Abstract
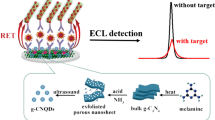
An electrochemiluminescence resonance energy transfer (ECL-RET) system that detects prostate-specific antigen (PSA) was created. Near-infrared (NIR)-emitting NaYF4:Yb,Tm/Mn upconverting nanoparticles (UCNPs) are used as donors, and gold nanorods (GNRs) are used as acceptors. The ECL was enhanced nearly threefold by Mn2+ doping, with an emission peak appearing at an NIR wavelength of 808 nm. Anti-PSA 1 (Ab1) was bound to the surfaces of UCNPs after being modified with poly(acrylic acid) (PAA). As for acceptors, cetyltrimethylammonium bromide (CTAB)-capped GNRs were treated with 11-mercaptoundecanoic acid (MUDA) and then conjugated with Anti-PSA 2 (Ab2). When PSA was added, donors and acceptors were brought in close proximity through specific interactions of antibodies and antigens, resulting in high quenching efficiency levels. Under optimal conditions, the linear range of detection was 3.75–938 pg/mL for PSA (R = 0.999), with a detection limit as low as 3.16 pg/mL. This method can be applied to detect PSA in human serums with satisfactory results.

An electrochemiluminescence resonance energy transfer system was developed for determination of prostate-specific antigen using near-infrared-emitting NaYF4:Yb,Tm/Mn upconverting nanoparticles as donors and gold nanorods as acceptors.








Similar content being viewed by others
Reference
Pansare V, Hejazi S, Faenza W, Prud’homme RK. Review of long-wavelength optical and NIR imaging materials: contrast agents, fluorophores and multifunctional nano carriers. Chem Mater. 2012;24:812–27.
Hutteman M, van der Vorst JR, Gaarenstroom KN, Peters AA, Mieog JS, Schaafsma BE, et al. Optimization of near-infrared fluorescent sentinel lymph node mapping for vulvar cancer. Am J Obstet Gynecol. 2012;206:89 e1–5.
Kim S, Lim YT, Soltesz EG, De Grand AM, Lee J, Nakayama A, et al. Near-infrared fluorescent type II quantum dots for sentinel lymph node mapping. Nat Biotechnol. 2004;22:93–7.
Frangioni J. In vivo near-infrared fluorescence imaging. Curr Opin Chem Biol. 2003;7:626–34.
Gill R, Zayats M, Willner I. Semiconductor quantum dots for bioanalysis. Angew Chem Int Ed Engl. 2008;47:7602–25.
Sapsford KE, Berti L, Medintz IL. Materials for fluorescence resonance energy transfer analysis: beyond traditional donor-acceptor combinations. Angew Chem Int Ed Engl. 2006;45:4562–89.
Qin G, Zhao S, Huang Y, Jiang J, Ye F. Magnetic bead-sensing-platform-based chemiluminescence resonance energy transfer and its immunoassay application. Anal Chem. 2012;84:2708–12.
Huang X, Li L, Qian H, Dong C, Ren J. A resonance energy transfer between chemiluminescent donors and luminescent quantum-dots as acceptors (CRET). Angew Chem. 2006;118:5264–67.
Zhao S, Huang Y, Liu R, Shi M, Liu YM. A nonenzymatic chemiluminescent reaction enabling chemiluminescence resonance energy transfer to quantum dots. Chemistry. 2010;16:6142–5.
Algar WR, Wegner D, Huston AL, Blanco-Canosa JB, Stewart MH, Armstrong A, et al. Quantum dots as simultaneous acceptors and donors in time-gated forster resonance energy transfer relays: characterization and biosensing. J Am Chem Soc. 2012;134:1876–91.
So MK, Xu C, Loening AM, Gambhir SS, Rao J. Self-illuminating quantum dot conjugates for in vivo imaging. Nat Biotechnol. 2006;24:339–43.
He LJ, Wu MS, Xu JJ, Chen HY. A reusable potassium ion biosensor based on electrochemiluminescence resonance energy transfer. Chem Commun (Camb). 2013;49:1539–41.
Qi W, Wu D, Zhao J, Liu Z, Zhang W, Zhang L, et al. Electrochemiluminescence resonance energy transfer based on ru(phen)3 2+-doped silica nanoparticles and its application in "turn-on" detection of ozone. Anal Chem. 2013;85:3207–12.
Shan Y, Xu JJ, Chen HY. Distance-dependent quenching and enhancing of electrochemiluminescence from a CdS:Mn nanocrystal film by Au nanoparticles for highly sensitive detection of DNA. Chem Commun (Camb) .2009;905-7.
Shan Y, Xu JJ, Chen HY. Electrochemiluminescence quenching by CdTe quantum dots through energy scavenging for ultrasensitive detection of antigen. Chem Commun (Camb). 2010;46:5079–81.
Wu MS, Shi HW, He LJ, Xu JJ, Chen HY. Microchip device with 64-site electrode array for multiplexed immunoassay of cell surface antigens based on electrochemiluminescence resonance energy transfer. Anal Chem. 2012;84:4207–13.
Wang F, Liu X. Recent advances in the chemistry of lanthanide-doped upconversion nanocrystals. Chem Soc Rev. 2009;38:976–89.
An M, Cui J, He Q, Wang L. Down-/up-conversion luminescence nanocomposites for dual-modal cell imaging. J Mater Chem B. 2013;1:1333–9.
Wang L, Li Y. Green upconversion nanocrystals for DNA detection. Chem Commun (Camb). 2006;2557-9.
Chen H, Guan Y, Wang S, Ji Y, Gong M, Wang L. Turn-on detection of a cancer marker based on near-infrared luminescence energy transfer from NaYF4:Yb, Tm/NaGdF4 core-shell upconverting nanoparticles to gold nanorods. Langmuir. 2014;30:13085–91.
Yuan F, Chen H, Xu J, Zhang Y, Wu Y, Wang L. Aptamer-based luminescence energy transfer from near-infrared-to-near-infrared upconverting nanoparticles to gold nanorods and its application for the detection of thrombin. Chemistry. 2014;20:2888–94.
Zhang J, Wang S, Gao N, Feng D, Wang L, Chen H. Luminescence energy transfer detection of PSA in red region based on Mn2+-enhanced NaYF4:Yb, Er upconversion nanorods. Biosens Bioelectron. 2015;72:282–7.
Guo X, Wu S, Duan N, Wang Z. Mn2+-doped NaYF4:Yb/Er upconversion nanoparticle-based electrochemiluminescent aptasensor for bisphenol A. Anal Bioanal Chem. 2016;408:3823–31.
Liu M, Ye Y, Yao C, Zhao W, Huang X. Mn2+-doped NaYF4:Yb/Er upconversion nanoparticles with amplified electrogenerated chemiluminescence for tumor biomarker detection. J Mater Chem B. 2014;2:6626–33.
Sim HG, Cheng CW. Changing demography of prostate cancer in Asia. Eur J Cancer. 2005;41:834–45.
Li L; Chen Y; Lu Q; Ji J; Shen Y; Xu M; Fei R; Yang G; Zhang K; Zhang J.-R; Zhu J-J Electrochemiluminescence energy transfer-promoted ultrasensitive immunoassay using near-infrared-emitting CdSeTe/CdS/ZnS quantum dots and gold nanorods. Scientific reports 2013:3.
Wang L, Zhang Y, Zhu Y. One-pot synthesis and strong near-infrared upconversion luminescence of poly(acrylic acid)-functionalized YF3:Yb3+/Er3+ nanocrystals. Nano Res. 2010;3:317–25.
Tian G, Gu Z, Zhou L, Yin W, Liu X, Yan L, et al. Mn2+ dopant-controlled synthesis of NaYF4:Yb/Er upconversion nanoparticles for in vivo imaging and drug delivery. Adv Mater. 2012;24:1226–31.
Wu S, Duan N, Zhang H, Wang Z. Simultaneous detection of microcysin-LR and okadaic acid using a dual fluorescence resonance energy transfer aptasensor. Anal Bioanal Chem. 2014;407:1303–12.
Dai Y, Xiao H, Liu J, Yuan Q, Ma P, Yang D, et al. In vivo multimodality imaging and cancer therapy by near-infrared light-triggered trans-platinum pro-drug-conjugated upconverison nanoparticles. J Am Chem Soc. 2013;135:18920–9.
Li LL, Wu P, Hwang K, Lu Y. An exceptionally simple strategy for DNA-functionalized up-conversion nanoparticles as biocompatible agents for nanoassembly, DNA delivery, and imaging. J Am Chem Soc. 2013;135:2411–4.
Li Z, Lv S, Wang Y, Chen S, Liu Z. Construction of LRET-based nanoprobe using upconversion nanoparticles with confined emitters and bared surface as luminophore. J Am Chem Soc. 2015;137:3421–7.
Yang J, Shen D, Li X, Li W, Fang Y, Wei Y, et al. One-step hydrothermal synthesis of carboxyl-functionalized upconversion phosphors for bioapplications. Chem Eur J. 2012;18:13642–50.
Ding Y, Zhu H, Zhang X, Zhu JJ, Burda C. Rhodamine B derivative-functionalized upconversion nanoparticles for FRET-based Fe(3+)-sensing. Chem Commun (Camb). 2013;49:7797–9.
Li H, Wang L. NaYF4:Yb3+/Er3+ nanoparticle-based upconversion luminescence resonance energy transfer sensor for mercury(II) quantification. Analyst. 2013;138:1589–95.
Orendorff CJ, Murphy CJ. Quantitation of metal content in the silver-assisted growth of gold nanorods. J Phys Chem B. 2006;110:3990–4.
Nikoobakht B, El-Sayed MA. Preparation and growth mechanism of gold nanorods (NRs) using seed-mediated growth method. Chem Mater. 2003;15:1957–62.
Yu C, Irudayaraj J. Quantitative evaluation of sensitivity and selectivity of multiplex nanoSPR biosensor assays. Biophys J. 2007;93:3684–92.
Zhang R, Fan L, Fang Y, Yang S. Electrochemical route to the preparation of highly dispersed composites of ZnO/carbon nanotubes with significantly enhanced electrochemiluminescence from ZnO. J Mater Chem. 2008;18:4964–70.
Huang H, Li J, Zhu J-J. Electrochemiluminescence based on quantum dots and their analytical application. Anal Methods. 2011;3:33–42.
Wang L, Xu S, Li H, Yue Q, Gu X, Zhang S, et al. Study for the electrochemical deposition on single carbon fiber and electrochemiluminescence of ZnO nanostructures. CrystEngComm. 2013;15:8444–9.
Li L-L, Ji J, Fei R, Wang C-Z, Lu Q, Zhang J-R, et al. A facile microwave avenue to electrochemiluminescent two-color graphene quantum dots. Adv Funct Mater. 2012;22:2971–79.
Chen Y, Yang M, Xiang Y, Yuan R, Chai Y. Binding-induced autonomous disassembly of aptamer-DNAzyme supersandwich nanostructures for sensitive electrochemiluminescence turn-on detection of ochratoxin A. Nanoscale. 2014;6:1099–104.
Ma MN, Zhang X, Zhuo Y, Chai YQ, Yuan R. An amplified electrochemiluminescent aptasensor using Au nanoparticles capped by 3,4,9,10-perylene tetracarboxylic acid-thiosemicarbazide functionalized C60 nanocomposites as a signal enhancement tag. Nanoscale. 2015;7:2085–92.
Shang L, Dong SJ. Design of fluorescent assays for cyanide and hydrogen peroxide based on the inner filter effect of metal nanoparticles. Anal Chem. 2009;81:1465–70.
Lang QL, Wang F, Yin L, Liu MJ, Petrenko VA, Liu AH. Specific probe selection from landscape phage display library and its application in enzyme-linked immunosorbent assay of free prostate-specific antigen. Anal Chem. 2014;86:2767–74.
Kong RM, Ding L, Wang ZJ, You JM, Qu FL. A novel aptamer-functionalized MoS2 nanosheet fluorescent biosensor for sensitive detection of prostate specific antigen. Anal Bioanal Chem. 2015;407:369–77.
Choi HK, Lee JH. Role of magnetic Fe3O4 graphene oxide in chemiluminescent aptasensors capable of sensing tumor markers in human serum. Anal Methods-UK. 2013;5:6964–8.
Zhang JG, Wang SZ, Gao N, Feng DX, Wang L, Chen HQ. Luminescence energy transfer detection of PSA in red region based on Mn2+-enhanced NaYF4:Yb, Er upconversion nanorods. Biosens Bioelectron. 2015;72:282–7.
Acknowledgements
This work was financially supported by the Natural Science Foundation of China (21475001, 21675002), Anhui Provincial Natural Science Foundation (1408085QB40), Foundation for Innovation Team of Bioanalytical Chemistry and Special and Excellent Research Fund of Anhui Normal University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 219 kb)
Rights and permissions
About this article
Cite this article
Gao, N., Ling, B., Gao, Z. et al. Near-infrared-emitting NaYF4:Yb,Tm/Mn upconverting nanoparticle/gold nanorod electrochemiluminescence resonance energy transfer system for sensitive prostate-specific antigen detection. Anal Bioanal Chem 409, 2675–2683 (2017). https://doi.org/10.1007/s00216-017-0212-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-017-0212-2