Abstract
Introduction
Evidence regarding the factors affecting the geographical variation of antimicrobial use (AMU) is relatively scarce. This study aimed to evaluate factors potentially associated with geographical variability of AMU per day per 1000 habitants in the 47 prefectures of Japan.
Methods
This is an observational ecological study using the Japanese national database in 2019. The outcome was the defined daily doses per 1000 inhabitants per day by prefecture. Multivariable negative binomial regression analysis was conducted using patient- and physician-level variables.
Results
The study included 605,391,054 defined daily doses of AMU in 2019 from the 47 prefectures. In the multivariable negative binomial regression analyses for the outcome of total AMU, the proportion of female individuals (adjusted rate ratio [aRR] 1.04 [1.01–1.08] per 1% increase, p = 0.021), the proportion of upper secondary graduates going to further education (aRR 1.01 [1.00–1.01] per 1% increase, p = 0.005), and the annual number of diagnoses related to upper respiratory infections (URIs) per 1000 inhabitants per day (aRR 1.21 [1.10–1.34], p < 0.001) were significantly correlated with total AMU.
Conclusions
In this ecological study, the variability of total AMU by Japanese prefecture was associated with the proportion of female individuals, education level, and the number of URI diagnoses per population. The results suggest the potential need for additional stewardship efforts to reduce unnecessary antimicrobial prescriptions for URI.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Factors affecting the geographical variation of antimicrobial use (AMU) are not clear. | |
This observational ecological study used the Japanese national database in 2019. | |
The number of URI diagnoses per population was strongly associated with AMU. | |
This suggests the potential need for additional antimicrobial stewardship efforts. |
Introduction
Antimicrobial resistance (AMR) has caused significant global morbidity and mortality [1]. A larger volume of antimicrobial use (AMU) was related to the higher rate of AMR in some ecological studies [2, 3]. Many countries have established their AMR national action plans [4]. Japan’s AMR national action plan was established in 2016, aiming to reduce AMU per 1000 inhabitants per day in 2020 to two-thirds of that in 2013 [5, 6]. There has been major progress, including the achievement of the goal to reduce AMU in 2020, although the COVID-19 pandemic may have had a significant impact on the reduction of AMU [7]. However, because there are still needs to further mitigate the negative impact of AMR, the investigation of potential factors associated with AMU is important to further guide future AMR-related measures.
While geographical variabilities of AMU within a country were reported in some studies, evidence regarding the factors affecting the geographical variation of AMU is relatively scarce [8,9,10,11,12,13]. In general, physician-level factors (e.g., physician’s experiences) and patient-level factors (e.g., patient age, sex, and socioeconomic status) have been reported to impact physician’s behavior regarding antimicrobial prescription [13,14,15,16,17,18]. The investigation of these factors to evaluate their potential impact on geographical variability using nationwide data may contribute to the identification of potential measures to reduce AMU and the promotion of appropriate AMU in the future. A previous study using AMU data until the year of the introduction of the AMR national action plan in Japan (2013–2016) showed that oral macrolides and quinolones are more frequently used in Western Japan compared to Eastern Japan (East Japan includes Tokyo, Kanto region (The Greater Tokyo Area), and Northern Japan, whereas West Japan includes Kansai region, the second most populated area in Japan, and Southern Japan) [11, 12]. However, factors associated with the geographical variation of AMU remain to be investigated. Given the significant reduction in AMU after the implementation of the national action plan since 2016, our study focused on the assessment of factors contributing to high regional AMU using the data after the implementation of the national action plan to elucidate potential interventions in addition to the action plan implementation.
In this ecological study, we aimed to evaluate factors potentially associated with the geographical variability of AMU per day per 1000 habitants in all 47 prefectures of Japan.
Methods
Ethical Approval
The study was conducted in accordance with the declaration of Helsinki and approved by the Institutional Review Board at National Center for Global Health and Medicine (Approval No. NCGM-G-003098-01). Patient identifiers were not included in our data used in this study and therefore no informed consent was required to conduct the present study.
Study Design
This was an observational ecological study using the Japanese national database (NDB) in 2019. All methods were carried out in accordance with the STROBE statement. The NDB is a database of anonymized electronic health insurance claims covering more than 99% of all national claims with data regarding medical outpatient and inpatient services, diagnostic procedural combinations, dental services, and dispensed medications [19, 20]. Our proposal document for the use of the NDB was reviewed by the Japanese Ministry of Health, Labour and Welfare (MHLW), which granted approval for its use. The outcome of this study was the defined daily doses (DDD) per 1000 inhabitants per day (DID). DDD was calculated according to the World Health Organization (WHO) definition in 2022 [21]. Population referred to that on October 1 in the study year [22]. Using dependent variables as described below, a multivariable negative binomial regression analysis was conducted to evaluate the impact of each variable on the outcome.
Outcome
All systemic antimicrobials, defined as J01 in the Anatomical Therapeutic Chemical (ATC) classification, were included in the study [23]. AMU data from January 1 to December 31, 2019 were extracted for all 47 prefectures. The location of prescribing healthcare facilities was used to identify the prefecture of each prescription. Then, the DID was calculated by prefecture. AMU data extracted from the NDB was categorized using ATC classification [23].
Variables
We hypothesized that antimicrobial stewardship measures are associated with reduced AMU. For example, the national action plan includes the enhancement of education related to AMR as well as the development of infectious disease specialists. Therefore, the average age of physicians (assuming young physicians receive more AMR-related education) and the number of infectious disease physicians are selected as variables. In Japan, physicians are the only practitioners who can prescribe antimicrobials. In hospital settings, antimicrobial stewardship teams often comprise physicians (e.g., infectious disease physicians), pharmacists (e.g., board certified pharmacists in infection control), nurses (e.g., certified nurses in infection control), and clinical laboratory technicians (e.g., infection control microbiological technologists) that support and advise physicians on antimicrobial prescriptions. In community settings, community pharmacists assist and advise physicians on antimicrobial prescribing.
As the action plan also focuses on the reduction of AMU for upper respiratory infections (URIs), the number of URIs was also selected as a variable in this study. In addition, the action plan encourages center hospitals to take initiative in their local stewardship efforts (additional reimbursement for infection prevention in Supplemental Table S1). The adjustment was conducted with population factors (population age, sex, nationality, income, and education level) and other healthcare facility factors (the number of clinics, the number of hospitals, and the proportion of large hospitals).
On the basis of the hypotheses described above, the variables evaluated in this study included population sex (the proportion of female individuals), population age (the proportion of population < 15 years and ≥ 65 years), the average income per household, the population education level (the proportion of upper secondary graduates going to further education), the number of clinics per 10,000 inhabitants, the number of hospitals per 100,000 inhabitants, the proportion of hospitals with ≥ 500 beds, the average age of physicians, the number of certified infectious disease specialist physicians per 100,000 inhabitants, the number of hospitals with additional reimbursement for infection prevention 1 per 100,000 inhabitants, the number of hospitals with additional reimbursement for infection prevention 2 per 100,000 inhabitants, and the annual number of diagnoses related to URI per 1000 inhabitants per day. In this study, all variables were presented at the prefecture level.
Population age and sex, the average income per household, the proportion of upper secondary graduates going to further education, and the average age of physicians were obtained from the Statistic Bureau, Ministry of Internal Affairs and Communications [24]. The physician’s age was included in the variables because some studies showed physicians’ experience is related to antibiotic prescriptions [13, 17]. The number of clinics, the number of hospitals, the proportion of hospitals with > 500 beds, and the number of healthcare facilities with additional reimbursement for infection prevention 1 and 2 were obtained from regional bureaus of Health and Welfare (e.g., Kanto-Shinetsu) [25, 26]. The requirements for hospitals with additional reimbursement for infection prevention in 2019 are presented in Supplemental Table S1 [27]. The number of certified infectious disease specialist physicians was obtained from the Japanese Association for Infectious Diseases [28]. The annual number of diagnoses related to URIs was extracted from the NDB. The URI-related diagnoses included in this study are presented in Table S2.
Statistical Analysis
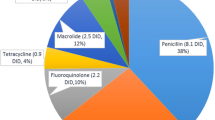
Characteristics of variables were presented with median and interquartile range (IQR). To investigate the relationship between the outcome of DID and each variable, multivariable negative binomial regression analyses were performed to calculate adjusted rate ratios (aRR) with 95% confidence intervals. The aRRs of proportional variables were presented for a 1% increase in each variable. The negative binomial regression model was selected because of the significant overdispersion in the multivariable Poisson regression model and because of DDD being the count data. To avoid multicollinearity, variance inflation factors (VIFs) in all variables were evaluated. After discussion amongst the authors, the proportion of ≥ 65 years, the average income per household, and the number of hospitals per 10,000 inhabitants, factors with high VIF, were excluded from the multivariable analysis. The log of the population in the prefecture was used as an offset term in the negative binomial regression analyses. The analyses were implemented for all systemic AMU, and AMU by class (1, J01C penicillins; 2, J01D other beta-lactams; 3, J01M quinolones; 4, J01F macrolides, lincosamides, and streptogramins; 5, J01 all other systemic antimicrobials).
Two-sided p values less than 0.05 were considered to show statistical significance. The statistical analyses described above were conducted using Stata 14.2 (College Station, TX, USA).
Results
The study included 605,391,054 defined daily doses of AMU in 2019 from the 47 prefectures. The characteristics of the prefecture-level AMU and variables are presented in Table 1. The AMUs in the 47 prefectures varied with a median of 13.1 DID (IQR 12.0–14.3). Figure 1 shows the geographical variation of AMU (DID) for all systemic antimicrobials. The trend of higher AMU in Western Japan was observed compared with Eastern Japan.
In the multivariable negative binomial regression analyses for the outcome of total AMU, the proportion of female individuals (aRR 1.04 [1.01–1.08] per 1% increase, p = 0.021), the proportion of upper secondary graduates going to further education (aRR 1.01 [1.00–1.01] per 1% increase, p = 0.005), and the annual number of diagnoses related to URIs per 1000 inhabitants per day (aRR 1.21 [1.10–1.34], p < 0.001) were significantly associated with total AMU (Fig. 2). Other variables were not significantly associated with total AMU.
Adjusted rate ratios of prefecture-level total antimicrobial use (DID) in 2019 by investigated variables. The dots and lines represent the point estimates and 95% confidence intervals, respectively. The adjusted rate ratios of proportional variables are presented for a 1% increase in each variable. The variable labels are defined as follows. Female, the proportion of female individuals in the population; < 15 years, the proportion of people aged < 15 years in the population; Education, the proportion of upper secondary graduates going to further education; Clinic, the number of clinics per 1000 inhabitants; ≥ 500 beds, the proportion of hospitals with > 500 beds; Reimbursement 1, the number of hospitals with additional reimbursement for infection prevention 1 per 100,000 inhabitants; Reimbursement 2, the number of hospitals with additional reimbursement for infection prevention 2 per 100,000 inhabitants; ID physician, the number of certified infectious disease specialist physicians per 1000 inhabitants; Physician age, the average age of physicians; URI, the annual number of diagnoses related to upper respiratory infections per inhabitant
The results of multivariable negative binomial regression analyses by antimicrobial class are presented in Supplemental Figs. S1–S5. For the outcome of AMU in penicillins, the proportion of < 15 years (aRR 1.10 [1.05–1.14] per 1% increase, p < 0.001) had a positive correlation, while the average age of physicians (aRR 0.93 [0.90–0.97], p = 0.001) had a significant negative correlation (Fig. S1). In the AMU of other beta-lactam antimicrobials, the proportion of female individuals (aRR 1.04 [1.00–1.08], p = 0.027), the proportion of upper secondary graduates going to further education (aRR 1.01 [1.00–1.02], p < 0.001), and the annual number of diagnoses related to URIs per 1000 inhabitants per day (aRR 1.19 [1.07–1.31], p = 0.001) were significantly correlated (Fig. S2). These three variables were also statistically significant in quinolones (Fig. S3), with aRR 1.07 [1.01–1.013] and p = 0.030 for the female proportion, aRR 1.01 [1.00–1.02] and p = 0.012 for the proportion of upper secondary graduates going to further education, and aRR 1.48 [1.26–1.74] and p < 0.001 for the annual number of diagnoses related to URIs per 1000 inhabitants per day. The number of hospitals with additional reimbursement for infection prevention 1 per 100,000 inhabitants was positively correlated with the AMU of macrolides, lincosamides, and streptogramins (aRR 4.32 [1.87–10.01], p = 0.001 in Fig. S4) and all other systemic antimicrobials (aRR 4.56 [1.16–17.85], p = 0.029 in Fig. S5).
Discussion
The study evaluated the relationship between AMU and patient- and physician-level variables in 47 Japanese prefectures. The proportion of female individuals, education level, and the number of URI diagnoses were positively correlated with the total AMU as well as the AMU beta-lactams other than penicillins and quinolones in the analysis by antimicrobial class. On the other hand, the use of penicillins was positively correlated with the proportion of the pediatric population and negatively correlated with physicians’ age. Compared with other countries, the median 13.1 DID in our study was in lower than those of most European countries, but was still higher than the lowest countries [29].
Our study resulted in a higher AMU in Western Japan than that in Eastern Japan, which was described in the previous study using data before the action plan implementation. For the AMU in general, previous studies, including a systematic review, revealed that female individuals were more likely to receive antimicrobials compared with male individuals [30,31,32,33], although other studies showed that male individuals had more antibiotic prescriptions than female individuals [34, 35]. The study could not evaluate the impact of urinary tract infection, which was more frequently reported in female individuals compared to male individuals [36]. While the impact of gender on the AMU varies by situation, further investigations, including the impact of urinary tract infection [37], regarding the factors related to gender differences in the population are warranted. Our study suggests that higher education levels may be correlated with more AMU. Previous studies outside of Japan showed that lower education was correlated with more AMU or misuse of antimicrobials [38, 39]. However, patient pressure to prescribe antibiotics is a recognized issue in inappropriate antimicrobial prescriptions [40, 41]. A potential hypothesis explaining our result is that higher patient education is correlated with high demand for antimicrobial prescriptions in Japan. Additional studies with a different study design from ours are important to evaluate the impact of overall education level on the Japanese population.
While the vast majority of URIs are caused by viruses, inappropriate antimicrobial prescriptions are frequently reported by many studies [42,43,44,45]. A previous Japanese study revealed that antimicrobials were prescribed in 60% of cases with claim diagnoses of non-bacterial URIs [42]. To reduce unnecessary AMU for URIs in Japan, the national guideline was developed in 2017. However, a recent study after the dissemination of the guideline showed that AMU for URIs was still common [46]. Our study also showed a positive correlation between the number of visits with URI diagnoses and AMU, suggesting the need for further efforts, including the enhancement of education regarding appropriate AMU targeting the general population, to reduce antibiotic prescriptions for URI [47]. In addition, it is important to note that most URIs are diagnosed and managed in outpatient settings. Stewardship measures specifically focusing on these outpatient settings are required to reduce antibiotic prescriptions for URI.
Regarding AMU by antibiotic class, beta-lactams other than penicillins and quinolones comprise approximately half of total AMU. Given that the three factors associated with the AMU of these two antibiotic classes are the same as those of total AMU, the same rationales of the associations with the total AMU may be applied to these two antibiotic classes. Stewardship measures to reduce unnecessary prescriptions of these two antibiotic classes are vital to further reduce total AMU in the future. On the other hand, AMU in penicillins was correlated with a larger proportion of the pediatric population and younger physician age. As recommended by a Japanese guideline, pediatric patients may often receive penicillins for certain types of respiratory infections [48]. Because our study used DID as the outcome, AMU for children may have been underestimated given some studies from other countries reported more frequent antimicrobial prescriptions for children than those for adults [49]. Therefore, the lack of significant correlation between the pediatric population and DID in overall AMU and AMU in other classes may have not been applied if the number of antimicrobial prescriptions had been selected as our outcome. In general, the spectrums of penicillins in the ATC classification are narrower than those of other beta-lactams with amoxicillin index used as an indicator of quality measure regarding AMU [50]. Some studies revealed that physicians with a long career duration were more likely to prescribe antimicrobials than younger physicians [17, 44].
The number of hospitals with additional reimbursement for infection prevention 1 per population was positively correlated with the larger AMUs in macrolides, lincosamides, streptogramins, and other systemic antimicrobials. While these hospitals may be more likely to see complex patients requiring the antimicrobial treatments of these classes, the reason for the result is currently being investigated.
There are some limitations in the study. First, because this is an ecological study, the possibility of an ecological fallacy cannot be eliminated. Other factors which were not investigated in this study may have affected the AMU. Second, the study used the data before the COVID-19 pandemic. The exploration of the impact of the COVID-19 pandemic on the factors of antibiotic prescription is an interesting future research topic. Third, the study findings from Japan may not be generalized to other countries. However, our result that the number of URI diagnoses was significantly correlated with higher AMU after adjusting population and healthcare-related factors may provide a global insight into the need to emphasize antimicrobial stewardship for URIs. While the results need to be interpreted with caution given these limitations, our results generated hypotheses to address the geographical variability of AMU in Japan.
Conclusions
In this ecological study, the variability of total AMU by Japanese prefecture was associated with the proportion of female individuals, education level, and the number of URI diagnoses per population. The results suggest the potential need for additional stewardship efforts to reduce unnecessary antimicrobial prescriptions for URI. The analysis by antimicrobial class showed that the younger age of physicians was correlated with the AMU of penicillins, suggesting that the impact of stewardship education for young physicians.
Data Availability
Data from this study cannot be shared. Data are available from the Ministry of Health, Labour, and Welfare of Japan for researchers who meet the criteria for access to confidential data. Please contact the corresponding author for further information.
References
Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022;399(10325):629–55. https://doi.org/10.1016/S0140-6736(21)02724-0.
Goossens H, Ferech M, Vander Stichele R, Elseviers M, ESAC Project Group. Outpatient AMU in Europe and association with resistance: a cross-national database study. Lancet. 2005;365(9459):579–87. https://doi.org/10.1016/S0140-6736(05)17907-0.
Pouwels KB, Chatterjee A, Cooper BS, Robotham JV. Antimicrobial resistance, stewardship, and consumption. Lancet Planet Health. 2019;3(2):e66. https://doi.org/10.1016/S2542-5196(18)30283-3.
Willemsen A, Reid S, Assefa Y. A review of national action plans on antimicrobial resistance: strengths and weaknesses. Antimicrob Resist Infect Control. 2022;11(1):90. https://doi.org/10.1186/s13756-022-01130-x.
The Government of Japan. National Action Plan on Antimicrobial Resistance (AMR) 2016–2020. April 5, 2016. https://www.mhlw.go.jp/file/06-Seisakujouhou-10900000-Kenkoukyoku/0000138942.pdf. Accessed 2 Aug 2022.
Ohmagari N. National action plan on antimicrobial resistance (AMR) 2016–2020 and relevant activities in Japan. Glob Health Med. 2019;1(2):71–7. https://doi.org/10.35772/ghm.2019.01017.
Gu Y, Fujitomo Y, Ohmagari N. Outcomes and future prospect of Japan’s national action plan on antimicrobial resistance (2016–2020). Antimicrobials (Basel). 2021;10(11):1293. https://doi.org/10.3390/antimicrobials10111293.
Kristensen PK, Johnsen SP, Thomsen RW. Decreasing trends, and geographical variation in outpatient AMU: a population-based study in Central Denmark. BMC Infect Dis. 2019;19(1):337. https://doi.org/10.1186/s12879-019-3964-9.
Zhang Y, Steinman MA, Kaplan CM. Geographic variation in outpatient antimicrobial prescribing among older adults. Arch Intern Med. 2012;172(19):1465–71. https://doi.org/10.1001/archinternmed.2012.3717.
Roberts RM, Hicks LA, Bartoces M. Variation in US outpatient antimicrobial prescribing quality measures according to health plan and geography. Am J Manag Care. 2016;22(8):519–23.
Kusama Y, Ishikane M, Tanaka C, et al. Regional variation of antimicrobial use in Japan from 2013–2016, as estimated by the sales data. Jpn J Infect Dis. 2019;72(5):326–9. https://doi.org/10.7883/yoken.JJID.2018.417.
Gahbauer AM, Gonzales ML, Guglielmo BJ. Patterns of antibacterial use and impact of age, race/ethnicity, and geographic region on antibacterial use in an outpatient Medicaid cohort. Pharmacotherapy. 2014;34(7):677–85. https://doi.org/10.1002/phar.1425.
Di Martino M, Lallo A, Kirchmayer U, Davoli M, Fusco D. Prevalence of antimicrobial prescription in pediatric outpatients in Italy: the role of local health districts and primary care physicians in determining variation. A multilevel design for healthcare decision support. BMC Public Health. 2017;17(1):886. https://doi.org/10.1186/s12889-017-4905-4.
McGurn A, Watchmaker B, Adam K, et al. Socioeconomic status and determinants of pediatric AMU. Clin Pediatr (Phila). 2021;60(1):32–41. https://doi.org/10.1177/0009922820941629.
Russo V, Monetti VM, Guerriero F, et al. Prevalence of antimicrobial prescription in southern Italian outpatients: real-world data analysis of socioeconomic and sociodemographic variables at a municipality level. Clinicoecon Outcomes Res. 2018;10:251–8. https://doi.org/10.2147/CEOR.S161299.
Depew RE, Gonzales G. Differences in US antimicrobial prescription use by facility and patient characteristics: evidence from the national ambulatory medical care survey. Fam Pract. 2020;37(2):180–6. https://doi.org/10.1093/fampra/cmz049.
Fernandez-Lazaro CI, Brown KA, Langford BJ, Daneman N, Garber G, Schwartz KL. Late-career physicians prescribe longer courses of antimicrobials. Clin Infect Dis. 2019;69(9):1467–75. https://doi.org/10.1093/cid/ciy1130.
Saliba-Gustafsson EA, Dunberger Hampton A, Zarb P, Orsini N, Borg MA, Stålsby LC. Factors associated with antimicrobial prescribing in patients with acute respiratory tract complaints in Malta: a 1-year repeated cross-sectional surveillance study. BMJ Open. 2019;9(12):e032704. https://doi.org/10.1136/bmjopen-2019-032704.
Ministry of Health, Labour and Welfare. NDB Open Data [In Japanese]. 2021. https://www.mhlw.go.jp/content/12400000/000821378.pdf. Accessed 5 Aug 2022.
Ishikawa KB. Medical big data for research use: current status and related issues. Japan Med Assoc J. 2016;59(2–3):110–24.
World Health Organization. Defined daily dose (DDD). https://www.who.int/tools/atc-ddd-toolkit/about-ddd. Accessed 5 Aug 2022.
Statistic Bureau, Ministry of Internal Affairs and Communications. E-Stat. https://www.e-stat.go.jp/en. Accessed 5 Aug 2022.
WHO Collaborating Centre for Drug Statistics Methodology. Norwegian Institute of Public Health. Antiinfectives for systemic use. https://www.whocc.no/atc_ddd_index/?code=j01. Accessed 2 Aug 2022.
Statistic Bureau, Ministry of Internal Affairs and Communications. e-Stat. Population estimates. https://www.e-stat.go.jp/en/stat-search/files?page=1&toukei=00200524&tstat=000000090001. Accessed 3 Aug 2022
Kanto-Shinetsu Regional Bureau of Health and Welfare. List of insurance medical institutions, insurance pharmacies, and newly registered insurance physicians and pharmacists. [In Japanese]. https://kouseikyoku.mhlw.go.jp/kantoshinetsu/chousa/shitei.html. Accessed 29 Aug 2022.
Kanto-Shinetsu Regional Bureau of Health and Welfare. Notification status of facility standards (overall) (list of medical institutions that have received notification). [In Japanese]. https://kouseikyoku.mhlw.go.jp/kantoshinetsu/chousa/kijyun.html. Accessed 29 Aug 2022.
Ministry of Health, Labour and Welfare. Individual Matters. No. 16. Additional Reimbursement for Infection Control. [In Japanese]. 2019. https://www.mhlw.go.jp/content/12404000/000583841.pdf. Accessed 22 Aug 2022.
The Japanese Association for Infectious Diseases. Roster list of certified physicians. July 1, 2022 https://www.kansensho.or.jp/uploads/files/senmoni/meibo_220701.pdf. Accessed 3 Aug 2022.
European Centre for Disease Prevention and Control. European Surveillance of Antimicrobial Consumption Network (ESAC-Net). https://www.ecdc.europa.eu/en/about-us/partnerships-and-networks/disease-and-laboratory-networks/esac-net. Accessed 2 Nov 2023.
Schröder W, Sommer H, Gladstone BP, et al. Gender differences in antibiotic prescribing in the community: a systematic review and meta-analysis. J Antimicrob Chemother. 2016;71(7):1800–6. https://doi.org/10.1093/jac/dkw054.
Hashimoto H, Saito M, Sato J, et al. Indications and classes of outpatient antibiotic prescriptions in Japan: a descriptive study using the national database of electronic health insurance claims, 2012–2015. Int J Infect Dis. 2020;91:1–8. https://doi.org/10.1016/j.ijid.2019.11.009.
Franchi C, Mandelli S, Fortino I, Nobili A. Antibiotic use and associated factors in adult outpatients from 2000 to 2019. Pharmacol Res Perspect. 2021;9(6):e00878. https://doi.org/10.1002/prp2.878.
Smith DRM, Dolk FCK, Smieszek T, Robotham JV, Pouwels KB. Understanding the gender gap in antibiotic prescribing: a cross-sectional analysis of English primary care. BMJ Open. 2018;8(2):e020203. https://doi.org/10.1136/bmjopen-2017-020203.
Araki Y, Momo K, Yasu T, et al. Prescription pattern analysis for antibiotics in working-age workers diagnosed with common cold. Sci Rep. 2021;11(1):22701. https://doi.org/10.1038/s41598-021-02204-3.
Hashimoto H, Matsui H, Sasabuchi Y, et al. Antibiotic prescription among outpatients in a prefecture of Japan, 2012–2013: a retrospective claims database study. BMJ Open. 2019;9(4):e026251. https://doi.org/10.1136/bmjopen-2018-026251.
Sako A, Yasunaga H, Matsui H, et al. Hospitalization for urinary tract infections in Japan, 2010–2015: a retrospective study using a national inpatient database. BMC Infect Dis. 2021;21(1):1048. https://doi.org/10.1186/s12879-021-06735-y.
Asmusses JB. Statens Serum Institut. DANMAP reports, 2022. https://www.danmap.org/reports. Accessed 2 Nov 2023.
Mallah N, Orsini N, Figueiras A, Takkouche B. Education level and misuse of antibiotics in the general population: a systematic review and dose-response meta-analysis. Antimicrob Resist Infect Control. 2022;11(1):24. https://doi.org/10.1186/s13756-022-01063-5.
Henaine AM, Lahoud N, Abdo R, et al. Knowledge of antibiotics use among lebanese adults: a study on the influence of sociodemographic characteristics. Sultan Qaboos Univ Med J. 2021;21(3):442–9. https://doi.org/10.18295/squmj.4.2021.006.
Stivers T, Timmermans S. Arriving at no: patient pressure to prescribe antibiotics and physicians’ responses. Soc Sci Med. 2021;290: 114007. https://doi.org/10.1016/j.socscimed.2021.114007.
Pan X, Slater M, Beacco A, et al. The responses of medical general practitioners to unreasonable patient demand for antibiotics—a study of medical ethics using immersive virtual reality. PLoS One. 2016;11(2):e0146837. https://doi.org/10.1371/journal.pone.0146837.
Higashi T, Fukuhara S. Antibiotic prescriptions for upper respiratory tract infection in Japan. Intern Med. 2009;48(16):1369–75. https://doi.org/10.2169/internalmedicine.48.1893.
Butt AA, Navasero CS, Thomas B, et al. Antibiotic prescription patterns for upper respiratory tract infections in the outpatient Qatari population in the private sector. Int J Infect Dis. 2017;55:20–3. https://doi.org/10.1016/j.ijid.2016.12.004.
Silverman M, Povitz M, Sontrop JM, et al. Antibiotic prescribing for nonbacterial acute upper respiratory infections in elderly persons. Ann Intern Med. 2017;166(11):765–74. https://doi.org/10.7326/M16-1131.
Skow M, Fossum GH, Høye S, Straand J, Emilsson L, Brænd AM. Antibiotic treatment of respiratory tract infections in adults in Norwegian general practice. JAC Antimicrob Resist. 2023;5(1):dlac135. https://doi.org/10.1093/jacamr/dlac135.
Sato D, Goto T, Uda K, Kumazawa R, Matsui H, Yasunaga H. Impact of national guidelines for antimicrobial stewardship to reduce antibiotic use in upper respiratory tract infection and gastroenteritis. Infect Control Hosp Epidemiol. 2021;42(3):280–6. https://doi.org/10.1017/ice.2020.427.
Tsuzuki S, Fujitsuka N, Horiuchi K, et al. Factors associated with sufficient knowledge of antibiotics and antimicrobial resistance in the Japanese general population. Sci Rep. 2020;10(1):3502. https://doi.org/10.1038/s41598-020-60444-1.
Committee to prepare the guidelines for the management of respiratory infectious diseases in children. The guidelines for the management of respiratory infectious diseases in children in Japan 2017, KYOWA KIKAKU Ltd., Tokyo (2016) (in Japanese).
Struwe J, Olsson-Liljequist B. Short summary of Swedres 2008, a report on antimicrobial utilisation and resistance in humans in Sweden. Euro Surveill. 2009;14(25):19252.
Hsia Y, Sharland M, Jackson C, Wong ICK, Magrini N, Bielicki JA. Consumption of oral antibiotic formulations for young children according to the WHO access, watch, reserve (AWaRe) antibiotic groups: an analysis of sales data from 70 middle-income and high-income countries. Lancet Infect Dis. 2019;19(1):67–75. https://doi.org/10.1016/S1473-3099(18)30547-4.
Medical Writing/Editorial Assistance
Editing was provided by ThinkSCIENCE, Inc. The cost of the editing service was supported by research grants from the Ministry of Health, Labour and Welfare (23HA2002 and 23HA1004).
Funding
This study was supported by a research grant from the Ministry of Health, Labour and Welfare (23HA2002 and 23HA1004). The journal’s fee was waived.
Author information
Authors and Affiliations
Contributions
Taito Kitano: Study design, Data analysis, Writing the first draft; Shinya Tsuzuki: Study design, Data analysis, Editing, supervision; Ryuji koizumi: Data collection, Data curation, Editing; Kensuke Aoyagi: Data collection, Data curation; Yusuke Asai: Data analysis, Editing; Yoshiki Kusama: Study design, Editing; Norio Ohmagari: Editing, Supervision.
Corresponding author
Ethics declarations
Conflict of Interest
Taito Kitano, Shinya Tsuzuki, Ryuji Koizumi, Kensuke Aoyagi, Yusuke Asai, Yoshiki Kusama, and Norio Ohmagari have nothing to disclose.
Ethical Approval
The study was conducted in accordance with the declaration of Helsinki and approved by the Institutional Review Board at National Center for Global Health and Medicine (Approval No. NCGM-G-003098-01). Patient identifiers were not included in our data used in this study and therefore no informed consent was required to conduct the present study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Kitano, T., Tsuzuki, S., Koizumi, R. et al. Factors Associated with Geographical Variability of Antimicrobial Use in Japan. Infect Dis Ther 12, 2745–2755 (2023). https://doi.org/10.1007/s40121-023-00893-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40121-023-00893-z