Abstract
Purpose
We investigated the prognostic value of the tumour heterogeneity index determined on preoperative [18F]FDG PET/CT in patients with uterine leiomyosarcoma (LMS).
Methods
We retrospectively reviewed patients with uterine LMS who underwent preoperative [18F]FDG PET/CT scans at three tertiary referral hospitals. The PET/CT parameters maximum standardized uptake value of the primary tumour (SUVmax), metabolic tumour volume (MTV) and total lesion glycolysis were assessed. The negative values of the MTV linear regression slope (nMLRS) according to the SUV thresholds of 2.5 and 3.0 were determined as the tumour heterogeneity index. The value of PET/CT-derived parameters in predicting progression-free survival (PFS) and overall survival (OS) were determined in regression analyses.
Results
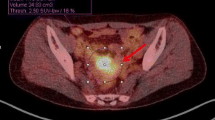
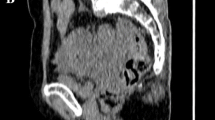
Clinicopathological and PET/CT data from 16 patients were reviewed. The median postsurgical follow-up was 21 months (range 4–82 months), and 12 patients (75.0%) experienced recurrence. Tumour size (P = 0.017), SUVmax (P = 0.019), MTV (P = 0.016) and nMLRS (P = 0.008) were significant prognostic factors for recurrence. MTV (P = 0.048) and nMLRS (P = 0.045) were significant prognostic factors for patient survival. nMLRS was correlated with clinicopathological parameters including tumour size (Pearson’s correlation coefficient γ = 0.825, P < 0.001) and lymph node metastasis (γ = 0.721, P = 0.004). Patient groups categorized according to the nMLRS cut-off value showed significant differences in PFS (P = 0.033) and OS (P = 0.044).
Conclusion
The preoperative tumour heterogeneity index obtained using the MTV linear regression slope may be a novel and useful prognostic marker in uterine LMS.




Similar content being viewed by others
References
D'Angelo E, Prat J. Uterine sarcomas: a review. Gynecol Oncol. 2010;116(1):131–9. https://doi.org/10.1016/j.ygyno.2009.09.023.
Abeler VM, Royne O, Thoresen S, Danielsen HE, Nesland JM, Kristensen GB. Uterine sarcomas in Norway. A histopathological and prognostic survey of a total population from 1970 to 2000 including 419 patients. Histopathology. 2009;54(3):355–64. https://doi.org/10.1111/j.1365-2559.2009.03231.x.
Amant F, Moerman P, Neven P, Timmerman D, Van Limbergen E, Vergote I. Endometrial cancer. Lancet. 2005;366(9484):491–505. https://doi.org/10.1016/S0140-6736(05)67063-8.
Brooks SE, Zhan M, Cote T, Baquet CR. Surveillance, epidemiology, and end results analysis of 2677 cases of uterine sarcoma 1989-1999. Gynecol Oncol. 2004;93(1):204–8. https://doi.org/10.1016/j.ygyno.2003.12.029.
Gockley AA, Rauh-Hain JA, del Carmen MG. Uterine leiomyosarcoma: a review article. Int J Gynecol Cancer. 2014;24(9):1538–42. https://doi.org/10.1097/IGC.0000000000000290.
Brenner W, Conrad EU, Eary JF. FDG PET imaging for grading and prediction of outcome in chondrosarcoma patients. Eur J Nucl Med Mol Imaging. 2004;31(2):189–95. https://doi.org/10.1007/s00259-003-1353-4.
Brenner W, Eary JF, Hwang W, Vernon C, Conrad EU. Risk assessment in liposarcoma patients based on FDG PET imaging. Eur J Nucl Med Mol Imaging. 2006;33(11):1290–5. https://doi.org/10.1007/s00259-006-0170-y.
Lisle JW, Eary JF, O'Sullivan J, Conrad EU. Risk assessment based on FDG-PET imaging in patients with synovial sarcoma. Clin Orthop Relat Res. 2009;467(6):1605–11. https://doi.org/10.1007/s11999-008-0647-z.
Yamane T, Takaoka A, Kita M, Imai Y, Senda M. 18F-FLT PET performs better than 18F-FDG PET in differentiating malignant uterine corpus tumors from benign leiomyoma. Ann Nucl Med. 2012;26(6):478–84. https://doi.org/10.1007/s12149-012-0597-0.
Park JY, Lee JW, Lee HJ, Lee JJ, Moon SH, Kang SY, et al. Prognostic significance of preoperative 18F-FDG PET/CT in uterine leiomyosarcoma. J Gynecol Oncol. 2017;28(3):e28. https://doi.org/10.3802/jgo.2017.28.e28.
Nowell PC. The clonal evolution of tumor cell populations. Science. 1976;194(4260):23–8.
Heppner GH. Tumor heterogeneity. Cancer Res. 1984;44(6):2259–65.
Tixier F, Le Rest CC, Hatt M, Albarghach N, Pradier O, Metges JP, et al. Intratumor heterogeneity characterized by textural features on baseline 18F-FDG PET images predicts response to concomitant radiochemotherapy in esophageal cancer. J Nucl Med. 2011;52(3):369–78. https://doi.org/10.2967/jnumed.110.082404.
Gerlinger M, Rowan AJ, Horswell S, Math M, Larkin J, Endesfelder D, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. 2012;366(10):883–92. https://doi.org/10.1056/NEJMoa1113205.
Asselin MC, O'Connor JP, Boellaard R, Thacker NA, Jackson A. Quantifying heterogeneity in human tumours using MRI and PET. Eur J Cancer. 2012;48(4):447–55. https://doi.org/10.1016/j.ejca.2011.12.025.
Lee M, Lee H, Cheon GJ, Kim HS, Chung HH, Kim JW, et al. Prognostic value of preoperative intratumoral FDG uptake heterogeneity in patients with epithelial ovarian cancer. Eur Radiol. 2017;27(1):16–23. https://doi.org/10.1007/s00330-016-4368-5.
Kang SY, Cheon GJ, Lee M, Kim HS, Kim JW, Park NH, et al. Prediction of recurrence by preoperative intratumoral FDG uptake heterogeneity in endometrioid endometrial cancer. Transl Oncol. 2017;10(2):178–83. https://doi.org/10.1016/j.tranon.2017.01.002.
Chung HH, Kang SY, Ha S, Kim JW, Park NH, Song YS, et al. Prognostic value of preoperative intratumoral FDG uptake heterogeneity in early stage uterine cervical cancer. J Gynecol Oncol. 2016;27(2):e15. https://doi.org/10.3802/jgo.2016.27.e15.
Kim YI, Kim YJ, Paeng JC, Cheon GJ, Lee DS, Chung JK, et al. Heterogeneity index evaluated by slope of linear regression on 18F-FDG PET/CT as a prognostic marker for predicting tumor recurrence in pancreatic ductal adenocarcinoma. Eur J Nucl Med Mol Imaging. 2017;44(12):1995–2003. https://doi.org/10.1007/s00259-017-3755-8.
Huang B, Chan T, Kwong DL, Chan WK, Khong PL. Nasopharyngeal carcinoma: investigation of intratumoral heterogeneity with FDG PET/CT. AJR Am J Roentgenol. 2012;199(1):169–74. https://doi.org/10.2214/AJR.11.7336.
Kim TH, Yoon JK, Kang DK, Lee SJ, Jung YS, Yim H, et al. Correlation between F-18 fluorodeoxyglucose positron emission tomography metabolic parameters and dynamic contrast-enhanced MRI-derived perfusion data in patients with invasive ductal breast carcinoma. Ann Surg Oncol. 2015;22(12):3866–72. https://doi.org/10.1245/s10434-015-4526-z.
Kwon SH, Yoon JK, An YS, Shin YS, Kim CH, Lee DH, et al. Prognostic significance of the intratumoral heterogeneity of 18F-FDG uptake in oral cavity cancer. J Surg Oncol. 2014;110(6):702–6. https://doi.org/10.1002/jso.23703.
Prat J. FIGO staging for uterine sarcomas. Int J Gynaecol Obstet. 2009;104(3):177–8. https://doi.org/10.1016/j.ijgo.2008.12.008.
Altman DG, Gore SM, Gardner MJ, Pocock SJ. Statistical guidelines for contributors to medical journals. Br Med J. 1983;286(6376):1489–93.
Lucignani G. SUV and segmentation: pressing challenges in tumour assessment and treatment. Eur J Nucl Med Mol Imaging. 2009;36(4):715–20. https://doi.org/10.1007/s00259-009-1085-1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. For this type of retrospective study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Lee, JW., Park, JY., Lee, H.J. et al. Preoperative [18F]FDG PET/CT tumour heterogeneity index in patients with uterine leiomyosarcoma: a multicentre retrospective study. Eur J Nucl Med Mol Imaging 45, 1309–1316 (2018). https://doi.org/10.1007/s00259-018-3975-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-018-3975-6