Abstract
The efficient isolation and the accurate phenotype discrimination of circulating tumor cells (CTCs) are expected to provide much valuable information for the understanding of tumor metastasis and to play an important role in personalized treatment of cancer patients in the future. In this study, we developed a novel, rapid, and simple method for efficient capture and accurate identification of CTCs using aptamer conjugated magnetic beads and surface-enhanced Raman scattering (SERS) imaging technique. Using aptamer conjugated magnetic beads, rare target cancer cells can be captured efficiently from buffer and whole blood sample with capture efficiency of 73 % and 55 %, respectively. Meanwhile, captured cancer cells were labeled by specific SERS probes and can be identified readily and accurately by SERS imaging technique. Results of our experiment demonstrate the potential feasibility of aptamer conjugated magnetic beads coupled with SERS imaging technique for the efficient capture and accurate discrimination of CTCs in clinical whole blood sample.

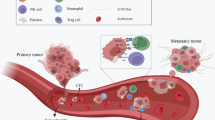
Schematic Representation of CTCs Capture and Identification Using Apt-MBs and SERS Imaging







Similar content being viewed by others
References
Maheswaran S, Haber DA (2010) Circulating tumor cells: a window into cancer biology and metastasis. Curr Opin Genet Dev 20(1):96–99
Paterlini-Brechot P, Benali NL (2007) Circulating tumor cells (CTC) detection: clinical impact and future directions. Cancer Lett 253(2):180–204
Ring A, Smith IE, Dowsett M (2004) Circulating tumor cells in breast cancer. Lancet Oncol 5(2):79–88
den Toonder J (2011) Circulating tumor cells: the grand challenge. Lab Chip 11(3):375–377
Pantel K, Alix-Panabie C (2010) Circulating tumor cells in cancer patients: challenges and perspectives. Trends Mol Med 16(9):398–406
Racila E, Euhus D, Weiss AJ, Rao C, McConnell J, Terstappen LW, Uhr JW (1998) Detection and characterization of carcinoma cells in the blood. Proc Natl Acad Sci U S A 95(8):4589–4594
Talasaz AH, Powell AA, Huber DE, Berbee JG, Roh KH, Yu W, Xiao W, Davis MM, Pease RF, Mindrinos MN, Jeffrey SS, Davis RW (2009) Isolating highly enriched populations of circulating epithelial cells and other rare cells from blood using a magnetic sweeper device. Proc Natl Acad Sci U S A 106(10):3970–3975
Flatmark K, Bjørnland K, Johannessen HO, Hegstad E, Rosales R, Hårklau L, Solhaug JH, Faye RS, Søreide O, Fodstad Ø (2002) Immunomagnetic detection of micrometastatic cells in bone marrow of colorectal cancer patients. Clin Cancer Res 8(2):444–449
Zigeuner RE, Riesenberg R, Pohla H, Hofstetter A, Oberneder R (2003) Isolation of circulating cancer cells from whole blood by immunomagnetic cell enrichment and unenriched immunocytochemistry in vitro. J Urol 169(2):701–705
Wang X, Qian XM, Beitler JJ, Chen ZG, Khuri FR, Lewis MM, Shin HJC, Nie SM, Shin DM (2011) Detection of circulating tumor cells in human peripheral blood using surface-enhanced Raman scattering nanoparticles. Cancer Res 71(5):1526–1532
Zheng S, Lin H, Liu JQ, Balic M, Datar R, Cote RJ, Tai YC (2007) Membrane microfilter device for selective capture electrolysis and genomic analysis of human circulating tumor cells. J Chromatogr A 1162(2):154–161
Wang Y, Zhou F, Liu X, Yuan L, Li D, Wang Y, Chen H (2013) Aptamer-modified micro/nanostructured surfaces: efficient capture of Ramos cells in serum environment. ACS Appl Mater Interfaces 5(9):3816–3823
Sheng W, Chen T, Kamath R, Xiong X, Tan W, Fan ZH (2012) Aptamer-enabled efficient isolation of cancer cells from whole blood using a microfluidic device. Anal Chem 84(9):4199–4206
Ozkumur E, Shah AM, Ciciliano JC, Emmink BL, Miyamoto DT, Brachtel E, Yu M, Chen PI, Morgan B, Trautwein J, Kimura A, Sengupta S, Stott SL, Karabacak NM, Barber TA, Walsh JR, Smith K, Spuhler PS, Sullivan JP, Lee RJ, Ting DT, Luo X, Shaw AT, Bardia A, Sequist LV, Louis DN, Maheswaran S, Kapur R, Haber DA, Toner M (2013) Inertial focusing for tumor antigen-dependent and -independent sorting of rare circulating tumor cells. Sci Transl Med 5(179), 179ra47
Riethdorf S, Fritsche H, Müller V, Rau T, Schindlbeck C, Rack B, Janni W, Coith C, Beck K, Jänicke F, Jackson S, Gornet T, Cristofanilli M, Pantel K (2007) Detection of circulating tumor cells in peripheral blood of patients with metastatic breast cancer: a validation study of the Cell Search system. Clin Cancer Res 13(3):920–928
Sefah K, Tang ZW, Shangguan DH, Chen H, Lopez-Colon D, Li Y, Parekh P, Martin J, Meng L, Phillips JA, Kim YM, Tan WH (2009) Molecular recognition of acute myeloid leukemia using aptamers. Leukemia 23(2):235–244
Patel DJ, Suri AK, Jiang L, Fan P, Kumar RA, Nonin S (1997) Structure recognition and adaptive binding in RNA aptamer complexes. J Mol Biol 272(5):645–664
Mairal T, Özalp VC, Sánchez PL, Mir M, Katakis I, O'Sullivan CK (2008) Aptamers: molecular tools for analytical applications. Anal Bioanal Chem 390(4):989–1007
Hu M, Zhang K (2013) The Application of aptamers in cancer research: an up-to-date review. Future Oncol 9(3):369–376
Daniels DA, Chen H, Hicke BJ, Swiderek KM, Gold L (2003) A tenascin-C aptamer identified by tumor cell SELEX: systematic evolution of ligands by exponential enrichment. Proc Natl Acad Sci U S A 100(26):15416–15421
Shangguan D, Li Y, Tang Z, Cao ZC, Chen HW, Mallikaratchy P, Sefah K, Yang CJ, Tan W (2006) Aptamers evolved from live cells as effective molecular probes for cancer study. Proc Natl Acad Sci U S A 103(32):11838–11843
Shangguan D, Cao ZC, Li Y, Tan W (2007) Aptamers evolved from cultured cancer cells reveal molecular differences of cancer cells in patient samples. Clin Chem 53(6):1153–1155
Sefah K, Meng L, Lopez-Colon D, Jimenez E, Liu C, Tan W (2010) DNA aptamers as molecular probes for colorectal cancer study. PLoS One 5(12), e14269
Zhang R, Zhang Y, Dong ZC, Jiang S, Zhang C, Chen LG, Zhang L, Liao Y, Aizpurua J, Luo Y, Yang JL, Hou JG (2013) Chemical mapping of a single molecule by plasmon-enhanced Raman scattering. Nature 498(7452):82–86
Vendrell M, Maiti KK, Dhaliwal K, Chang YT (2013) Surface-enhanced Raman scattering in cancer detection and imaging. Trends Biotechnol 31(4):249–257
Li MQ, Xu J, Romero-Gonzalez M, Banwart SA, Huang WE (2012) Single cell Raman spectroscopy for cell sorting and imaging. Curr Opin Biotechnol 23(1):56–63
Bantz KC, Meyer AF, Wittenberg NJ, Im H, Kurtulus O, Lee SH, Lindquist NC, Oh SH, Haynes CL (2011) Recent progress in SERS biosensing. Phys Chem Chem Phys 13(24):11551–11567
Pallaoro A, Hoonejani MR, Braun GB, Meinhart CD, Moskovits M (2015) Rapid identification by surface-enhanced Raman spectroscopy of cancer cells at low concentrations flowing in a microfluidic channel. ACS Nano 9(4):4328–4336
Nolan JP, Duggan E, Liu E, Condello D, Dave I, Stoner SA (2012) Single cell analysis using surface enhanced Raman scattering (SERS) tags. Methods 57(3):272–279
MacLaughlin CM, Mullaithilaga N, Yang GS, Ip SY, Wang C, Walker GC (2013) Surface-enhanced Raman scattering dye-labeled Au nanoparticles for triplexed detection of leukemia and lymphoma cells and SERS flow cytometry. Langmuir 29(6):1908–1919
Lee S, Chon H, Lee M, Choo J, Shin SY, Lee YH, Rhyu IJ, Son SW, Oh CH (2009) Surface-enhanced Raman scattering imaging of HER2 cancer markers overexpressed in single MCF7 cells using antibody conjugated hollow gold nanospheres. Biosens Bioelectron 24(7):2260–2263
Park H, Lee S, Chen L, Lee EK, Shin SY, Lee YH, Son SW, Oh CH, Song JM, Kang SH, Choo J (2009) SERS imaging of HER2-overexpressed MCF7 cells using antibody-conjugated gold nanorods. Phys Chem Chem Phys 11(34):7444–7449
Lee S, Kim S, Choo J, Shin SY, Lee YH, Choi HY, Ha S, Kang K, Oh CH (2007) Biological imaging of HEK293 cells expressing PLC gamma 1 using surface-enhanced Raman microscopy. Anal Chem 79(3):916–922
Freitag I, Matthaus C, Csaki A, Clement JH, Cialla-May D, Weber K, Krafft C, Popp J (2015) Differentiation of MCF-7 tumor cells from leukocytes and fibroblast cells using epithelial cell adhesion molecule targeted multicore surface-enhanced Raman spectroscopy labels. J Biomed Opt 20(5):055002. doi:10.1117/1.JBO.20.5.055002
Pallaoro A, Braun GB, Moskovits M (2011) Quantitative ratiometric discrimination between noncancerous and cancerous prostate cells based on neuropilin-1 overexpression. Proc Natl Acad Sci U S A 108(40):16559–16564
Maiti KK, Samanta A, Vendrell M, Soh KS, Olivo M, Chang YT (2011) Multiplex cancer cell detection by SERS nanotags with cyanine and triphenylmethine Raman reporters. Chem Commun 47(12):3514–3516
Yang J, Wang Z, Zong S, Song C, Zhang R, Cui Y (2012) Distinguishing breast cancer cells using surface-enhanced Raman scattering. Anal Bioanal Chem 402(3):1093–1100
Zhang P, Zhang R, Gao M, Zhang X (2014) Novel nitrocellulose membrane substrate for efficient analysis of circulating tumor cells coupled with surface-enhanced Raman scattering imaging. ACS Appl Mater Interfaces 6(1):370–376
Frens G (1973) Controlled nucleation for regulation of particle size in monodisperse gold suspensions. Nat Phys Sci 241(105):20–22
Acknowledgments
This work was supported by National Basic Research Program of China (Project 2012CB910604), the National High-Tech R&D Program (Project 2012AA020202), and the National Natural Science Foundation of China (Project: 21275034 and 21475027).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Compliance with Ethical Standards
ᅟ
Conflict of interest
The authors declare no competing financial interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 1.76 MB)
Rights and permissions
About this article
Cite this article
Sun, C., Zhang, R., Gao, M. et al. A rapid and simple method for efficient capture and accurate discrimination of circulating tumor cells using aptamer conjugated magnetic beads and surface-enhanced Raman scattering imaging. Anal Bioanal Chem 407, 8883–8892 (2015). https://doi.org/10.1007/s00216-015-9049-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-015-9049-8