Abstract
Purpose
MKNR3 is a paternally expressed gene whose mutations are the main cause of central precocious puberty (CPP). Protein circulating levels can be easily measured, as demonstrated in idiopathic CPP and healthy controls. No data are available for patients harboring an MKRN3 mutation. Our aim was to perform MKRN3 mutation screening and to investigate if circulating protein levels could be a screening tool to identify MKRN3 mutation in CPP patients.
Methods
We enrolled 140 CPP girls and performed MKRN3 mutation analysis. Patients were stratified into two groups: idiopathic CPP (iCPP) and MKRN3 mutation-related CPP (MKRN3-CPP). Clinical characteristics were collected. Serum MKRN3 values were measured by a commercially available ELISA assay kit in MKRN3-CPP and a subgroup of 15 iCPP patients.
Results
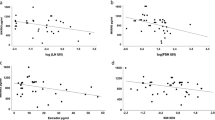
We identified 5 patients with MKRN3 mutations: one was a novel mutation (p.Gln352Arg) while the others were previously reported (p.Arg328Cys, p.Arg345Cys, p.Pro160Cysfs*14, p.Cys410Ter). There was a significant difference in circulating MKRN3 values in MKRN3-CPP compared to iCPP (p < 0.001). In MKRN3-CPP, the subject harboring Pro160Cysfs*14 presented undetectable levels. Subjects carrying the missense mutations p.Arg328Cys and p.Gln352Arg showed divergent circulating protein levels, respectively 40.56 pg/mL and undetectable. The patient with the non-sense mutation reported low but measurable MKRN3 levels (12.72 pg/mL).
Conclusions
MKRN3 defect in patients with CPP cannot be predicted by MKRN3 circulating levels, although those patients presented lower protein levels than iCPP. Due to the great inter-individual variability of the assay and the lack of reference values, no precise cut-off can be identified to suspect MKRN3 defect.

Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Avendaño MS, Vazquez MJ, Tena-Sempere M (2017) Disentangling puberty: novel neuroendocrine pathways and mechanisms for the control of mammalian puberty. Hum Reprod Update. https://doi.org/10.1093/humupd/dmx025. (PMID: 28961976)
Brito VN, Canton APM, Seraphim CE, Abreu AP, Macedo DB, Mendonca BB, Kaiser UB, Argente J, Latronico AC (2022) The congenital and acquired mechanisms implicated in the etiology of central precocious puberty. Endocr Rev. https://doi.org/10.1210/endrev/bnac020. (Epub ahead of print. PMID: 35930274)
Tinano FR, Canton APM, Montenegro LR, de Castro LA, Faria AG, Seraphim CE, Brauner R, Jorge AA, Mendonca BB, Argente J, Brito VN, Latronico AC (2023) Clinical and genetic characterization of familial central precocious puberty. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgac763
Valadares LP, Meireles CG, De Toledo IP et al (2019) MKRN3 Mutations in central precocious puberty: a systematic review and meta-analysis. J Endocr Soc. https://doi.org/10.1210/js.2019-00041
Tinano FR, Canton APM, Montenegro LR et al (2023) Clinical and genetic characterization of familial central precocious puberty. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgac763
Palumbo S, Cirillo G, Aiello F, Papparella A, Miraglia Del Giudice E, Grandone A (2022) MKRN3 role in regulating pubertal onset: the state of art of functional studies. Front Endocrinol (Lausanne). https://doi.org/10.3389/fendo.2022.991322
Bessa DS, Macedo DB, Brito VN, França MM, Montenegro LR, Cunha-Silva M et al (2017) High frequency of MKRN3 mutations in Male central precocious puberty previously classified as idiopathic. Neuroendocrinology. https://doi.org/10.1159/000446963
Pagani S, Calcaterra V, Acquafredda G et al (2020) MKRN3 and KISS1R mutations in precocious and early puberty. Ital J Pediatr. https://doi.org/10.1186/s13052-020-0808-6
Hughes IA (2013) Releasing the brake on puberty. N Engl J Med. https://doi.org/10.1056/NEJMe1306743
Abreu AP, Toro CA, Song YB et al (2020) MKRN3 inhibits the reproductive axis through actions in kisspeptin-expressing neurons. J Clin Invest. https://doi.org/10.1172/JCI136564
Liu H, Kong X, Chen F (2017) Mkrn3 functions as a novel ubiquitin E3 ligase to inhibit Nptx1 during puberty initiation. Oncotarget. https://doi.org/10.18632/oncotarget.19347
Omwancha J, Zhou XF, Chen SY, Baslan T, Fisher CJ, Zheng Z et al (2006) Makorin RING finger protein 1 (MKRN1) has negative and positive effects on RNA polymerase II-dependent transcription. Endocrine. https://doi.org/10.1385/ENDO:29:2:363
Li C, Lu W, Yang L, Li Z, Zhou X, Guo R et al (2020) MKRN3 regulates the epigenetic switch of mammalian puberty via ubiquitination of MBD3. Natl Sci Rev. https://doi.org/10.1093/nsr/nwaa023
Hagen CP, Sørensen K, Mieritz MG, Johannsen TH, Almstrup K, Juul A (2015) Circulating MKRN3 levels decline prior to pubertal onset and through puberty: a longitudinal study of healthy girls. J Clin Endocrinol Metab. https://doi.org/10.1210/jc.2014-4462
Busch AS, Hagen CP, Almstrup K, Juul A (2016) Circulating MKRN3 levels decline during puberty in healthy boys. J Clin Endocrinol Metab. https://doi.org/10.1210/jc.2016-1488
Grandone A, Cirillo G, Sasso M, Tornese G, Luongo C, Festa A, Marzuillo P, Miraglia Del Giudice E (2018) MKRN3 levels in girls with central precocious puberty during GnRHa treatment: a longitudinal study. Horm Res Paediatr. https://doi.org/10.1159/000493134
Macedo DB, Abreu AP, Reis ACS et al (2014) Central precocious puberty that appears to be sporadic caused by paternally inherited mutations in the imprinted gene makorin ring finger 3. J Clin Endocrinol Metab. https://doi.org/10.1210/jc.2013-3126
Simon D, Ba I, Mekhail N et al (2016) Mutations in the maternally imprinted gene MKRN3 are common in familial central precocious puberty. Eur J Endocrinol. https://doi.org/10.1530/EJE-15-0488
Seraphim CE, Canton APM, Montenegro L, Piovesan MR, Macedo DB, Cunha M, Guimaraes A, Ramos CO, Benedetti AFF, de Castro LA, Gagliardi PC, Antonini SR, Gryngarten M, Arcari AJ, Abreu AP, Kaiser UB, Soriano-Guillén L, Escribano-Muñoz A, Corripio R, Labarta JI, Travieso-Suárez L, Ortiz-Cabrera NV, Argente J, Mendonca BB, Brito VN, Latronico AC (2021) Genotype-phenotype correlations in central precocious puberty caused by MKRN3 mutations. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgaa955
Magnotto JC, Mancini A, Bird K et al (2023) Novel MKRN3 missense mutations associated with central precocious puberty reveal distinct effects on ubiquitination. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgad151
Cole TJ, Lobstein T (2012) Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity. Pediatr Obes. https://doi.org/10.1111/j.2047-6310.2012.00064
Tanner JM (1976) Whitehouse RHClinical longitudinal standards for height, weight, height velocity, weight velocity, and stages of puberty. Arch Dis Child 51:170–179
Valadares LP, Meireles CG, De Toledo IP, Santarem de Oliveira R, Gonçalves de Castro LC, Abreu AP, Carroll RS, Latronico AC, Kaiser UB, Guerra ENS, Lofrano-Porto A (2019) MKRN3 mutations in central precocious puberty: a systematic review and meta-analysis. J Endocr Soc. https://doi.org/10.1210/js.2019-00041
Grandone A, Capristo C, Cirillo G, Sasso M, Umano GR, Mariani M, Miraglia Del Giudice E, Perrone L (2017) Molecular screening of MKRN3, DLK1, and KCNK9 genes in girls with idiopathic central precocious puberty. Horm Res Paediatr. https://doi.org/10.1159/000477441
Magnotto JC, Mancini A, Bird K, Montenegro L, Tütüncüler F, Pereira SA, Simas V, Garcia L, Roberts SA, Macedo D, Magnuson M, Gagliardi P, Mauras N, Witchel SF, Carroll RS, Latronico AC, Kaiser UB, Abreu AP (2023) Novel MKRN3 missense mutations associated with central precocious puberty reveal distinct effects on ubiquitination. The J Clin Endocrinol Metabol 108(7):1646–1656. https://doi.org/10.1210/clinem/dgad151
Aycan Z, Savaş-Erdeve Ş, Çetinkaya S, Kurnaz E, Keskin M, Muratoğlu Şahin N, Bayramoğlu E, Ceylaner G (2018) Investigation of MKRN3 mutation in patients with familial central precocious puberty. J Clin Res Pediatr Endocrinol. https://doi.org/10.4274/jcrpe.5506
Ortiz-Cabrera NV, Riveiro-Álvarez R, López-Martínez MÁ, Pérez-Segura P, Aragón-Gómez I, Trujillo-Tiebas MJ, Soriano-Guillén L (2017) Clinical exome sequencing reveals MKRN3 pathogenic variants in familial and nonfamilial idiopathic central precocious puberty. Horm Res Paediatr. https://doi.org/10.1159/000453262
Kırkgöz T, Kaygusuz SB, Alavanda C, Helvacıoğlu D, Abalı ZY, Tosun BG, Eltan M, Menevşe TS, Guran T, Arman A, Turan S, Bereket A (2023) Molecular analysis of MKRN3 gene in Turkish girls with sporadic and familial idiopathic central precocious puberty. J Pediatr Endocrinol Metab. https://doi.org/10.1515/jpem-2022-0645
Roberts SA, Kaiser UB (2020) Genetics in endocrinology: genetic etiologies of central precocious puberty and the role of imprinted genes. Eur J Endocrinol. https://doi.org/10.1530/EJE-20-0103
Suzuki E, Shima H, Kagami M, Soneda S, Tanaka T, Yatsuga S et al (2019) (Epi)genetic defects of MKRN3 are rare in Asian patients with central precocious puberty. Hum Genome Var. https://doi.org/10.1038/s41439-019-0039-9
Dauber A, Cunha-Silva M, Macedo DB, Brito VN, Abreu AP, Roberts SA, Montenegro LR, Andrew M, Kirby A, Weirauch MT, Labilloy G, Bessa DS, Carroll RS, Jacobs DC, Chappell PE, Mendonca BB, Haig D, Kaiser UB, Latronico AC (2017) Paternally inherited DLK1 deletion associated with familial central precocious puberty. J Clin Endocrinol Metab. https://doi.org/10.1210/jc.2016-3677
Montenegro L, Labarta JI, Piovesan M, Canton APM, Corripio R, Soriano-Guillén L, Travieso-Suárez L, Martín-Rivada Á, Barrios V, Seraphim CE, Brito VN, Latronico AC, Argente J (2020) Novel genetic and biochemical findings of DLK1 in children with central precocious puberty: a brazilian-spanish study. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgaa461
Palumbo S, Cirillo G, Sanchez G, Aiello F, Fachin A, Baldo F, Pellegrin MC, Cassio A, Salerno M, Maghnie M, Faienza MF, Wasniewska M, Fintini D, Giacomozzi C, Ciccone S, Miraglia Del Giudice E, Tornese G, Grandone A (2022) A new DLK1 defect in a family with idiopathic central precocious puberty: elucidation of the male phenotype. J Endocrinol Invest. https://doi.org/10.1007/s40618-022-01997-y
Roberts SA, Abreu AP, Navarro VM, Liang JN, Maguire CA, Kim HK, Carroll RS, Kaiser UB (2020) The Peripubertal decline in makorin ring finger protein 3 expression is independent of leptin action. J Endocr Soc. 4(7):059. https://doi.org/10.1210/jendso/bvaa059. (PMID: 32587933; PMCID: PMC7304661)
Mariani M, Fintini D, Cirillo G, Palumbo S, Del Giudice EM, Bocchini S, Manco M, Cappa M, Grandone A (2022) MKRN3 circulating levels in Prader-Willi syndrome: a pilot study. J Endocrinol Invest. https://doi.org/10.1007/s40618-022-01860-0
Ge W, Wang HL, Shao HJ, Liu HW, Xu RY (2020) Evaluation of serum makorin ring finger protein 3 (MKRN3) levels in girls with idiopathic central precocious puberty and premature thelarche. Physiol Res. https://doi.org/10.33549/physiolres.934222
Acknowledgements
We gratefully thank all the patients, family members, and staff from all the units that participated in the study. We thank all members of the Italian Society for Pediatric Endocrinology and Diabetology (SIEDP) study group on growth and pubertal development for supporting the study. A special thanks to all the Italian Medical Center who participated in enrolling the patients, below the list: Endocrinological Unit of AOU “Federico II”, Naples; Clinic of Pediatrics, University of Chieti, Chieti; Pediatric Unit, University of Modena and Reggio Emilia, Modena; Endocrinological Unit, Department of Pediatrics, Pediatric Hospital Giovanni XXIII, University of Bari, Bari; Unit of Pediatrics, Department of Medicine and Surgery, University of Parma, Parma; Department of Pediatric Endocrinology, Unit of Pediatrics, Department of Medical & Surgical Sciences, University of Bologna, Bologna; Medical Genetic and Endocrinology of Cesena, AUSL Romagna, Pediatric Endocrinology unit, Department of Pediatrics, "Carlo Poma" Hospital, Mantova; Endocrinology Unit, Pediatric University Department, Bambino Gesù Children's Hospital, Rome; Pediatric Endocrinology Unit, Department of Pediatrics, Santobono-Pausilipon Children's Hospital, Pediatric Unit, University of Modena and Reggio Emilia, Modena, Italy.
Funding
This work was supported by a grant (390) funded by “VALERE: VAnviteLli pEr la RicErca” program of University of Campania “L. Vanvitelli”.
Author information
Authors and Affiliations
Contributions
AG conceived the study idea and supervised the work process. AG, SP and GC elaborated the design. Recruitment and clinical management were performed by GT, DF, MW, MFF, CL, AF, EMG. Genetic analysis was performed by SP and GC. FA reviewed the literature, collected the data, wrote the first draft of the manuscript. All authors commented on previous versions of the manuscript. FA prepared the drafts for publication. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethical approval
This study involved human participants, their data and biological material. Approval was obtained from the ethics committee of University “Luigi Vanvitelli”, Naples, Italy. The procedures used in this study adhere to the tenets of the 1964 Declaration of Helsinki and its later amendments.
Research involving human participants and/or animals
The modified the ethical approval disclosure to be more specific about this research including human participants.
Consent to participate
Written informed consent was obtained from all parents of individual participants included in the study. An oral assent was obtained by the patients themselves.
Consent to publish
The authors affirm that human research participants provided informed consent for publication of all the clinical and molecular data inhere presented in the document.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aiello, F., Palumbo, S., Cirillo, G. et al. MKRN3 circulating levels in girls with central precocious puberty caused by MKRN3 gene mutations. J Endocrinol Invest (2023). https://doi.org/10.1007/s40618-023-02255-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40618-023-02255-5