Abstract
Purpose
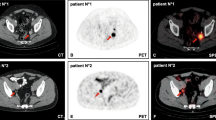
Intraoperative identification of lymph node (LN) metastases (LNM) detected on preoperative PSMA PET/CT may be facilitated by PSMA radioguided surgery with the use of a gamma probe. We evaluated the uptake of 111In-labelled PSMA ligand DKFZ-617 (referred to as 111In-PSMA-617) in unaffected LN and LNM at the level of single LN.
Methods
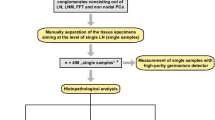
Six patients with prostate cancer (PCa) with suspicion of LNM on preoperative PSMA PET/CT underwent 111In-PSMA-617-guided lymphadenectomy (LA; four salvage LA and two primary LA). 111In-PSMA-617 (109 ± 5 MBq). was injected Intravenously 48 h prior to surgery Template LAs were performed in small subregions: common, external, obturator and internal iliac vessels, and presacral and retroperitoneal subregions (n = 4). Samples from each subregion were isolated aiming at the level of single LN. Uptake was measured ex situ using a germanium detector. Receiver operating characteristic (ROC) analysis was performed based on 111In-PSMA-617 uptake expressed as standardized uptake values normalized to lean body mass (SUL).
Results
Overall 310 LN (mean 52 ± 19.7) were removed from 74 subregions (mean 12 ± 3.7). Of the 310 LN, 35 turned out to be LNM on histopathology. Separation of the samples from all subregions resulted in 318 single specimens: 182 PCa-negative LN samples with 275 LN, 35 single LNM samples, 3 non-nodal PCa tissue samples and 98 fibrofatty tissue samples. The median SULs of nonaffected LN (0.16) and affected LN (13.2) were significantly different (p < 0.0001). Based on 38 tumour-containing and 182 tumour-free specimens, ROC analysis revealed an area under the curve of 0.976 (95% CI 0.95–1.00, p < 0.0001). Using a SUL cut-off value of 1.136, sensitivity, specificity, positive predictive value, negative predictive value and accuracy in discriminating affected from nonaffected LN were 92.1% (35/38), 98.9% (180/182), 94.6% (35/37), 98.4% (180/183) and 97.7% (215/220), respectively.
Conclusion
Ex situ analysis at the level of single LN showed that 111In-PSMA-617 had excellent ability to discriminate between affected and nonaffected LN in our patients with PCa. This tracer characteristic is a prerequisite for in vivo real-time measurements during surgery.





Similar content being viewed by others
References
American Cancer Society. Cancer facts & figures 2018. Atlanta, GA: American Cancer Society; 2018.
Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft DK, AWMF). Interdisziplinäre Leitlinie der Qualität S3 zur Früherkennung, Diagnose und Therapie der verschiedenen Stadien des Prostatakarzinoms, Langversion 5.0,. AWMF Registernummer: 043/022OL, http://www.leitlinienprogramm-onkolo-giede/leitlinien/prostatakarzinom/ (abgerufen am: 10052018). 2018.
Cornford P, Bellmunt J, Bolla M, Briers E, De Santis M, Gross T, et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part II: Treatment of relapsing, metastatic, and castration-resistant prostate cancer. Eur Urol. 2017;71(4):630–42.
Mottet N, Bellmunt J, Bolla M, Briers E, Cumberbatch MG, De Santis M, et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: Screening, diagnosis, and local treatment with curative intent. Eur Urol. 2017;71(4):618–29.
Abdollah F, Gandaglia G, Suardi N, Capitanio U, Salonia A, Nini A, et al. More extensive pelvic lymph node dissection improves survival in patients with node-positive prostate cancer. Eur Urol. 2015;67(2):212–9.
Jilg CA, Drendel V, Rischke HC, Beck T, Vach W, Schaal K, et al. Diagnostic accuracy of Ga-68-HBED-CC-PSMA-ligand-PET/CT before salvage lymph node dissection for recurrent prostate cancer. Theranostics. 2017;7(6):1770–80.
Herlemann A, Wenter V, Kretschmer A, Thierfelder KM, Bartenstein P, Faber C, et al. (68)Ga-PSMA positron emission tomography/computed tomography provides accurate staging of lymph node regions prior to lymph node dissection in patients with prostate cancer. Eur Urol. 2016;70(4):553–7.
Afshar-Oromieh A, Zechmann CM, Malcher A, Eder M, Eisenhut M, Linhart HG, et al. Comparison of PET imaging with a (68)Ga-labelled PSMA ligand and (18)F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2014;41(1):11–20.
Eiber M, Maurer T, Souvatzoglou M, Beer AJ, Ruffani A, Haller B, et al. Evaluation of hybrid 68Ga-PSMA ligand PET/CT in 248 patients with biochemical recurrence after radical prostatectomy. J Nucl Med. 2015;56(5):668–74.
Maurer T, Gschwend JE, Rauscher I, Souvatzoglou M, Haller B, Weirich G, et al. Diagnostic efficacy of (68)gallium-PSMA positron emission tomography compared to conventional imaging for lymph node staging of 130 consecutive patients with intermediate to high risk prostate cancer. J Urol. 2016;195(5):1436–43.
Abdollah F, Briganti A, Montorsi F, Stenzl A, Stief C, Tombal B, et al. Contemporary role of salvage lymphadenectomy in patients with recurrence following radical prostatectomy. Eur Urol. 2015;67(5):839–49.
Zschaeck S, Wust P, Beck M, Wlodarczyk W, Kaul D, Rogasch J, et al. Intermediate-term outcome after PSMA-PET guided high-dose radiotherapy of recurrent high-risk prostate cancer patients. Radiat Oncol. 2017;12(1):140.
Maurer T, Robu S, Schottelius M, Schwamborn K, Rauscher I, van den Berg NS, et al. (99m)Technetium-based prostate-specific membrane antigen-radioguided surgery in recurrent prostate cancer. Eur Urol. 2018. https://doi.org/10.1016/j.eururo.2018.03.013.
Maurer T, Weirich G, Schottelius M, Weineisen M, Frisch B, Okur A, et al. Prostate-specific membrane antigen-radioguided surgery for metastatic lymph nodes in prostate cancer. Eur Urol. 2015;68(3):530–4.
Rauscher I, Duwel C, Wirtz M, Schottelius M, Wester HJ, Schwamborn K, et al. Value of 111In-prostate-specific membrane antigen (PSMA)-radioguided surgery for salvage lymphadenectomy in recurrent prostate cancer: correlation with histopathology and clinical follow-up. BJU Int. 2017;120(1):40–7.
Gourni E, Canovas C, Goncalves V, Denat F, Meyer PT, Maecke HR. (R)-NODAGA-PSMA: a versatile precursor for radiometal labeling and nuclear imaging of PSMA-positive tumors. PLoS One. 2015;10(12):e0145755.
Unterweger MP, Hoppes DD, Schima FJ. New and revised half-life measurements results. Nuclear instruments and methods in physics research section a: accelerators, spectrometers, detectors and associated equipment. Nucl Instrum Methods Phys Res A. 1992;312(1-2):349–52.
Janmahasatian S, Duffull SB, Ash S, Ward LC, Byrne NM, Green B. Quantification of lean bodyweight. Clin Pharmacokinet. 2005;44(10):1051–65.
D'Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280(11):969–74.
Rauscher I, Horn T, Eiber M, Gschwend JE, Maurer T. Novel technology of molecular radio-guidance for lymph node dissection in recurrent prostate cancer by PSMA-ligands. World J Urol. 2018;36(4):603–8.
Hillier SM, Maresca KP, Lu G, Merkin RD, Marquis JC, Zimmerman CN, et al. 99mTc-labeled small-molecule inhibitors of prostate-specific membrane antigen for molecular imaging of prostate cancer. J Nucl Med. 2013;54(8):1369–76.
Banerjee SR, Foss CA, Horhota A, Pullambhatla M, McDonnell K, Zale S, et al. 111In- and IRDye800CW-labeled PLA-PEG nanoparticle for imaging prostate-specific membrane antigen-expressing tissues. Biomacromolecules. 2017;18(1):201–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Additional information
Michael Mix and Kathrin Reichel share authorship.
Rights and permissions
About this article
Cite this article
Mix, M., Reichel, K., Stoykow, C. et al. Performance of 111In-labelled PSMA ligand in patients with nodal metastatic prostate cancer: correlation between tracer uptake and histopathology from lymphadenectomy. Eur J Nucl Med Mol Imaging 45, 2062–2070 (2018). https://doi.org/10.1007/s00259-018-4094-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-018-4094-0