Abstract
Background
Qualitative research is fundamental for designing discrete choice experiments (DCEs) but is often underreported in the preference literature. We developed a DCE to elicit preferences for vaccination against invasive meningococcal disease (IMD) among adolescents and young people (AYP) and parents and legal guardians (PLG) in the United States. This article reports the targeted literature review and qualitative interviews that informed the DCE design and demonstrates how to apply the recent reporting guidelines for qualitative developmental work in preference studies.
Methods
This study included two parts: a targeted literature review and qualitative interviews. The Medline and Embase databases were searched for quantitative and qualitative studies on IMD and immunization. The results of the targeted literature review informed a qualitative interview guide. Sixty-minute, online, semi-structured interviews with AYP and PLG were used to identify themes related to willingness to be vaccinated against IMD. Participants were recruited through a third-party recruiter’s database and commercial online panels. Interviews included vignettes about IMD and vaccinations and three thresholding exercises examining the effect of incidence rate, disability rate, and fatality rate on vaccination preferences. Participant responses related to the themes were counted.
Results
The targeted literature review identified 31 concepts that were synthesized into six topics for the qualitative interviews. Twenty AYP aged 16–23 years and 20 PLG of adolescents aged 11–17 years were interviewed. Four themes related to willingness to be vaccinated emerged: attitudes towards vaccination, knowledge and information, perception of IMD, and vaccine attributes. Most participants were concerned about IMD (AYP 60%; PLG 85%) and had positive views of vaccination (AYP 80%; PLG 60%). Ninety percent of AYP and 75% of PLG always chose vaccination over no vaccination, independent of IMD incidence rate, disability rate, or fatality rate.
Conclusion
Willingness to be vaccinated against IMD was affected by vaccine attributes but largely insensitive to IMD incidence and severity. This article provides an example of how to apply the recent reporting guidelines for qualitative developmental work in preference studies, with 21 out of 22 items in the guidelines being considered.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The design of quantitative patient preference elicitation instruments is often informed by qualitative research, but this process is often inadequately reported. |
This article serves as an example of how to apply the new reporting guidelines for qualitative developmental work in preference studies. |
Four themes related to willingness to be vaccinated against invasive meningococcal disease emerged from the qualitative research: attitudes towards vaccination, knowledge and information, perception of invasive meningococcal disease, and vaccine attributes. These themes informed the design of a discrete choice experiment. |
1 Introduction
Qualitative research is fundamental for designing robust and reliable quantitative patient preference elicitation instruments and is increasingly published as standalone research [1, 2]. However, systematic underreporting of the process that informed the final design of a preference elicitation instrument persists in the preference literature. A systematic review of published discrete choice experiments (DCEs) found that 89% (n = 225) of the reviewed studies did not report the qualitative developmental component of the research ‘in detail’ [1].
General guidelines for the reporting of qualitative research have been available [3,4,5,6], but guidelines for reporting qualitative research specific to the design of preference elicitation instruments are relatively new [2]. The patient preference-specific guidelines require transparent disclosure of the qualitative data generation, analysis approach, and processes that led to the final instrument design. This is important to clearly understand the steps that resulted in the final instrument design (e.g., selection of considered treatment attributes). By promoting transparency, these guidelines aim to help researchers, reviewers, and decision makers to evaluate the validity of preference elicitation instruments and interpret the results [2]. In this study, we provide an example of how to apply these reporting guidelines.
Although qualitative methods are generally used, and necessary, for developing DCE attributes, non-qualitative methods are also important for designing DCEs to address a research question with a practical impact [7]. For example, Helter and Boehler proposed a systematic approach to developing attributes for DCE studies that combined qualitative and non-qualitative methods [7]. The real-world decision context is a key consideration in developing DCE attributes [8]. Specifically, DCE attributes should differentiate between real-world alternatives to make findings actionable for decision makers.
We developed a DCE to elicit preferences for vaccination against invasive meningococcal disease (IMD), which was systematically informed by a targeted literature review and qualitative patient preference research by following the guidelines [2]. In the United States (US), MenB (monovalent) vaccine uptake is poor, with only 29.4% of 17-year-olds having received at least one dose and 11.9% having received at least two doses in 2022 [9]. MenACWY (quadrivalent) vaccine uptake is better, with 88.6% of 13–17-year-olds having received at least one dose and 60.8% having received at least two doses [9].
Understanding IMD vaccination preferences is necessary for increasing vaccine uptake. For example, if people prefer fewer injections, this suggests that the availability of MenABCWY (pentavalent) vaccines may increase vaccine uptake, since they provide the same coverage with two injections instead of three. Qualitative research on this topic is required, particularly to understand the preferences of adolescents who are central to vaccination programs but are frequently underrepresented in qualitative literature [10]. One previous qualitative study has explored MenACWY vaccine-related knowledge, attitudes, and decision making. Painter and colleagues found that lack of knowledge on vaccines was a barrier to vaccine uptake, and mothers were the main decision makers for their adolescent daughters regarding vaccine uptake [11].
We developed a DCE to quantify the drivers of IMD vaccination preferences and willingness to be vaccinated among adolescents and young people (AYP) and parents and legal guardians (PLG) in the US. In this study, by following the recently published reporting guidelines [2], we report the targeted literature review and qualitative interviews that informed the development of the DCE attributes and attribute levels. In doing so, we specifically demonstrate how the recent reporting guidelines can be implemented.
2 Targeted Literature Review
2.1 Methods
A targeted literature review of published quantitative preference studies and qualitative studies that investigated attitudes towards IMD and willingness to be vaccinated was conducted [12]. Findings from this review were supplemented with clinical data about IMD vaccinations from the US labels [13]. The review aimed to identify preference-relevant aspects of IMD and IMD vaccination for discussion in the semi-structured interviews.
2.1.1 Search Strategy
Two searches were performed in Ovid (https://ovidsp.ovid.com/) using the Medline and Embase databases. The search strategy for quantitative preference studies included terms related to IMD and immunization, acceptable risks and benefits, and willingness to accept and pay for a vaccine (Online Resource Table S1). The search strategy for qualitative studies included terms related to IMD and immunization, beliefs about disease impact and vaccines, and qualitative methods, including interviews and focus groups (Online Resource Table S2).
2.1.2 Study Eligibility and Data Extraction
Two authors (CMI and CW) screened the studies and extracted the data. Studies were screened in two stages by title and abstract (‘screening stage’), then by full text (‘eligibility stage’). Studies identified in the search were screened based on the following prespecified eligibility criteria: English language peer-reviewed publication from 2000 to 2021 or conference abstract from 2012 to 2021; quantitative or qualitative methods; and participants who were potential vaccine recipients, parents of potential vaccine recipients, or health care providers involved in vaccine delivery or decision making (Online Resource Table S3). Study objectives, year, location, respondent type, vaccine population, sample size, and results were extracted from eligible studies (Online Resource Tables S4–S6). The attributes and attribute levels were recorded from any eligible study that used a quantitative preference method, such as a DCE. Attributes represented included effectiveness, duration of vaccine effect, adverse events, number of injections, cost, and serogroups covered.
2.2 Results
2.2.1 Characteristics of the Included Studies
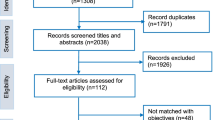
Nineteen quantitative studies and one qualitative study were eligible for inclusion in the targeted literature review (Online Resource Fig. S1). Quantitative studies used either a DCE (n = 4) or a survey (n = 15). Studies included PLG (n = 13), physicians/other healthcare providers (n = 6), adolescents (n = 3), and/or young adults (n = 1) from the US (n = 7), France (n = 4), Italy (n = 3), Germany (n = 2), Australia (n = 2), Canada (n = 1), The Netherlands (n = 1), and/or the UK (n = 1).
2.2.2 Factors Related to Meningococcal Vaccination
Eleven factors related to attitudes towards meningococcal vaccination were identified from quantitative studies that used a DCE design, and 12 factors associated with willingness to be vaccinated were identified from quantitative studies that did not use a DCE design (Table 1). Cost was the factor most frequently related to attitudes towards IMD vaccination, and perceptions of seriousness or severity of IMD was the factor most frequently related to willingness to be vaccinated. Three domains were identified from the qualitative study in the literature review: overall attitudes towards vaccines, knowledge and attitudes toward adolescent vaccines, and vaccine decision making (Online Resource Table S6).
The results of the targeted literature review directly informed the design of the qualitative interview guide (Online Resource), consistent with recommendations in the reporting guidelines [2]. A total of 31 concepts potentially affecting individuals’ perceptions of IMD or preferences for vaccination were extracted and synthesized into six topics (Table 2). These six topics were included in the qualitative interviews through three mediums: an open discussion with participants using a semi-structured interview guide, three vignettes, and three thresholding exercises. The semi-structured interview guide included probing questions about general attitudes towards adolescent/young person vaccinations and awareness of vaccines to determine participants’ level of understanding and opinions. The vignettes presented information about IMD and vaccinations, and participants were asked prespecified questions related to the vignettes. The thresholding exercises examined the influence of IMD incidence rate, disability rate, and fatality rate on willingness to be vaccinated.
3 Qualitative Interviews
3.1 Methods
3.1.1 Study Design
Semi-structured interviews were conducted to inform the subsequent DCE design. The qualitative study received ethical approval from Ethical and Independent Review Services (reference: 22049-01, 29 March 2022). The full interview guide is provided in the Online Resource.
3.1.2 Participants
The recruitment target was 40 participants (20 AYP and 20 PLG). This sample size was based on our experience in conducting qualitative research to inform the development of quantitative preference instruments, and it is also aligned with previous research and guidelines [14]. Recent publications have demonstrated that 97% of all concepts emerge by the 20th interview [15]. Therefore, 20 participants were recruited from each subgroup as an estimate of the number of interviews that might be needed to achieve data saturation. An interim analysis of 50% of completed interviews (AYP, n = 10; PLG, n = 10) determined that data saturation of all major themes had not been reached; therefore, the remaining 50% of interviews were conducted. The sample size (n = 40) was adequate for the thresholding exercises because the data are elicited at the individual level, meaning that individual-level modeling can be conducted.
Participants were identified through a third-party recruiter’s database and commercial online panels. A non-probability target sampling approach was used. Sample targets were generated based on the 2022 US census to recruit a diverse but representative sample. Sample targets included sex, race, ethnicity, parental education, location (rural, suburban), US region (Northeast, Midwest, South, West), and vaccination experience (Online Resource Table S7). To be eligible to participate, individuals had to either be aged 16–23 years or a parent of an adolescent aged 11–17 years, as IMD vaccination is recommended for these age groups [9]. Participants had to be residing in the US, fluent in English, and able to provide online assent (if aged 16–17 years) or consent (if aged ≥18 years). Individuals aged 16–17 years also required online consent from a PLG to participate. Participants were not excluded based on being related to other participants. Interviews took place in June 2022, and each participant was reimbursed $100 for their time.
3.1.3 Measures and Procedures
After providing assent/consent, potential participants answered prescreening questions by phone. Participants then completed a short online screening questionnaire that also captured participant characteristics and vaccine hesitancy, as measured by the Vaccine Hesitancy Scale [16]. Sixty-minute semi-structured interviews were conducted virtually via a web-assisted conferencing platform (Microsoft Teams) by researchers with qualitative research experience, and interviews were audio-recorded.
Interviews had three parts and followed a semi-structured interview guide (Online Resource). Part A included a study introduction and an open discussion to understand participants’ attitudes towards childhood/adolescent vaccination. Part B included a discussion of three vignettes (Figs. 1, 2, 3) to understand factors affecting AYP’s willingness to be vaccinated or PLG’ willingness for their child to be vaccinated. Vignette 1 covered the overall disease context, IMD-related health outcomes, and information about IMD and meningococcal vaccinations. Vignette 2 covered currently available MenACWY and MenB vaccines, the potential MenABCWY vaccine, recommended dosing schedules, and willingness to be vaccinated against IMD. Vignette 3 covered current US Advisory Committee on Immunization Practices (ACIP) guidance for IMD vaccinations, influence of physicians’ recommendations, and perceptions of access to vaccine information and vaccines. Participants read a vignette topic (e.g., ‘What is meningitis’) and interviewers then asked questions (‘discussion probes’) relating to that vignette topic before moving to the next.
Part C included three thresholding exercises [17] (see Fig. 4 for an example) that assessed how AYP’s willingness to be vaccinated or PLG’ willingness for their child to be vaccinated was affected by IMD incidence rate, disability rate, and fatality rate. Incidence rate was defined as the number of new cases of meningitis within a time period out of the number of people at risk. Disability rate was defined as the number of people who have long-term consequences from meningitis, despite treatment, out of the number of people who get meningitis. Fatality rate was defined as the number of people who die from meningitis, despite treatment, out of the number of people who get meningitis.
Each thresholding exercise used three binary choice questions, where participants selected either a ‘no vaccination’ option or a hypothetical ‘vaccination for MenACWY and MenB’ option. The first thresholding exercise varied the IMD incidence rate between 10 and 500 out of 100 million people, with the IMD incidence rate in the initial choice question set to 200 out of 100 million people. The second thresholding exercise varied the disability rate between 5 and 80%, with the disability rate in the initial choice question set to 20%. The third thresholding exercise varied the fatality rate between 5 and 60%, with the fatality rate in the initial choice question set to 20%. Choice questions were adaptive, such that IMD incidence rate, disability rate, and fatality rate were increased in the next question when ‘no vaccination’ was chosen as the response to a given question and decreased when ‘vaccination’ was chosen. The 10-year vaccine efficacy was fixed at 80% and the vaccine was assumed to have no impact on disability rate or fatality rate. For each thresholding exercise, each participant made three choices out of a possible seven choice tasks (i.e., Choice 1, Choice 2A, Choice 2B, Choice 3AA, Choice 3AB, Choice 3BA, Choice 3BB). Two example choice tasks are presented in the supplementary information.
3.1.4 Analyses and Interpretation
Qualitative data were analyzed thematically and managed using ATLAS.ti version 8 [18]. The analysis included a deductive ‘top–down’ element involving prespecified codes in a coding dictionary developed from the interview guide and an inductive ‘bottom-up’ element involving expanding and adapting the coding dictionary based on emerging themes. Qualitative analyses were conducted in three rounds. In round 1, two analysts (CW, CMI) coded two transcripts independently. In round 2, the same two analysts coded two further transcripts independently using the expanded coding dictionary from round 1. In round 3, all remaining transcripts were split across analysts and coded using the expanded coding dictionary from round 2. At each round, coding dictionaries were merged across analysts, and inconsistencies were resolved by discussion.
Main themes and subthemes were grouped and summarized in a conceptual map, which was iteratively designed using input from interview moderators and qualitative analysts to summarize codes and groups of codes hierarchically. Merging and adding codes were discussed and harmonized among analysts, and disagreements were resolved by a senior researcher (CMI). The conceptual map summarizes the key drivers of treatment valuation. The conceptual map aims to generate a data-driven image of the research findings. Similar processes have been used in the literature to summarize themes and subthemes emerging from qualitative research [19]. Participant responses related to the themes were counted. Data from the thresholding exercise and questionnaire were analyzed with descriptive statistics using R version 4.0.5 (The R Foundation for Statistical Computing, Vienna, Austria).
3.2 Results
3.2.1 Participant Characteristics
The sample consisted of 40 participants residing in the US: n = 20 AYP aged 16–23 years, and n = 20 PLG of adolescents aged 11–17 years. Generally, sample targets were met, except that a higher proportion of participants had completed a degree, lived in suburban locations and Southern US regions, and had received an MenB vaccine (Online Resource Table S7). AYP (n = 20) were, on average, 18.5 years (standard deviation [SD] 2.5) of age and most were female (n = 12, 60%). PLG (n = 20) were, on average, 46.5 years (SD 7.7) of age and most were male (n = 16, 80%). Most AYP reported having received multiple vaccines in the past, including the MenACWY vaccine (n = 13, 65%) and the MenB vaccine (n = 12, 60%), and most PLG reported that their children had received multiple vaccines, including the MenACWY vaccine (n = 15, 75%) and the MenB vaccine (n = 17, 85%). Three participants (8%) scored 25 points or higher on the Vaccine Hesitancy Scale, meeting the definition of being ‘vaccine hesitant’ [20] (Table 3).
3.2.2 Thematic Analysis
Four themes were derived from the qualitative data and are summarized in the conceptual map (Fig. 5). Example quotations from the qualitative interviews are provided in Online Resource Table S8.
Theme 1 was ‘attitudes towards vaccination’. Most participants (AYP: n = 16, 80%; PLG: n = 12, 60%) expressed positive attitudes towards vaccinations, reporting that vaccinations provided protection against diseases for themselves, their families, and communities. More PLG than AYP had negative (AYP: n = 1, 5%; PLG: n = 3, 15%), mixed (AYP: n = 1, 5%; PLG: n = 2, 10%), or unsure (AYP: n = 0; PLG: n = 2, 10%) attitudes towards vaccinations. Some participants (AYP: n = 3, 15%; PLG: n = 4, 20%) reported that the length of time a vaccine has been on the market would affect their decision to be vaccinated or for their child to be vaccinated, and some PLG (n = 3, 15%) were wary of ‘rushed’ vaccine developments and approvals, citing coronavirus disease 2019 (COVID-19) as an example. Two participants (AYP: n = 1, 5%; PLG: n = 1, 5%) expressed general mistrust in the pharmaceutical industry. Although most AYP reported following PLG guidance, some PLG (n = 3, 15%) said that their child’s unwillingness had contributed or would contribute to vaccination decisions.
Theme 2 was ‘knowledge and information’. Participants reported that they received information about vaccines and IMD from healthcare providers, clinics, the internet, media, schools, family, and friends. Some participants struggled to remember which vaccines they or their children had received (AYP: n = 4, 20%; PLG: n = 4, 20%), and some struggled to distinguish between MenB and MenACWY vaccines (PLG: n = 5, 25%), relying on healthcare providers or clinics to keep track. Government or doctor-specific recommendations about IMD vaccinations were perceived positively by most participants (AYP: n = 12, 60%; PLG: n = 15, 75%). However, a few participants (AYP: n = 1, 5%; PLG: n = 3, 15%) expressed mistrust in the recommendations. Four PLG (20%) knew someone who had died or had serious complications from meningitis—all four had positive outlooks on current recommendations.
Theme 3 was ‘perception of IMD’. Most participants (AYP: n = 12, 60%; PLG: n = 17, 85%) reported being very concerned about IMD after reading the vignette. Others (AYP: n = 7, 35%; PLG: n = 3, 15%) reported little or no concern; one AYP reported having no clear opinion. Key concerns about IMD included speed of onset, risk of mistaking it for flu, and contagiousness in school settings. One PLG thought that the IMD risk could be reduced through non-medical measures, such as increased hygiene. Most participants (AYP: n = 12, 60%; PLG: n = 16, 80%) described their perceived risk or their perceived child’s risk of developing meningitis as low.
Theme 4 was ‘vaccine attributes’. Concerns about adverse effects such as nausea, injection site reactions, headaches, and fever were reported barriers to IMD vaccination (AYP: n = 16, 80%; PLG: n = 14, 70%). Participants’ willingness to be vaccinated or willingness for their child to be vaccinated was influenced by vaccine efficacy (i.e., level of protection) (AYP: n = 4, 20%; PLG: n = 5, 25%) and number of doses (AYP: n = 6, 30%; PLG: n = 15, 75%). However, some participants (AYP: n = 11, 55%; PLG: n = 6, 30%) reported that number of doses did not matter if vaccines were effective with minimal adverse effects. Thirty-one participants (78%) would not accept more than three doses (AYP: n = 16, 80%; PLG: n = 15, 75%), while 9 (23%) would not accept more than two doses overall (AYP: n = 3, 15%; PLG: n = 6, 30%). Most participants were interested in the pentavalent (MenABCWY) vaccine (AYP: n = 18, 90%; PLG: n = 13, 65%) and most reported that they would choose a pentavalent (MenABCWY) vaccine over separate MenACWY and MenB vaccines (AYP: n = 14, 70%; PLG: n = 11, 55%). All themes directly informed the design of the DCE survey instrument and the statistical analysis plan, consistent with recommendations in the reporting guidelines [2].
3.2.3 Thresholding Exercises
Eighteen AYP (90%) and 15 PLG (75%) always chose the ‘vaccination’ option over the ‘no vaccination’ option, independent of IMD incidence rate, disability rate, or fatality rate. One AYP (5%) and two PLG (10%) always chose ‘no vaccination’. One AYP (5%) and three PLG (15%) varied their choices depending on incidence rate, disability rate, and fatality rate. Ninety-three percent of participants (n = 37) chose vaccination at least once.
4 Discrete Choice Experiment
The qualitative interviews informed the design of a DCE developed to quantify preferences for hypothetical IMD vaccination profiles (i.e., MenACWY + MenB or MenABCWY) versus no vaccination. The DCE quantified the trade-offs between drivers of vaccination preferences that individuals are willing to make and to predict the uptake of an MenABCWY vaccine. The vaccination attributes identified from the qualitative interviews (‘Vaccine attributes’ part of Fig. 5) were combined into three attribute categories: efficacy, adverse events, and number of doses.
Other qualitative interview results and the clinical data identified in the targeted literature review were also discussed. Cost and access were deemed out-of-scope for the DCE as they are individualized barriers to vaccination related to socioeconomic, insurance, or geographic status and are not fundamental attributes of the vaccines themselves. Furthermore, in the US, recommended vaccines during childhood are offered free of charge. Therefore, each hypothetical vaccination profile of the DCE will be defined by four attributes: level of protection, dosing, risk of mild-to-moderate adverse effects, and risk of severe adverse effects. Participants will be told to assume the hypothetical treatments are identical apart from the four considered attributes. The DCE will be iteratively pilot tested before being administered to study participants.
5 Adherence to Reporting Guidelines
We implemented the guidelines for reporting qualitative research that informs the design of quantitative preference elicitation instruments [2] (Online Resource Table S9). Twenty-one out of 22 items were fulfilled. ‘Researcher characteristics and reflexivity’, reporting community engagement used to facilitate study design or researcher characteristics that may influence the research, was the only item not fulfilled. Due to the professional expertise required, individuals from our targeted participant groups in the public (AYP or PLG) or patient advocates were not involved in the literature review. Giving representatives from these groups the opportunity to review the interview guide may have been beneficial, however our interviews were designed to be completed in three rounds. The interview guides were designed to be updated based on feedback from the previous rounds, ensuring participant influence on the relevance and readability of the questions. However, changes were ultimately not required, and the semi-structured interview guide did not change over the course of the study. Data on researcher characteristics were not collected due to employee privacy.
6 Discussion
The targeted literature review identified six topics that informed the qualitative interviews: general attitudes towards childhood/adolescent vaccination, prior knowledge of IMD, perceived risk, awareness of vaccines, willingness to be vaccinated, and recommendations. Qualitative analysis demonstrated that most participants were very concerned about IMD and had positive attitudes towards vaccinations. More AYP than PLG had positive attitudes towards vaccinations. Participants received information about vaccines and IMD from healthcare providers, clinics, the internet, media, schools, family, and friends. Recommendations from healthcare providers were generally perceived positively, although three participants expressed general mistrust in the pharmaceutical development process, being wary of ‘rushed’ products, or stating that ‘older’ drugs were ‘more trustworthy’. Willingness to be vaccinated was affected by several vaccine attributes, including dosing requirements, level of protection and efficacy, and adverse effects. More AYP than PLG would choose a new pentavalent vaccine over currently available vaccines. Efficacy and serogroup appeared to influence this choice. In the thresholding exercise, more than 80% of participants preferred a meningococcal vaccination over no vaccination, independent of a hypothetical incidence rate, disability rate, or fatality rate. More AYP than PLG chose vaccination over no vaccination at any incidence rate, disability rate, and fatality rate. These results align with a 2021 survey, where more than 80% of parents in six countries thought it was very important for children to be vaccinated against meningitis [21].
Generally, our findings are consistent with other recent qualitative studies on attitudes towards meningococcal vaccines and willingness to be vaccinated. For example, Richardson and colleagues also found that common barriers to vaccination were concerns about adverse effects and uncertainty about their child’s susceptibility to IMD [22], in agreement with our study findings.
However, there are some differences. For example, focus groups with US parents reported by Richardson and colleagues indicated that parents trusted physicians’ recommendations about MenACWY and MenB vaccines [22], whereas the current study found that trust in recommendations varied among participants. In another study using semi-structured interviews, parents in The Netherlands reported that they influenced their children’s decisions to accept or reject MenACWY vaccination; their children’s engagement with this decision was minimal [23]. By contrast, the current study found that adolescents’ unwillingness to be vaccinated influenced vaccination decisions even if parents were willing, but this was only in a minority of cases. The COVID-19 pandemic has influenced vaccine attitudes [24], which may explain some differences in our findings compared with findings from earlier studies.
By gathering qualitative data, a rich picture of themes was created relating to attitudes towards IMD and willingness to be vaccinated, which have implications for vaccine development and vaccination strategies. The observed preference for pentavalent vaccines over separate monovalent and quadrivalent vaccines may be explained by people’s preferences for fewer doses. Efficacy and serogroup coverage appeared to influence some participants’ preferences for a new pentavalent vaccine over currently available vaccines, but further research is needed to understand the relationships among these attributes. Our findings highlight differences between AYP’s and PLG’ attitudes towards IMD and willingness to be vaccinated, which suggests that public health initiatives may need to tailor their content for these different audiences to increase vaccine uptake.
In conducting this qualitative research, 21 out of 22 items from the reporting guidelines were fulfilled [2] and we systematically reported the process that informed the design of our DCE. The ‘researcher characteristics and reflexivity’ item from the reporting guidelines was not fulfilled. This item could have been fully fulfilled if the interview guide had been reviewed in advance by members from our participant groups; however, the study team took steps to ensure that the interview guide was relevant to people in the general population who may be eligible to make vaccination decisions for themselves or for their children. Community engagement may be more crucial in patient preference studies to identify specific patient experiences, rather than in studies where participants are members of the general public. Although previous studies have used qualitative research to design preference elicitation instruments, this process is often underreported [1]. We hope that this study can serve as an example of how to design a DCE based on qualitative research and how to sufficiently report this methodological process. The key element of reporting such studies is transparency.
This study has several limitations. Participants were residing in the US only, therefore findings may not be transferrable to other countries. However, we recruited a diverse sample based on different target characteristics to capture different perspectives and experiences. Differences in perspectives between AYP and PLG were explored, but given the small sample, no firm conclusions can be drawn about any differences. Only 8% of the sample were classified as vaccine-hesitant. Although 73% of the sample reported receiving an MenB vaccine, this may have been overreported given the general uptake of the MenB vaccine in the US [9]. Attitudes towards IMD vaccination may be more negative in samples with higher vaccine hesitancy and less IMD vaccination experience. Future qualitative studies should examine the views of AYP and PLG in other countries and in populations with lower meningococcal vaccine uptake, and should use patient and public involvement activities to facilitate study design, since researcher characteristics can influence the research. All clinical data were self-reported and subject to recall bias. For example, several participants struggled to remember which vaccines they or their children had received, limiting the reliability of these data.
Targeted literature reviews and qualitative research are vital for identifying attributes and levels for quantitative patient preference elicitation instruments. Our targeted literature review revealed that no previous DCEs had investigated young people’s and parents’ preferences for IMD vaccination in the US, confirming the need to address this research gap. Implementing small-sample or individual-level preference methods such as the thresholding exercise and best-worst scaling may help researchers to quantitatively select attributes and levels for a DCE, and test whether patients are willing to make trade-offs between attributes of vaccination options [17, 25]. Testing trade-offs is important for understanding participant preferences.
7 Conclusions
Most participants were concerned about IMD and had positive views of vaccines and vaccination recommendations. Furthermore, most participants would choose a pentavalent vaccine over a combination of currently available monovalent and quadrivalent vaccines. Willingness to be vaccinated was affected by a vaccine’s level of protection and other vaccination attributes, but largely insensitive to IMD incidence and likelihood of disability or death. These findings highlight the need to design vaccination strategies that account for the views of parents and adolescents, to maximize vaccine uptake. This study serves as an example of how to design a DCE based on qualitative research and how to adequately report this methodological process.
Availability of data and material
The informed consent obtained from participants does not allow for individual data to be made publicly available.
Code availability
Not applicable.
References
Vass C, Rigby D, Payne K. The role of qualitative research methods in discrete choice experiments. Med Decis Making. 2017;37(3):298–313.
Hollin IL, Craig BM, Coast J, Beusterien K, Vass C, DiSantostefano R, Peay H. Reporting formative qualitative research to support the development of quantitative preference study protocols and corresponding survey instruments: guidelines for authors and reviewers. Patient. 2020;13(1):121–36.
Tong A, Sainsbury P, Craig J. Consolidated criteria for reporting qualitative research (COREQ): a 32-item checklist for interviews and focus groups. Int J Qual Health Care. 2007;19(6):349–57.
Tong A, Flemming K, McInnes E, Oliver S, Craig J. Enhancing transparency in reporting the synthesis of qualitative research: ENTREQ. BMC Med Res Methodol. 2012;12:181.
de Jong Y, van der Willik EM, Milders J, Voorend CGN, Morton RL, Dekker FW, et al. A meta-review demonstrates improved reporting quality of qualitative reviews following the publication of COREQ- and ENTREQ-checklists, regardless of modest uptake. BMC Med Res Methodol. 2021;21(1):184.
O’Brien BC, Harris IB, Beckman TJ, Reed DA, Cook DA. Standards for reporting qualitative research: a synthesis of recommendations. Acad Med. 2014;89(9):1245–51.
Helter TM, Boehler CE. Developing attributes for discrete choice experiments in health: a systematic literature review and case study of alcohol misuse interventions. J Subst Use. 2016;21(6):662–8.
de Bekker-Grob EW, Swait JD, Kassahun HT, Bliemer MCJ, Jonker MF, Veldwijk J, et al. Are healthcare choices predictable? The impact of discrete choice experiment designs and models. Value Health. 2019;22(9):1050–62.
Pingali C, Yankey D, Elam-Evans LD, Markowitz LE, Valier MR, Fredua B, et al. Vaccination coverage among adolescents aged 13–17 Years—National Immunization Survey-Teen, US, 2022. Morb Mortal Wkly Rep. 2023;72(34):912–9.
Mitchell H, Lim R, Gill PK, Dhanoa J, Dube E, Bettinger JA. What do adolescents think about vaccines? Systematic review of qualitative studies. PLOS Glob Public Health. 2022;2(9): e0001109.
Painter JE, De Viana OMS, Jimenez L, Avila AA, Sutter CJ, Sutter R. Vaccine-related attitudes and decision-making among uninsured, Latin American immigrant mothers of adolescent daughters: a qualitative study. Hum Vaccin Immunother. 2019;15(1):121–33.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Syst Rev. 2021;10(1):89.
US Food and Drug Administration. Package leaflet: TRUMENBA (Meningococcal Group B Vaccine). Pfizer. 2021.
Patrick DL, Burke LB, Gwaltney CJ, Leidy NK, Martin ML, Molsen E, Ring L. Content validity–establishing and reporting the evidence in newly developed patient-reported outcomes (PRO) instruments for medical product evaluation: ISPOR PRO Good Research Practices Task Force report: part 2–assessing respondent understanding. Value Health. 2011;14(8):978–88.
Turner-Bowker DM, Lamoureux RE, Stokes J, Litcher-Kelly L, Galipeau N, Yaworsky A, et al. Informing a priori sample size estimation in qualitative concept elicitation interview studies for clinical outcome assessment instrument development. Value Health. 2018;21(7):839–42.
Shapiro GK, Tatar O, Dube E, Amsel R, Knauper B, Naz A, et al. The vaccine hesitancy scale: psychometric properties and validation. Vaccine. 2018;36(5):660–7.
Hauber B, Coulter J. Using the threshold technique to elicit patient preferences: an introduction to the method and an overview of existing empirical applications. Appl Health Econ Health Policy. 2020;18(1):31–46.
Patton MQ. Qualitative research and evaluation methods: Integrating theory and practice. 4th ed. Sage Publications. 2014.
Ikenwilo D, Heidenreich S, Ryan M, Mankowski C, Nazir J, Watson V. The best of both worlds: an example mixed methods approach to understand men’s preferences for the treatment of lower urinary tract symptoms. Patient. 2018;11(1):55–67.
Akel KB, Masters NB, Shih SF, Lu Y, Wagner AL. Modification of a vaccine hesitancy scale for use in adult vaccinations in the United States and China. Hum Vaccin Immunother. 2021;17(8):2639–46.
Tan LLJ, Safadi MAP, Horn M, Regojo Balboa C, Moya E, Schanbaum J, et al. Pandemic’s influence on parents’ attitudes and behaviors toward meningococcal vaccination. Hum Vaccin Immunother. 2023;19(1):2179840.
Richardson E, Ryan KA, Lawrence RM, Harle CA, Young A, Livingston MD, et al. Perceptions and knowledge about the MenB vaccine among parents of high school students. J Community Health. 2021;46(4):808–16.
Oostdijk C, Van Zoonen K, Ruijs WLM, Mollema L. Household decision-making for the MenACWY vaccination: how parents and adolescents deal with an adolescent vaccination decision. Vaccine. 2021;39(31):4283–90.
Altman JD, Miner DS, Lee AA, Asay AE, Nielson BU, Rose AM, et al. Factors affecting vaccine attitudes influenced by the COVID-19 pandemic. Vacc (Basel). 2023;11(3):516.
Cheung KL, Wijnen BF, Hollin IL, Janssen EM, Bridges JF, Evers SM, Hiligsmann M. Using best-worst scaling to investigate preferences in health care. Pharmacoeconomics. 2016;34(12):1195–209.
Bishai D, Brice R, Girod I, Saleh A, Ehreth J. Conjoint analysis of French and German parents’ willingness to pay for meningococcal vaccine. Pharmacoeconomics. 2007;25(2):143–54.
Marshall HS, Chen G, Clarke M, Ratcliffe J. Adolescent, parent and societal preferences and willingness to pay for meningococcal B vaccine: a discrete choice experiment. Vaccine. 2016;34(5):671–7.
Poulos C, Reed Johnson F, Krishnarajah G, Anonychuk A, Misurski D. Pediatricians’ preferences for infant meningococcal vaccination. Value Health. 2015;18(1):67–77.
Seanehia J, Treibich C, Holmberg C, Muller-Nordhorn J, Casin V, Raude J, Mueller JE. Quantifying population preferences around vaccination against severe but rare diseases: a conjoint analysis among French university students, 2016. Vaccine. 2017;35(20):2676–84.
Basta NE, Becker AB, Li Q, Nederhoff D. Parental awareness of Meningococcal B vaccines and willingness to vaccinate their teens. Vaccine. 2019;37(4):670–6.
Bedford H, Lansley M. More vaccines for children? Parents’ views. Vaccine. 2007;25(45):7818–23.
Clark SJ, Cowan AE, Stokley S, Bilukha O, Davis MM. Physician perspectives to inform a new recommendation for meningococcal conjugate vaccine (MCV4). J Adolesc Health. 2006;39(6):850–5.
Dube E, Gagnon D, Hamel D, Belley S, Gagne H, Boulianne N, et al. Parents’ and adolescents’ willingness to be vaccinated against serogroup B meningococcal disease during a mass vaccination in Saguenay-Lac-St-Jean (Quebec). Can J Infect Dis Med Microbiol. 2015;26(3):163–7.
Gargano LM, Weiss P, Underwood NL, Seib K, Sales JM, Vogt TM, et al. School-located vaccination clinics for adolescents: correlates of acceptance among parents. J Community Health. 2015;40(4):660–9.
Le Ngoc TS, Ader F, Ferry T, Floret D, Arnal M, Fargeas S, et al. Vaccination against serogroup B Neisseria meningitidis: perceptions and attitudes of parents. Vaccine. 2015;33(30):3463–70.
Mameli C, Faccini M, Mazzali C, Picca M, Colella G, Duca PG, Zuccotti GV. Acceptability of meningococcal serogroup B vaccine among parents and health care workers in Italy: a survey. Hum Vaccin Immunother. 2014;10(10):3004–10.
Marshall H, Clarke M, Sullivan T. Parental and community acceptance of the benefits and risks associated with meningococcal B vaccines. Vaccine. 2014;32(3):338–44.
Pelullo CP, Napolitano F, Di Giuseppe G. Meningococcal disease and vaccination: knowledge and acceptability among adolescents in Italy. Hum Vaccin Immunother. 2018;14(5):1197–202.
Ponticelli D, D’Ambrosio A, Cancellieri M, Agozzino E. Do HCWs adequately know about meningitis and 4CMenB vaccine and recommend its use to parents? A cross sectional analysis in Campania Region, Italy. J Prev Med Hyg. 2019;60(2):E147–57.
Roberts JR, Thompson D, Rogacki B, Hale JJ, Jacobson RM, Opel DJ, Darden PM. Vaccine hesitancy among parents of adolescents and its association with vaccine uptake. Vaccine. 2015;33(14):1748–55.
Srivastava A, Dempsey A, Galitsky A, Fahimi M, Huang L. Parental awareness and utilization of meningococcal serogroup B vaccines in the United States. BMC Public Health. 2020;20(1):1109.
Takla A, Wichmann O, Koch J, Terhardt M, Hellenbrand W. Survey of pediatricians in Germany reveals important challenges for possible implementation of meningococcal B vaccination. Vaccine. 2014;32(48):6349–55.
van Lier A, Ferreira JA, Mollema L, Sanders EAM, de Melker HE. Intention to vaccinate universally against varicella, rotavirus gastroenteritis, meningococcal B disease and seasonal influenza among parents in the Netherlands: an internet survey. BMC Res Notes. 2017;10(1):672.
Verger P, Fressard L, Collange F, Gautier A, Jestin C, Launay O, et al. Vaccine hesitancy among general practitioners and its determinants during controversies: a national cross-sectional survey in France. EBioMedicine. 2015;2(8):891–7.
Acknowledgments
Medical writing was provided by Maddy L. Dyer, PhD (Evidera) and Stephen Gilliver, PhD (Evidera) and funded by Pfizer. Recruitment and screening were provided by Global Perspectives, and Evidera staff (Chiara Whichello, Christine Michaels-Igbokwe, Malavika Venkatraman, Vicky Turner, Rodolfo Matos, Mary-Claire Miller, and Deana Trimble) completed the interviews.
Funding
This study was conducted by Evidera under the direction of Pfizer and was funded by Pfizer. Editorial and graphics services were provided by Evidera.
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of the research. CW, CMI, and MV conducted the targeted literature review and the qualitative interviews, and acquired the data. CW, CMI, MV, and SH analyzed the data, and all authors interpreted the data. All authors approved the version to be published and agree to be accountable for all aspects of the research.
Corresponding author
Ethics declarations
Conflicts of interest
Joshua Coulter, Brett Hauber, Joseph C. Cappelleri, Paula Peyrani, Jessica Vespa Presa and Katharina Schley are employees and may hold stocks or stock options in Pfizer Inc.. Chiara Whichello, Sebastian Heidenreich, Christine Michaels-Igbokwe, and Malavika Venkatraman are employees of Evidera, which was paid by Pfizer for the development of this manuscript.
Ethics approval
The study was conducted in accordance with accepted research practices described in Good Practices for Outcomes Research, issued by the International Society for Pharmacoeconomics and Outcomes Research. Compliance with regulatory standards assured that the rights, safety, and well-being of participants were protected and that the study data were credible and responsibly reported. This study was designed, implemented, and reported in accordance with US ethical guidelines and with the ethical principles laid down in the Declaration of Helsinki. The study protocol was submitted to Ethical and Independent Review Services, a fully accredited institutional review board, for central ethical approval. Institutional Review Board approval was granted on 29 March 2022 (reference: 22049-01).
Consent to participate
Participants provided oral and digitally collected written consent.
Consent for publication
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Coulter, J., Whichello, C., Heidenreich, S. et al. From Qualitative Research to Quantitative Preference Elicitation: An Example in Invasive Meningococcal Disease. Patient 17, 319–333 (2024). https://doi.org/10.1007/s40271-024-00677-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40271-024-00677-8