Abstract
Purpose
Early side effects including oesophagitis are potential prognostic factors in patients undergoing radiochemotherapy (RCT) for locally advanced oesophageal cancer (LAEC). We assessed the prognostic value of 18F-fluorodeoxyglucose (FDG) uptake within irradiated non-tumour-affected oesophagus (NTO) during restaging positron emission tomography (PET) as a surrogate for inflammation/oesophagitis.
Methods
This retrospective evaluation included 64 patients with LAEC who had completed neoadjuvant RCT and had successful oncological resection. All patients underwent FDG PET/CT before and after RCT. In the restaging PET scan maximum and mean standardized uptake values (SUVmax, SUVmean) were determined in the tumour and NTO. Univariate Cox regression with respect to overall survival, local control, distant metastases and treatment failure was performed. Independence of clinically relevant parameters was tested in a multivariate Cox regression analysis.
Results
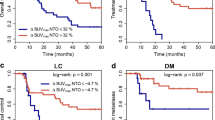
Increased FDG uptake, measured in terms of SUVmean in NTO during restaging was significantly associated with complete pathological remission (p = 0.002) and did not show a high correlation with FDG response of the tumour (rho < 0.3). In the univariate analysis, increased SUVmax and SUVmean in NTO was associated with improved overall survival (p = 0.011, p = 0.004), better local control (p = 0.051, p = 0.044), a lower rate of treatment failure (p < 0.001 for both) and development of distant metastases (p = 0.012, p = 0.001). In the multivariate analysis, SUVmax and SUVmean in NTO remained a significant prognostic factor for treatment failure (p < 0.001, p = 0.004) and distant metastases (p = 0.040, p = 0.011).
Conclusions
FDG uptake in irradiated normal tissues measured on restaging PET has significant prognostic value in patients undergoing neoadjuvant RCT for LAEC. This effect may potentially be of use in treatment personalization.




Similar content being viewed by others
References
van Hagen P, Hulshof MC, van Lanschot JJ, Steyerberg EW, van Berge Henegouwen MI, Wijnhoven BP, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366:2074–84.
Kranzfelder M, Schuster T, Geinitz H, Friess H, Büchler P. Meta-analysis of neoadjuvant treatment modalities and definitive non-surgical therapy for oesophageal squamous cell cancer. Br J Surg. 2011;98:768–83.
Gurusamy KS, Pallari E, Midya S, Mughal M. Laparoscopic versus open transhiatal oesophagectomy for oesophageal cancer. Cochrane Database Syst Rev. 2016;3:CD011390.
Rebollo Aguirre AC, Ramos-Font C, Villegas Portero R, Cook GJR, Llamas Elvira JM, Tabares AR. 18F-fluorodeoxiglucose positron emission tomography for the evaluation of neoadjuvant therapy response in esophageal cancer: systematic review of the literature. Ann Surg. 2009;250:247–54.
Swisher SG, Erasmus J, Maish M, Correa AM, Macapinlac H, Ajani JA, et al. 2-Fluoro-2-deoxy-D-glucose positron emission tomography imaging is predictive of pathologic response and survival after preoperative chemoradiation in patients with esophageal carcinoma. Cancer. 2004;101:1776–85.
Jayachandran P, Pai RK, Quon A, Graves E, Krakow TE, La T, et al. Postchemoradiotherapy positron emission tomography predicts pathologic response and survival in patients with esophageal cancer. Int J Radiat Oncol Biol Phys. 2012;84:471–7.
Elimova E, Wang X, Etchebehere E, Shiozaki H, Shimodaira Y, Wadhwa R, et al. 18-fluorodeoxy-glucose positron emission computed tomography as predictive of response after chemoradiation in oesophageal cancer patients. Eur J Cancer 1990. 2015;51:2545–52.
Malik V, Lucey JA, Duffy GJ, Wilson L, McNamara L, Keogan M, et al. Early repeated 18F-FDG PET scans during neoadjuvant chemoradiation fail to predict histopathologic response or survival benefit in adenocarcinoma of the esophagus. J Nucl Med. 2010;51:1863–9.
Palie O, Michel P, Ménard J-F, Rousseau C, Rio E, Bridji B, et al. The predictive value of treatment response using FDG PET performed on day 21 of chemoradiotherapy in patients with oesophageal squamous cell carcinoma. A prospective, multicentre study (RTEP3). Eur J Nucl Med Mol Imaging. 2013;40:1345–55.
Lordick F, Ott K, Krause B-J, Weber WA, Becker K, Stein HJ, et al. PET to assess early metabolic response and to guide treatment of adenocarcinoma of the oesophagogastric junction: the MUNICON phase II trial. Lancet Oncol. 2007;8:797–805.
Lloyd S, Chang BW. Current strategies in chemoradiation for esophageal cancer. J Gastrointest Oncol. 2014;5:156–65.
Wolff HA, Daldrup B, Jung K, Overbeck T, Hennies S, Matthias C, et al. High-grade acute organ toxicity as positive prognostic factor in adjuvant radiation and chemotherapy for locally advanced head and neck cancer. Radiology. 2011;258:864–71.
Wolff HA, Raus I, Jung K, Schüler P, Herrmann MK, Hennies S, et al. High-grade acute organ toxicity as a positive prognostic factor in primary radiochemotherapy for anal carcinoma. Int J Radiat Oncol Biol Phys. 2011;79:1467–78.
Hennies S, Hermann RM, Gaedcke J, Grade M, Hess CF, Christiansen H, et al. Increasing toxicity during neoadjuvant radiochemotherapy as positive prognostic factor for patients with esophageal carcinoma. Dis Esophagus. 2014;27:146–51.
Zschaeck S, Loeck S, Leger S, Haase R, Bandurska-Luque A, et al. FDG uptake in normal tissues assessed by PET during treatment has prognostic value for treatment results in head and neck squamous cell carcinomas undergoing radiochemotherapy. Radiother Oncol. 2017;122(3):437–44.
Mandard AM, Dalibard F, Mandard JC, Marnay J, Henry-Amar M, Petiot JF, et al. Pathologic assessment of tumor regression after preoperative chemoradiotherapy of esophageal carcinoma. Clinicopathologic correlations. Cancer. 1994;73:2680–6.
Becker K, Mueller JD, Schulmacher C, Ott K, Fink U, Busch R, et al. Histomorphology and grading of regression in gastric carcinoma treated with neoadjuvant chemotherapy. Cancer. 2003;98:1521–30.
Thies S, Langer R. Tumor regression grading of gastrointestinal carcinomas after neoadjuvant treatment. Front Oncol. 2013;3:262.
van den Hoff J, Lougovski A, Schramm G, Maus J, Oehme L, Petr J, et al. Correction of scan time dependence of standard uptake values in oncological PET. EJNMMI Res. 2014;4:18.
R Core Team. R: A Language and Environment for Statistical Computing. The R Foundation.
Duan X-F, Tang P, Yu Z-T. Neoadjuvant chemoradiotherapy for resectable esophageal cancer: an in-depth study of randomized controlled trials and literature review. Cancer Biol Med. 2014;11:191–201.
Buscombe J. PET imaging of inflammation. Q J Nucl Med Mol Imaging. 2014;58:284–9.
Hess S, Hansson SH, Pedersen KT, Basu S, Høilund-Carlsen PF. FDG-PET/CT in infectious and inflammatory diseases. PET Clin. 2014;9:497–519.
Niedzielski JS, Yang J, Liao Z, Gomez DR, Stingo F, Mohan R, et al. (18)F-Fluorodeoxyglucose positron emission tomography can quantify and predict esophageal injury during radiation therapy. Int J Radiat Oncol Biol Phys. 2016;96:670–8.
Schuell B, Gruenberger T, Kornek GV, Dworan N, Depisch D, Lang F, et al. Side effects during chemotherapy predict tumour response in advanced colorectal cancer. Br J Cancer. 2005;93:744–8.
Soveri LM, Hermunen K, de Gramont A, Poussa T, Quinaux E, Bono P, et al. Association of adverse events and survival in colorectal cancer patients treated with adjuvant 5-fluorouracil and leucovorin: is efficacy an impact of toxicity? Eur J Cancer. 2014;50:2966–74.
Dahl O, Horn A, Mella O. Do acute side-effects during radiotherapy predict tumour response in rectal carcinoma? Acta Oncol. 1994;33:409–13.
Wolff HA, Gaedcke J, Jung K, Hermann RM, Rothe H, Schirmer M, et al. High-grade acute organ toxicity during preoperative radiochemotherapy as positive predictor for complete histopathologic tumor regression in multimodal treatment of locally advanced rectal cancer. Strahlenther Onkol. 2010;186:30–5.
Budach W, Hartford A, Gioioso D, Freeman J, Taghian A, Suit HD. Tumors arising in SCID mice share enhanced radiation sensitivity of SCID normal tissues. Cancer Res. 1992;52:6292–6.
Barber JB, Burrill W, Spreadborough AR, Levine E, Warren C, Kiltie AE, et al. Relationship between in vitro chromosomal radiosensitivity of peripheral blood lymphocytes and the expression of normal tissue damage following radiotherapy for breast cancer. Radiother Oncol. 2000;55:179–86.
Borgmann K, Hoeller U, Nowack S, Bernhard M, Röper B, Brackrock S, et al. Individual radiosensitivity measured with lymphocytes may predict the risk of acute reaction after radiotherapy. Int J Radiat Oncol Biol Phys. 2008;71:256–64.
Peacock J, Ashton A, Bliss J, Bush C, Eady J, Jackson C, et al. Cellular radiosensitivity and complication risk after curative radiotherapy. Radiother Oncol. 2000;55:173–8.
Russell NS, Grummels A, Hart AA, Smolders IJ, Borger J, Bartelink H, et al. Low predictive value of intrinsic fibroblast radiosensitivity for fibrosis development following radiotherapy for breast cancer. Int J Radiat Biol. 1998;73:661–70.
Klug F, Prakash H, Huber PE, Seibel T, Bender N, Halama N, et al. Low-dose irradiation programs macrophage differentiation to an iNOS+/M1 phenotype that orchestrates effective T cell immunotherapy. Cancer Cell. 2013;24:589–602.
Balermpas P, Rödel F, Rödel C, Krause M, Linge A, Lohaus F, et al. CD8+ tumour-infiltrating lymphocytes in relation to HPV status and clinical outcome in patients with head and neck cancer after postoperative chemoradiotherapy: a multicentre study of the German cancer consortium radiation oncology group (DKTK-ROG). Int J Cancer. 2016;138:171–81.
Herskovic A, Martz K, al-Sarraf M, Leichman L, Brindle J, Vaitkevicius V, et al. Combined chemotherapy and radiotherapy compared with radiotherapy alone in patients with cancer of the esophagus. N Engl J Med. 1992;326:1593–8.
Cameron DA, Massie C, Kerr G, Leonard RCF. Moderate neutropenia with adjuvant CMF confers improved survival in early breast cancer. Br J Cancer. 2003;89:1837–42.
Zhang H, Tan S, Chen W, Kligerman S, Kim G, D'Souza WD, et al. Modeling pathologic response of esophageal cancer to chemoradiation therapy using spatial-temporal 18F-FDG PET features, clinical parameters, and demographics. Int J Radiat Oncol Biol Phys. 2014;88:195–203.
Yip SS, Coroller TP, Sanford NN, Mamon H, Aerts HJ, Berbeco RI. Relationship between the temporal changes in positron-emission-tomography-imaging-based textural features and pathologic response and survival in esophageal cancer patients. Front Oncol. 2016;6:72.
Beukinga RJ, Hulshoff JB, van Dijk LV, Muijs CT, Burgerhof JG, Kats-Ugurlu G, et al. Predicting response to neoadjuvant chemoradiotherapy in esophageal cancer with textural features derived from pre-treatment 18F-FDG PET/CT imaging. J Nucl Med. 2017;58:723–9.
Tixier F, Le Rest CC, Hatt M, Albarghach N, Pradier O, Metges JP, et al. Intratumor heterogeneity characterized by textural features on baseline 18F-FDG PET images predicts response to concomitant radiochemotherapy in esophageal cancer. J Nucl Med. 2011;52:369–78.
van Rossum PSN, Fried DV, Zhang L, Hofstetter WL, van Vulpen M, Meijer GJ, et al. The incremental value of subjective and quantitative assessment of 18F-FDG PET for the prediction of pathologic complete response to preoperative chemoradiotherapy in esophageal cancer. J Nucl Med. 2016;57:691–700.
Baumann M, Krause M, Overgaard J, Debus J, Bentzen SM, Daartz J, et al. Radiation oncology in the era of precision medicine. Nat Rev Cancer. 2016;16:234–49.
Acknowledgment
Falk Tillner is acknowledged for providing screenshots of treatment planning.
Author information
Authors and Affiliations
Contributions
S.Z. provided ideas for the study; S.Z. and F.H. performed the analysis, and drafted the manuscript, designed the figures and calculated the underlying statistics, and together with S.L.. C.J., K.Z., J.K., G.B. and J.W. were responsible for treatment, histology and imaging of patients. R.B., J.S. and S.Z. collected patient data and performed the follow-up examinations. M.K., K.Z., F.H. and M.B. provided ideas, supervised the analysis and interpretation of the data, and reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Funding
The study was partly supported by the German Federal Ministry of Education and Research (BMBF contract 03ZIK42/OncoRay). The funding source had no influence on data acquisition, evaluation or writing of the manuscript.
Conflicts of interest
None.
Ethical approval
The study was approved by the Institutional Ethics Committee and all patients signed written informed consent.
Rights and permissions
About this article
Cite this article
Zschaeck, S., Hofheinz, F., Zöphel, K. et al. Increased FDG uptake on late-treatment PET in non-tumour-affected oesophagus is prognostic for pathological complete response and disease recurrence in patients undergoing neoadjuvant radiochemotherapy. Eur J Nucl Med Mol Imaging 44, 1813–1822 (2017). https://doi.org/10.1007/s00259-017-3742-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-017-3742-0