Paternity and testicular function among testicular cancer survivors treated with two to four cycles of cisplatin-based chemotherapy. Eur Urol 58(1):134–140. doi:10.1016/j.eururo.2010.03.041
Brennemann W, Stoffel-Wagner B, Helmers A, Mezger J, Jager N, Klingmuller D (1997) Gonadal function of patients treated with cisplatin based chemotherapy for germ cell cancer. J Urol 158(3 Pt 1):844–850. doi:10.1016/S0022-5347(01)64333-7
Palmieri G, Lotrecchiano G, Ricci G, Spiezia R, Lombardi G, Bianco AR, Torino G (1996) Gonadal function after multimodality treatment in men with testicular germ cell cancer. Eur J Endocrinol 134(4):431–436
Howell SJ, Radford JA, Adams JE, Shalet SM (2000) The impact of mild leydig cell dysfunction following cytotoxic chemotherapy on bone mineral density (BMD) and body composition. Clin Endocrinol (Oxf) 52(5):609–616. doi:10.1046/j.1365-2265.2000.00997.x
Bieber AM, Marcon L, Hales BF, Robaire B (2006) Effects of chemotherapeutic agents for testicular cancer on the male rat reproductive system, spermatozoa, and fertility. J Androl 27(2):189–200. doi:10.2164/jandrol.05103
Delbes G, Chan D, Pakarinen P, Trasler JM, Hales BF, Robaire B (2009) Impact of the chemotherapy cocktail used to treat testicular cancer on the gene expression profile of germ cells from male brown-norway rats. Biol Reprod 80(2):320–327. doi:10.1095/biolreprod.108.072108
Masubuchi Y, Kawasaki M, Horie T (2006) Down-regulation of hepatic cytochrome P450 enzymes associated with cisplatin-induced acute renal failure in male rats. Arch Toxicol 80(6):347–353. doi:10.1007/s00204-006-0079-z
Howell SJ, Radford JA, Adams JE, Smets EM, Warburton R, Shalet SM (2001) Randomized placebo-controlled trial of testosterone replacement in men with mild leydig cell insufficiency following cytotoxic chemotherapy. Clin Endocrinol (Oxf) 55(3):315–324. doi:10.1046/j.1365-2265.2001.01297.x
Payne AH, Youngblood GL (1995) Regulation of expression of steroidogenic enzymes in leydig cells. Biol Reprod 52(2):217–225
Svechnikov K, Izzo G, Landreh L, Weisser J, Soder O (2010) Endocrine disruptors and Leydig cell function. J Biomed Biotechnol. doi:10.1155/2010/684504
Delbes G, Hales BF, Robaire B (2007) Effects of the chemotherapy cocktail used to treat testicular cancer on sperm chromatin integrity. J Androl 28 (2):241–249; discussion 250–241. doi:10.2164/jandrol.106.001487
Boekelheide K (2005) Mechanisms of toxic damage to spermatogenesis. J Natl Cancer Inst Monogr 34:6–8. doi:10.1093/jncimonographs/lgi006
Jahnukainen K, Ehmcke J, Hou M, Schlatt S (2011) Testicular function and fertility preservation in male cancer patients. Best Pract Res Clin Endocrinol Metab 25(2):287–302. doi:10.1016/j.beem.2010.09.007
Valacchi G, Sticozzi C, Lim Y, Pecorelli A (2011) Scavenger receptor class B type I: a multifunctional receptor. Ann N Y Acad Sci 1229:E1–E7. doi:10.1111/j.1749-6632.2011.06205.x
Reaven E, Zhan L, Nomoto A, Leers-Sucheta S, Azhar S (2000) Expression and microvillar localization of scavenger receptor class B, type I (SR-BI) and selective cholesteryl ester uptake in Leydig cells from rat testis. J Lipid Res 41(3):343–356
Light A, Hammes SR (2013) Membrane receptor cross talk in steroidogenesis: recent insights and clinical implications. Steroids 78(6):633–638. doi:10.1016/j.steroids.2012.12.016
Manna PR, Stocco DM (2005) Regulation of the steroidogenic acute regulatory protein expression: functional and physiological consequences. Curr Drug Targets Immune Endocr Metabol Disord 5(1):93–108
Bogan RL, Davis TL, Niswender GD (2007) Peripheral-type benzodiazepine receptor (PBR) aggregation and absence of steroidogenic acute regulatory protein (StAR)/PBR association in the mitochondrial membrane as determined by bioluminescence resonance energy transfer (BRET). J Steroid Biochem Mol Biol 104(1–2):61–67. doi:10.1016/j.jsbmb.2006.10.007
Garcia MM, Acquier A, Suarez G, Gomez NV, Gorostizaga A, Mendez CF, Paz C (2012) Cisplatin inhibits testosterone synthesis by a mechanism that includes the action of reactive oxygen species (ROS) at the level of P450scc. Chem Biol Interact 199(3):185–191. doi:10.1016/j.cbi.2012.08.012
Conley AJ, Bird IM (1997) The role of cytochrome P450 17 alpha-hydroxylase and 3 beta-hydroxysteroid dehydrogenase in the integration of gonadal and adrenal steroidogenesis via the delta 5 and delta 4 pathways of steroidogenesis in mammals. Biol Reprod 56(4):789–799
Faienza MF, Giordani L, Delvecchio M, Cavallo L (2008) Clinical, endocrine, and molecular findings in 17 beta-hydroxysteroid dehydrogenase type 3 deficiency. J Endocrinol Invest 31(1):85–91. doi:10.1016/0960-0750(96)00035-0
Miller WL (2012) The syndrome of 17,20 lyase deficiency. J Clin Endocrinol Metab 97(1):59–67. doi:10.1210/jc.2011-2161
Sergejew TF, Hartmann RW (1996) Effect of a diphenylethylenediamine platinum complex on steroidogenesis in rats. J Steroid Biochem Mol Biol 58(2):243–248. doi:10.1016/0960-0760(96)00035-0
Randall VA (1994) Role of 5 alpha-reductase in health and disease. Baillieres Clin Endocrinol Metab 8(2):405–431
Meinhardt U, Mullis PE (2002) The essential role of the aromatase/p450arom. Semin Reprod Med 20(3):277–284. doi:10.1055/s-2002-35374
Narayana K (2012) Effects of l-ascorbic acid on two cycles of cisplatin-induced DNA double-strand breaks and phosphorylation of p53 in the liver. Exp Toxicol Pathol 64(5):495–502. doi:10.1016/j.etp.2010.11.004
Salem EA, Salem NA, Maarouf AM, Serefoglu EC, Hellstrom WJ (2012) Selenium and lycopene attenuate cisplatin-induced testicular toxicity associated with oxidative stress in wistar rats. Urology 79(5):1184-e1–1186-e1. doi:10.1016/j.urology.2011.12.006
Beytur A, Ciftci O, Oguz F, Oguzturk H, Yilmaz F (2012) Montelukast attenuates side effects of cisplatin including testicular, spermatological, and hormonal damage in male rats. Cancer Chemother Pharmacol 69(1):207–213. doi:10.1007/s00280-011-1692-y
Ilbey YO, Ozbek E, Cekmen M, Simsek A, Otunctemur A, Somay A (2009) Protective effect of curcumin in cisplatin-induced oxidative injury in rat testis: mitogen-activated protein kinase and nuclear factor-kappa B signaling pathways. Hum Reprod 24(7):1717–1725. doi:10.1093/humrep/dep058dep058
Ilbey YO, Ozbek E, Simsek A, Otunctemur A, Cekmen M, Somay A (2009) Potential chemoprotective effect of melatonin in cyclophosphamide- and cisplatin-induced testicular damage in rats. Fertil Steril 92(3):1124–1132. doi:10.1016/j.fertnstert.2008.07.1758
Acknowledgments
This research work was funded by Kuwait University Grant Number MA02/08 and General Facility grant number SRUL02/13. The author is grateful to Ms. S. Verghese, Ms. J. Prashanth and Dr. S. Jacob for the technical assistance, and the Animal Resources Center for maintaining the experimental animals.
Conflict of interest
The author declares no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kilarkaje, N. Effects of combined treatment of α-tocopherol, l-ascorbic acid, selenium and zinc on bleomycin, etoposide and cisplatin-induced alterations in testosterone synthesis pathway in rats. Cancer Chemother Pharmacol 74, 1175–1189 (2014). https://doi.org/10.1007/s00280-014-2592-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-014-2592-8