Abstract
Purpose
Causes of ARF are numerous including drugs. In 2012, spontaneous reporting showed a possible association between dronedarone and ARF. To further investigate such association, a retrospective cohort study on health-service claim databases was performed taking amiodarone as comparison.
Methods
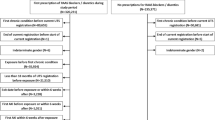
All patients receiving new prescription of amiodarone or dronedarone between September 2010 and December 2012 were selected. Cox regression models to estimate the hazard ratios (HRs), with 95 % confidence intervals (CIs), for dronedarone versus amiodarone were performed. HRs were calculated: (i) for the entire cohort; (ii) for matched cohorts using propensity score; (iii) and high-dimensional propensity score.
Results
New users without previous episodes of ARF were 56,739 and 1761 on dronedarone and 54,978 on amiodarone. After 1:1 matching for propensity score, new users with dronedarone and amiodarone were 1467 and 1467, respectively. The cumulative incidence rate of ARF was 1.6 % (95 % CI 0.7–3.6 %) among dronedarone group and 2.3 % (1.0–5.1 %) among amiodarone group (p from log rank test = 0.4884). The unadjusted HR of ARF was 0.34 (0.18–0.64) in dronedarone new users compared to amiodarone; in propensity score matched cohort, it was 0.75 (0.26–2.16), and in high-dimensional propensity score, it was 0.83 (0.25–2.73).
Conclusions
This large community-based study did not confirm the signal of an increased nephrotoxicity from dronedarone compared to amiodarone. Nevertheless, given the increasing number of reports collected from pharmacovigilance databases worldwide on this association, it is advisable for clinicians and patients to be aware of the possible kidney damage due to dronedarone in order to improve clinical outcomes with early intervention.

Similar content being viewed by others
References
Hsu C-Y, McCulloch CE, Fan D, Ordonez JD, Chertow GM, Go AS (2007) Community-based incidence of acute renal failure. Kidney Int 72(2):208–212
Naughton CA (2008) Drug-induced nephrotoxicity. Am Fam Physician 78(6):743–750
Biagi C, Venegoni M, Melis M, Buccellato E, Montanaro N, Motola D (2013) Dronedarone-associated acute renal failure: evidence coming from the Italian spontaneous ADR reporting database. Br J Clin Pharmacol 75(5):1351–1355
Schneeweiss S, Rassen JA, Glynn RJ, Avorn J, Mogun H, Brookhart MA (2011) NIH Public Access 20(4):512–522
Rassen JA, Glynn RJ, Brookhart MA, Schneeweiss S (2011) Covariate selection in high-dimensional propensity score analyses of treatment effects in small samples. Am J Epidemiol 173(12):1404–1413
Accessdata.fda.gov, “MULTAQ PRESCRIBING INFORMATION,” 2014. [Online]. Available:http://www.accessdata.fda.gov/drugsatfda_docs/label/2014/022425s025lbl.pdf
Tschuppert Y, Buclin T, Rothuizen LE et al (2007) Effect of dronedarone on renal function in healthy subjects. Br J Clin Pharmacol 64(6):785–791
Luciani R, Falcone C, Principe F, Punzo G, Mene P (2009) Acute renal failure due to amiodarone-induced hypothyroidism. Clin Nephrol 72(1):79–80
Boucher BJ (2009) Renal failure and rhabdomyolysis associated with sitagliptin and simvastatin use. But what about the amiodarone? Diabetic Med: J Br Diabet Assoc 26(2):192–193
Schmidt GA, Hoehns JD, Purcell JL, Friedman RL, Elhawi Y (2007) Severe rhabdomyolysis and acute renal failure secondary to concomitant use of simvastatin, amiodarone, and atazanavir. J Am Board Fam Med 20(4):411–416
Curran BJ, Havill JH (2002) Hepatic and renal failure associated with amiodarone infusion in a patient with hereditary fructose intolerance. Crit Care Resusc 4(2):112–115
Weber JCP (1984) Epidemiology of adverse reactions to nonsteroidal antiinflammatory drugs, vol 6. Raven Press, New York
Coons JC, Plauger KM, Seybert AL, Sokos GG (2010) Worsening heart failure in the setting of dronedarone initiation. Ann Pharmacother 44(9):1496–1500
Young C, Maruthappu M, Wayne RP, Leaver L (2013) Reversible acute kidney injury requiring haemodialysis five days after starting dronedarone in a stable 71-year-old man at risk of cardiovascular polypharmacy. J R Coll Physicians Edinb 43(2):122–125
B Healthcare (2011) Nexterone prescribing information. 1–16
Acknowledgments
We thank the Pharmaceutical Departments of Lombardy and Emilia-Romagna regions for providing the data. We would also like to thank Mariagrazia Monardo, PharmD, who actively participated in this research when preparing her graduation thesis at the Unit of Pharmacology, University of Bologna.
Funding
No founding source.
Conflict of interest
The authors declare that they have no conflict of interest.
Authors’ contributions
Study conception and planning: CB, VC, DM, MV. Acquisition, analysis, or interpretation of data: CB, VC, IF, DM, MV; MM, AV, MD. Drafting of the manuscript: VC, CB, DM, MV. Critical revision of the manuscript for important intellectual content: MM, MD, IF, AV.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. 1S
(PDF 50 kb)
Rights and permissions
About this article
Cite this article
Conti, V., Biagi, C., Melis, M. et al. Acute renal failure in patients treated with dronedarone or amiodarone: a large population-based cohort study in Italy. Eur J Clin Pharmacol 71, 1147–1153 (2015). https://doi.org/10.1007/s00228-015-1903-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-015-1903-2