Abstract
Purpose
This study examined the characteristics, incidence and prognostic factors of the first AIDS-defining condition developed after more than one year of continuous antiretroviral therapy (ART) among people living with HIV (PLHIV).
Methods
We used data from two multicentre observational cohorts of PLHIV in Germany between 1999 and 2018. Our outcome was the first AIDS-defining event that occurred during follow-up after more than one year of continuous ART. Descriptive analyses at ART initiation, at the time of the AIDS event and of the most frequently observed types of AIDS-defining illnesses were performed. We calculated the incidence rate (IR) per 1000 person-years (PY) and used a bootstrap stepwise selection procedure to identify predictors of the outcome.
Results
A total of 12,466 PLHIV were included in the analyses. 378 developed the outcome, constituting an overall IR of 5.6 (95% CI 5.1–6.2) AIDS events per 1000 PY. The majority of PLHIV was virally suppressed at the time of the event. Oesophageal candidiasis and wasting syndrome were the most frequently diagnosed AIDS-defining illnesses. We found a low CD4 count at ART initiation, a previous AIDS-defining condition and transmission through intravenous drug use to be meaningful prognostic factors of the outcome.
Conclusion
The overall rate of AIDS-defining events among PLHIV under long-term ART was low, highlighting the importance of continuous treatment. PLHIV who started ART with indicators of impaired immune functioning were more susceptible to disease progression, suggesting that the public health response should continue to focus on early and sustained treatment for all PLHIV.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
The introduction of combination antiretroviral therapy (ART) in 1996 has been a milestone in the treatment of HIV. Continuous improvements in ART efficacy and safety as well as adapted prescription guidelines have led to a substantial decline in HIV-related morbidity and mortality among people living with HIV (PLHIV) worldwide [1, 2]. In Germany as of 2021, 96% of all diagnosed PLHIV received HIV treatment of which another 96% had achieved durable viral suppression [3]. With regard to the Joint UN Program on HIV/AIDS (UNAIDS)’s postulated “95-95-95” targets that aim to have diagnosed 95% of all HIV-positive people, provided ART for 95% of those diagnosed and achieved viral suppression for 95% of those treated by 2030 [4], Germany has reached the two ART-related endpoints well ahead of the target date. On the basis of these goals, the overarching aim put forward by UNAIDS was to “end the AIDS epidemic by 2030” [4].
These encouraging developments are reflected in the incidence rates (IRs) of AIDS in Germany. In a previous study, we have analysed changes in the AIDS rates among PLHIV under clinical care by years of follow-up as well as calendar periods between 1999 and 2018 [5]. In line with other prior research conducted in the Western European and North American context, we found that the IR of a first AIDS-defining condition has continuously declined by both years of follow-up and calendar periods since the introduction of effective ART [5,6,7,8]. The majority of AIDS events were observed at baseline and within the first year of follow-up, suggesting that the progression to AIDS can largely be attributed to late HIV diagnoses [5]. This was further corroborated by our finding that indicators of advanced HIV infections, i.e., a low CD4 cell count, high viral load, and older age at baseline, were predictive of disease progression [5].
Despite these developments, there still remains a number of PLHIV in Germany who progress to AIDS even after long-term ART exposure [5]. Even though these cases can be considered rare since the advent of effective ART [9], it is important to examine them since their occurrence can neither be attributed to a still recovering immune response as AIDS events in the early phase after starting ART nor initial complications like the development of the immune reconstitution inflammatory syndrome (IRIS), a hyperinflammatory reaction that usually manifests within the first months after starting treatment [10, 11]. The incidence and specific characteristics of AIDS events developed after continuous long-term therapy in the era of effective ART have not been sufficiently examined yet.
The objective of the current study was, therefore, to analyse the occurrence of AIDS-defining illnesses among PLHIV with long-term regular ART intake from two German observational cohorts. More specifically, we were interested in examining the incidence of the first AIDS-defining condition developed after more than one year of continuous ART. Here, we aimed to analyse the characteristics of these PLHIV at the time of ART initiation, at the time of the AIDS event as well as the types of AIDS-defining illnesses that were diagnosed. In addition to that, we sought to identify potential predictors of the first AIDS-defining condition that occurred under regular ART. A better understanding of those AIDS cases is needed to assess the feasibility of UNAIDS’s goal of “ending the AIDS epidemic by 2030” [4] in the context of Germany and can help to direct the public health response on the route to achieving this goal.
Methods
Study design and population
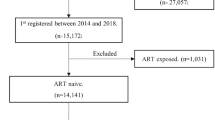
We used data from the HIV-1 Seroconverter Study and ClinSurv HIV Study, which are two German multicentre, open, prospective long-term observational cohorts initiated in 1997 and 1999, respectively [12, 13]. Both cohorts are hosted and coordinated at the Robert Koch Institute (RKI) where the data are used for the clinical surveillance of PLHIV in Germany. Participating HIV practitioners regularly report pseudonymised information on demographic properties, laboratory values, and other clinical events from routine visits. In the HIV-1 Seroconverter Study, eligibility criteria include the knowledge of a person’s approximate time of infection based on a negative HIV test within three years before the diagnosis or a diagnosis during acute seroconversion. In the ClinSurv HIV Study, all PLHIV who receive treatment in one of the participating HIV clinics are eligible for inclusion. The study designs have been described in detail in previous publications [5, 12, 13]. In the current study, we combined the data of both cohorts and included PLHIV who were enrolled between 1999 and 2018. We excluded PLHIV under the age of 18 years and PLHIV whose ART initiation date was missing or who had initiated ART before the enrolment in one of the cohorts. In the case of ART interruptions, we censored all patient observations that were recorded after the first interruption to only include person-time of PLHIV while being prescribed ART. We furthermore excluded all PLHIV from the analyses who did not complete a year of ART, which includes those who developed an AIDS event within the first year after ART initiation.
Outcome
Our outcome was the first AIDS-defining event that occurred during follow-up after more than one year of continuous ART since therapy initiation. An AIDS-defining event was comprised of a diagnosis of one or potentially more AIDS-defining illnesses in the same month as the cohort data are recorded monthly. We included all clinical conditions associated with AIDS as published by the Centers for Disease Control and Prevention (CDC) [14]. As opposed to the CDC’s expanded AIDS surveillance case definition, we did not characterise a CD4 cell count drop below 200 cells/µL as an AIDS-defining event [14].
Predictor selection
We selected potential predictors of the first AIDS-defining event developed after more than one year of continuous ART on the basis of prior research and availability in our dataset [15,16,17,18]. These included age at ART initiation, gender, transmission mode, country of origin, CD4 count at ART initiation, plasma HIV-1 RNA viral load (VL) at ART initiation, and a diagnosis of a previous AIDS event before or at ART initiation. Age, CD4 count and VL were treated as continuous variables. Gender was categorised into male vs. female, transmission mode into men who have sex with men (MSM), persons with heterosexual contact (HET), persons who inject drugs (PWID), persons from high-prevalence countries (PHPC) and “other”, country of origin into Germany vs. abroad, and a previous AIDS event into yes vs. no. The transmission group PHPC comprised PLHIV who migrated from countries with an HIV prevalence of > 1% among the population between 15 and 49 years.
Statistical analysis
We performed descriptive analyses at baseline, i.e., the month of ART initiation. The ART initiation date could fall on the same date as a person’s enrolment date in one of the two cohorts or later if ART was prescribed over the course of follow-up. We present the mean and standard deviation (SD) for the variable age and the median and interquartile range (IQR) for CD4 count and VL. In the case of missing CD4 or VL values at baseline, we used, if available, records up to three months after ART initiation. For the categorical variables, percentages are presented and corresponding 95% confidence intervals (95% CI) when proportions are described and compared. To examine whether the baseline data differed among PLHIV who experienced the outcome at different points in time throughout follow-up, we additionally stratified the baseline characteristics by years under continuous ART at the time of the AIDS event. The strata selected were AIDS after > 1–3 years, > 3–6 years, and > 6 years of continuous ART, with 3 years corresponding to the approximate median time and 6 years to the third quartile until the outcome.
We performed further descriptive analyses of the characteristics at the time of the first AIDS event under continuous ART. Here, we present the time-varying variables age, CD4 count and VL. In addition to that, we created the variable “Experienced viral load increase (> 200 copies/mL) before the AIDS event”, which indicated the occurrence of at least one episode of viraemia before the AIDS event after already having achieved viral suppression. Since a detectable VL at the time of the AIDS event can be an indicator of suboptimal ART adherence [19], we additionally stratified VL by transmission mode to examine potential differences between the groups. As in our baseline analyses, we also performed stratified analyses by the time until the AIDS event occurred.
Further descriptive analyses were performed with regard to the types of AIDS-defining illnesses. We present the five most frequently observed AIDS-defining illnesses among PLHIV who developed the AIDS event after more than one year of continuous ART intake. To further analyse whether certain illnesses were diagnosed more often at particular CD4 or VL categories, differed across the transmission groups or with respect to the time under continuous ART until the AIDS event, we also stratified the analyses of the AIDS-defining illnesses on these variables. We do not present the transmission groups “other” and “unknown” due to sparse data and limited interpretability.
In our time-to-event analyses, we first calculated the incidence rate (IR) and corresponding 95% CI of the first AIDS-defining event per 1000 person-years (PY). Follow-up started one year after ART initiation. PLHIV were censored at the first AIDS-defining event, the first ART interruption, loss-to-follow-up, death or administrative censorship (December 31, 2018). Seeing that with the continuous improvements in ART efficacy and safety treatment regimens and guidelines were amended over time, we split our study period and additionally calculated the IR in each period for comparison. We selected the time periods of 1999 to 2009 and 2010 to 2018, including only person-time of PLHIV within the respective period. 2010 was chosen as the dividing year as the US Department of Health and Human Services (DHHS) and the German and Austrian AIDS societies (DAIG, ÖAG) amended their guidelines at that time, recommending ART generally for all patients with CD4 counts below 500 cells/µL [20, 21].
To identify prognostically meaningful variables for the development of an AIDS event, we utilised the bootstrap stepwise selection procedure by Sauerbrei and Schumacher [22]. Here, we carried out 500 bootstrap replications of the original data set. In each replication, potential predictors were selected by a stepwise algorithm which only kept variables in the Cox proportional hazards regression model that did not exceed our a priori selected cut-off p-value of 0.1. The absolute and relative inclusion frequencies then served as a criterion for the predictive importance of the respective variables. We considered variables as predictive that were selected in the model in a high majority (> 80%) of the bootstrapped samples. As our potential predictors were all continuous or binary except for transmission mode, we dichotomised transmission mode and conducted sensitivity analyses repeating the selection procedure with separate binary variables for each transmission group. MSM served as the reference category for transmission mode and was not included as a variable for collinearity reasons. The transmission groups “other” and “unknown” were excluded due to sparse data and limited interpretability. This sensitivity check was conducted to examine whether one or more of the transmission groups stood out as a meaningful predictor of the outcome. To detect potential time trends, we also carried out the predictor analyses in each of our selected time periods (1999–2009 and 2010–2018).
All analyses were performed using Stata 17.0 (Stata Statistical Software: Release 17, United States).
Ethical approval
The HIV-1 Seroconverter Study was approved by the ethics commission at the Charité—Universitätsmedizin Berlin (EA2/105/05). The ClinSurv HIV Study data between 1999 and 2018 were collected anonymously in compliance with the German Infection Protection Act (IfSG) as of 2001. As the data collection adhered to the legal requirements of the IfSG, no written informed consent was required. Approval was granted by the RKI data protection officer and the Federal Commissioner for Data Protection and Freedom of Information.
Results
Baseline characteristics
A total of 12,466 PLHIV were included in the analyses (Fig. 1), of which 378 experienced an AIDS-defining event after more than one year of continuous ART and 12,088 did not. Table 1 presents the baseline characteristics of both groups, showing that they were comparable with regard to age, gender, country of origin, and VL. Mean age at ART initiation was 40.3 years (SD 10.6) among PLHIV with the outcome and 39.9 years (SD 10.8) among PLHIV without the outcome. The majority of PLHIV were male (80.4%, 95% CI 76.1–84.3 vs. 80.8%, 95% CI 80.1–81.5) and born in Germany (68.8%, 95% CI 63.8–73.4 vs. 68.9%, 95% CI 68.0–69.7). While the transmission groups HET (18.0%, 95% CI 14.2–22.2 vs. 15.3%, 95% CI 14.7–16.0) and PHPC (15.3%, 95% CI 11.9–19.4 vs. 13.3%, 95% CI 12.7–13.9) were comparable in size, MSM were less frequently represented among PLHIV who developed an AIDS-defining condition (45.8%, 95% CI 40.7–50.9 vs. 52.1%, 95% CI 51.2–53.0) and PWID more frequently (10.6%, 95% CI 7.7–14.1 vs. 5.3%, 95% CI 4.9–5.7). PLHIV with an AIDS event had lower CD4 counts at ART initiation compared to those without an AIDS event (median 178 cells/µL, IQR 73–313 vs. median 280 cells/µL, IQR 144–433), while the VLs were comparable (median 24,930 copies/mL, IQR 350–164,313 vs. median 10,000 copies/mL, IQR 219–110,000). Moreover, PLHIV with the outcome more often had already experienced a previous AIDS-defining condition at baseline compared to those without the outcome (32.3%, 95% CI 27.6–37.2 vs. 14.6%, 95% CI 14.0–15.3).
Our stratified analyses of PLHIV with an AIDS event showed that in the majority the event occurred after > 1–3 years since ART initiation (Table S1, Additional file 1). PLHIV who experienced the outcome after > 6 years were predominantly male (91.1%, 95% CI 83.2–96.1). CD4 counts at baseline were higher among PLHIV who developed an AIDS event later during follow-up (> 1–3 years: median 153 cells/µL, IQR 40–296, > 3–6 years: median 181 cells/µL, IQR 85–298, > 6 years: median 214 cells/µL, IQR 119–362).
Characteristics at the time of the AIDS-defining event
In Table 2, we present the characteristics at the time of the AIDS event among the 378 PLHIV who experienced the outcome. We observed a mean age of 44.6 years (SD 11.1) and a median CD4 count of 324 cells/µL (IQR 127–510). With regard to the CD4 counts and VLs, missing values were obtained in more than half of the cases. 66.3% of the PLHIV with available VL records were virally suppressed (< 50 copies/mL) when the AIDS-defining condition was diagnosed. Over 90% had achieved viral suppression over the course of follow-up before the AIDS event. Here, 58.5% had sustained durable viral suppression or experienced only minor VL increases at some point before the AIDS event, and 32.0% had experienced at least one episode of viraemia with more than 200 copies/mL. 9.5% had never achieved viral suppression before the event occurred (Table 2). In the additional stratified analyses of VL, we observed the lowest proportion of viral suppression at the time of the AIDS event among PWID (42.9%, 95% CI 17.7–71.1) (Table S2, Additional file 2).
Our stratified analyses by follow-up time showed that the later throughout follow-up the AIDS-defining condition developed, the higher the age (> 1–3 years: mean 41.1 years, SD 10.2, > 3–6 years: mean 46.3 years, SD 12.2, > 6 years: mean 49.3 years, SD 9.3). CD4 counts at the time of the AIDS event were highest among PLHIV who experienced the outcome after > 6 years of continuous ART (median 397 cells/µL, IQR 139–738). We observed no discernible pattern with regard to the proportion of PLHIV with undetectable VLs (> 1–3 years: 66.3%, 95% CI 55.5–76.0, > 3–6 years: 65.9%, 95% CI 49.4–79.9, > 6 years: 66.7%, 95% CI 49.8–80.9) (Table S3, Additional file 2). While 16.7% (95% CI 11.5–22.9) of the PLHIV who developed an AIDS condition after > 1–3 years had experienced at least one episode of viraemia above 200 copies/mL at some point before the outcome, this proportion increased to 64.4% (95% CI 53.7–74.3) among PLHIV who experienced the outcome after > 6 years (Table S3, Additional file 2).
Types of AIDS-defining illnesses
The five most frequently observed AIDS-defining illnesses were oesophageal candidiasis (14.7%), wasting syndrome (11.3%), pneumocystis-jirovecii-pneumonia (10.3%), HIV encephalitis (10.1%), and HSV ulcers (7.5%) (Table 3).
Several differences were found in the stratified analyses: Among PLHIV who developed the AIDS event after > 1–3 years of continuous ART, we observed oesophageal candidiasis (13.8%), wasting syndrome (10.8%), pneumocystis-jirovecii-pneumonia (9.7%), as well as extrapulmonary tuberculosis (9.2%) and pulmonary tuberculosis (8.2%) (Table S3, Additional file 3). While oesophageal candidiasis, wasting syndrome, and pneumocystis-jirovecii-pneumonia were also among the five most common illnesses of PLHIV who experienced the outcome after > 6 years, the tuberculoses were replaced by HIV encephalitis (13.0%) and atypical mycobacteria (8.0%). With respect to the transmission groups, we observed extrapulmonary tuberculosis and pulmonary tuberculosis only among the top five illnesses of PHPC with 25.0% and 11.7%, respectively, while the diagnosed illnesses among MSM, HET, and PWID were comparable and predominantly included wasting syndrome and oesophageal candidiasis. The types of illnesses also differed somewhat with regard to the CD4 counts at which they developed. Cerebral toxoplasmosis and cytomegalovirus manifested more frequently at lower CD4 counts (< 50 cells/µL: both 9.4%, 50–199 cells/µL: both 8.2%), while HSV ulcers and Kaposi’s sarcoma more frequently at higher CD4 counts of > 500 cells/µL (17.0% and 10.6%). No discernible patterns were found with regard to the VLs (Table S3, Additional file 3).
Incidence and predictors
After completing a year of ART, a total of 12,466 PLHIV contributed person-time and were followed-up for a median of 4.3 years (range 1 month to 18.8 years). We recorded 378 first AIDS-defining events that constituted an IR of 5.6 (95% CI 5.1–6.2) AIDS events per 1000 PY among PLHIV under continuous ART between 1999 and 2018. Median follow-up time until the AIDS event was 2.2 years (range 1 month to 17.2 years), which translates into a median time of 3.2 years (range 13 months to 18.2 years) from ART initiation until the AIDS event. In our stratified time period analyses, we obtained an IR of 11.1 (95% CI 9.6–12.8) AIDS events per 1000 PY between 1999 and 2009 and an IR of 3.9 (95% CI 3.1–4.8) between 2010 and 2018.
In Table 4, we present the absolute and relative inclusion frequencies of the potential predictors after 500 bootstrap replications. The variables CD4 count at ART initiation and a previous AIDS event before ART initiation were included in the model in 499 (99.8%) and 498 (99.6%) of the bootstrapped samples, suggesting that these two variables were strong prognostic factors for the development of an AIDS-defining condition after more than one year of continuous ART. The direction of the predictors was derived from our descriptive analyses, which indicated that the lower the CD4 count at baseline and “yes” in regard to a previous AIDS event, the higher the risk of developing an AIDS-defining condition (Table 1). The variables country of origin, transmission mode, gender, VL at ART initiation, and age at ART initiation followed with inclusion frequencies of 176 (35.2%), 170 (34.0%), 79 (15.8%), 55 (11.0%), and 43 (8.6%) times, respectively, suggesting that neither of those were of meaningful predictive importance for the outcome. While we observed a similar distribution of the variables in the time period between 1999 and 2009 (Table S5, Additional file 4), none of the variables were selected in > 80% of the bootstrapped samples in the later period between 2010 and 2018 (Table S6, Additional file 4).
Our sensitivity analyses in which we repeated the bootstrap stepwise selection procedure with separate binary variables for each transmission mode category showed that again, CD4 count at ART initiation and a previous AIDS event before ART initiation were selected in nearly all of the bootstrapped samples (Table S7, Additional file 4). In addition to that, the transmission group PWID was included in the model 407 (81.4%) times, suggesting that intravenous drug use can be considered another relevant predictor of the development of an AIDS-defining condition while receiving ART. HET and PHPC were included in only 156 (31.2%) and 103 (20.6%) of the bootstrap replications and were therefore not considered predictive of the outcome. In our time period analyses, we again observed a similar distribution in the first period between 1999 and 2009, in which a previous AIDS event and CD4 count were selected in the majority of the bootstrapped samples, however not PWID (Table S8, Additional file 4). In the second period between 2010 and 2018, again no variable crossed the 80% mark, suggesting that none of these variables were of meaningful predictive utility of the outcome in the later time period (Table S9, Additional file 4).
Discussion
This study investigated the characteristics, incidence and prognostic factors of a first AIDS-defining condition developed after more than one year of continuous ART among PLHIV in Germany between 1999 and 2018. Our main findings were that PLHIV with the outcome had lower CD4 counts at ART initiation, had more often already developed an AIDS event before ART was initiated and more often were intravenous drug users compared to those without the outcome. At the time of the AIDS event, the majority of PLHIV was virally suppressed and nearly 60% had sustained durable viral suppression or experienced only minor VL increases at some point before the event. In addition to that, we found that oesophageal candidiasis and wasting syndrome were the most frequently diagnosed AIDS-defining illnesses. While these were observed across all strata, other illnesses occurred more often among certain strata such as extrapulmonary and pulmonary tuberculosis, which were diagnosed more frequently among people from high-prevalence countries. We obtained an overall low IR of the outcome, which was lower in the later study period between the years 2010 and 2018 compared to the earlier period between 1999 and 2009. A low CD4 count, a previous AIDS-defining condition at ART initiation and transmission through intravenous drug use were identified as overall meaningful prognostic factors of the outcome.
What stood out in our descriptive analyses was the observation that the majority of PLHIV with available VL records was virally suppressed at the time of the AIDS event. This finding builds on existing evidence of prior studies, which have also reported diagnoses of AIDS-defining conditions in the presence of undetectable VLs [23,24,25,26]. While the proportion of virally suppressed PLHIV at the time of the AIDS event was the same across the time strata, the proportion of PLHIV who experienced at least one episode of viraemia at some point before the AIDS event increased substantially the later across follow-up the event occurred. Even though we only analysed person-time from patient records indicating continuous ART intake, occasional non-adherence which can lead to episodes of viraemia can happen due to various reasons [27, 28]. In addition to that, although rare, HIV drug resistance resulting in viral failure can emerge despite potent ART drugs and adherence [29]. We assume that both sporadic ART non-adherence and drug resistance could have played a role in our study population, seeing that we also observed a considerable number of PLHIV with high VLs at the time of the AIDS event. The fact that even short episodic viraemia can negatively affect the immune response [26] highlights the importance of supporting ART adherence and a close monitoring of the VLs to spot potential fluctuations.
Insights into further potential factors to be regarded over the course of treatment are provided by our findings of the analysis of the most frequently diagnosed AIDS-defining illnesses. The results were partially consistent with those of our previous study in which we had analysed the AIDS rates among PLHIV under clinical care irrespective of ART status or duration [5]. While oesophageal candidiasis, wasting syndrome, and pneumocystis-jirovecii-pneumonia were also found among the top five illnesses in our previous study, HIV encephalitis and HIV ulcers as in the present study were not. As our stratified analyses showed, both illnesses, particularly HIV encephalitis, developed more often later in time after several years of continuous ART. Previous research found that certain diseases such as cardiovascular diseases and diabetes can increase the risk of HIV encephalitis [30]. Seeing that such noncommunicable conditions which are widespread and age-related [31, 32] potentially increase the risk of AIDS-defining conditions emphasises that attention also needs to be given to the comorbidities of PLHIV in therapy.
This is further corroborated by our finding that tuberculosis was the most common illness only among migrants from high-prevalence countries. As pointed out by prior research, the prevalence of the different types of AIDS-defining illnesses differs by region with tuberculosis disproportionally affecting countries in Sub-Saharan Africa [33,34,35], which make up the majority of the high-prevalence countries [36]. Besides HIV being a prognostic factor of tuberculosis itself, other health-affecting factors that are more common in these regions such as lower socioeconomic status together with malnutrition, improper housing conditions, or the usage of contaminated water further increase the risk of tuberculosis [37,38,39]. This finding hence also underlines the significance of PLHIV’s overall health status in regard to disease progression, which should be regularly checked and strengthened.
The results of our prediction analysis provide new insights into certain prognostic characteristics at ART initiation. The most often selected predictors for the development of an AIDS event after more than one year of continuous treatment were a low CD4 count and a previous AIDS-defining condition, which are both indicators of advanced HIV infections and an already substantially impaired immune response [3, 40]. While our and other previous studies that considered all AIDS cases including those developed shortly after ART initiation reported additional predictors such as a high VL and older age [5, 15,16,17,18], the findings of the present study suggest that factors directly related to immune functioning are the most critical when it comes to AIDS events after continuous long-term ART exposure. Our stratified analyses by time period revealed that while these two factors were also the strongest predictors in the years between 1999 and 2009, none of our available potential predictors showed to be of meaningful predictive importance in the later time period between 2010 and 2018. This finding suggests that with improved therapy regimens and amended guidelines recommending earlier treatment, these factors at ART initiation have become less useful to predict the occurrence of AIDS events after continuous long-term therapy. Further analyses considering additional variables would be needed to identify relevant prognostic factors among PLHIV who have initiated treatment in the more recent years.
Even though the variable transmission mode was not predictive of the outcome and therefore has only a little importance in the clinical context when considering prognostic factors, our sensitivity analyses of the entire study period showed that one category stood out and was predictive, which was transmission through intravenous drug use. In line with other prior research, this supports the point that PWID represents a vulnerable population with a higher susceptibility to disease progression after long-term treatment, potentially due to additional challenges with respect to treatment adherence, overall health, and access to clinical care with a resulting higher risk of late diagnoses [41,42,43,44]. Our analyses have also shown that PWID were the group that most often had detectable VLs at the time of the AIDS event, possibly indicating suboptimal adherence. Adherence issues among PWID can be explained by social limitations such as homelessness which are observed at higher rates among this population [45]. In addition to that, PWID more often experience mental health disorders that can affect treatment adherence and are overall at an increased risk of developing various comorbidities [46, 47], making them more susceptible to HIV disease progression.
Overall, the results of our study are encouraging and confirm the substantial improvements with respect to HIV-related morbidity among PLHIV in therapy since the advent of effective ART. In our 20-year study period, we observed only 378 PLHIV with a first AIDS-defining event under continuous long-term ART that constituted an overall low rate which was even lower in the more recent study period, highlighting the importance of sustained clinical care and ART. The main objective therefore remains to ensure that all HIV positive persons are diagnosed early and receive continuous treatment, which is underlined by our finding that indicators of advanced HIV infections at the time of ART initiation were overall strong prognostic factors of the outcome. Even though the rate was low, our results however also demonstrated that AIDS-defining events among PLHIV under long-term ART still occur, and they do so across a broad time span, with a wide range of different types of illnesses as well as in the presence of viral suppression. This allows for the assumption that even if all UNAIDS “95-95-95” targets were met or exceeded, there would still remain a small number of AIDS cases. To advance towards UNAIDS’s goal of “ending the AIDS epidemic by 2030” [4], we therefore suggest that the public health response continues to focus on early HIV detection to minimise the number of late diagnoses and consequently the number of PLHIV starting treatment with impaired immune functioning. In addition to that, supporting ART adherence and closely monitoring HIV-related parameters remains of importance, in particular among PLHIV who are more susceptible to disease progression.
Strengths and limitations
Strengths of the present study include the long observation time as well as the large number of PLHIV included in the analyses. As our study period comprised almost the entire time of treatment with effective ART since its introduction, our results can be considered informative for Germany and other countries with similar characteristics in terms of ART coverage, HIV prevalence and other population demographics.
Limitations of our study include the substantial proportion of missing CD4 and VL values at the time of the AIDS event. This might have led to biased results with a potential shift in the distribution of these variables, which would have particularly affected the conclusions in regard to PLHIV with undetectable VLs at the time of the AIDS event. Since our stratified analyses however showed similar proportions of missing CD4 and VL values across the time strata and we have no further knowledge of a certain pattern regarding PLHIV whose values at the time of the outcome were measured or not, we assume that the overall results were not considerably affected by the missing data. Another limitation to be considered is that the participating HIV clinics are mostly specialised centres in which a higher number of PLHIV with advanced HIV infections is treated [13]. We might have therefore obtained somewhat higher IRs compared to the ones that would have been observed in the entire population of PLHIV in Germany. Finally, the common limitations of cohort studies also apply to our study, such as loss to follow-up including drop-outs that potentially resulted in missed outcomes and attrition bias.
Conclusion
Our study has shown that the overall rate of a first AIDS-defining event among PLHIV who have been receiving continuous ART for more than a year was low, highlighting the importance of sustained clinical care and ART. Nevertheless, AIDS events after long-term therapy still occur in the era of effective ART and can be expected across a broad time span as well as with different types of AIDS-defining illnesses. Indicators of impaired immune functioning at treatment initiation, i.e., a low CD4 count, a previous AIDS-defining condition and transmission through intravenous drug use, showed to be the most meaningful prognostic factors. Overall, our findings suggest that to further reduce the number of AIDS events among PLHIV receiving ART, the public health response should continue to focus on ensuring early and sustained treatment for all PLHIV.
Data availability
The data generated and/or analysed during the current study are not publicly available due to data protection and confidentiality agreements.
References
Katz IT, Maughan-Brown B. Improved life expectancy of people living with HIV: who is left behind? Lancet HIV. 2017;4:e324–6.
UNAIDS. Global HIV & AIDS statistics — Fact sheet. 2021 [cited 2023 Aug 2]. https://www.unaids.org/en/resources/fact-sheet.
HIV in Deutschland 2021. Epidemiologisches Bulletin: Robert Koch-Institut. 2022 [cited 2023 Aug 2]. https://www.rki.de/DE/Content/Infekt/EpidBull/Archiv/2022/Ausgaben/47_22.pdf?__blob=publicationFile.
UNAIDS. Fast-Track - Ending the AIDS epidemic by 2030. 2014 [cited 2023 Aug 2]. https://www.unaids.org/en/resources/documents/2014/JC2686_WAD2014report.
Pantke A, Kollan C, Gunsenheimer-Bartmeyer B, Jensen B-EO, Stephan C, Degen O, et al. AIDS in the era of antiretroviral therapy: Changes in incidence rates and predictors of AIDS among people living with HIV under clinical care in Germany, a cohort study 1999–2018. HIV Med. 2023;1–3.
HIV/AIDS surveillance in Europe 2020 – 2019 data. Copenhagen: WHO Regional Office for Europe; 2020.
Moore RD, Chaisson RE. Natural history of HIV infection in the_era of combination antiretroviral therapy. AIDS. 1999;13:1933–42.
Nizami S, Morales C, Hu K, Holzman R, Rapkiewicz A. Trends in mortality from human immunodeficiency virus infection, 1984–2016: an autopsy-based study. Arch Pathol Lab Med. 2019;144:572–9.
Deutsche Aidshilfe. HIV-Behandlung. 2023 [cited 2023 Aug 3]. https://www.aidshilfe.de/hiv-behandlung.
Brust JCM, McGowan JP, Fine SM, Merrick ST, Radix AE, Vail RM, et al. New York State Department of Health AIDS Institute Clinical Guidelines. Management of Immune Reconstitution Inflammatory Syndrome (IRIS). Baltimore (MD): Johns Hopkins University; 2021.
World Health Organization. Antiretroviral therapy of HIV infection in infants and children: towards universal access: recommendations for a public health approach—2010 revision. Geneva: World Health Organization; 2010.
HIV-Studien und HIV-Projekte am Robert Koch-Institut. Epidemiologisches Bulletin: Robert Koch-Institut. 2019 [cited 2023 Aug 9]. https://www.rki.de/DE/Content/Infekt/EpidBull/Archiv/2019/Ausgaben/49_19.pdf?__blob=publicationFile.
Bätzing-Feigenbaum J, Kollan C, Kühne A, Matysiak-Klose D, Gunsenheimer-Bartmeyer B, Hamouda O. Cohort profile: the German ClinSurv HIV project—a multicentre open clinical cohort study supplementing national HIV surveillance. HIV Med. 2010;12:269–78.
Centers for Disease Control and Prevention. 1993 Revised Classification System for HIV Infection and Expanded Surveillance Case Definition for AIDS Among Adolescents and Adults. MMRW 1992;41(No RR-17).
Egger M, May M, Chêne G, Phillips AN, Ledergerber B, Dabis F, et al. Prognosis of HIV-1-infected patients starting highly active antiretroviral therapy: a collaborative analysis of prospective studies. Lancet. 2002;360:119–29.
Langford SE, Ananworanich J, Cooper DA. Predictors of disease progression in HIV infection: a review. AIDS Res Ther. 2007;4:11.
May M, Sterne JAC, Sabin C, Costagliola D, Justice AC, Thiébaut R, et al. Prognosis of HIV-1-infected patients up to 5 years after initiation of HAART: collaborative analysis of prospective studies. AIDS. 2007;21:1185–97.
Wang J, Yuan T, Ding H, Xu J, Keusters WR, Ling X, et al. Development and external validation of a prognostic model for survival of people living with HIV/AIDS initiating antiretroviral therapy. Lancet Reg Health West Pac. 2021;16: 100269.
Usitalo A, Leister E, Tassiopoulos K, Allison S, Malee K, Paul ME, et al. Relationship between viral load and self-report measures of medication adherence among youth with perinatal HIV infection. AIDS Care. 2014;26:107–15.
Gulick RM. Antiretroviral treatment 2010: progress and controversies. J Acquir Immune Defic Syndr. 2010;55 Suppl 1(Suppl 1):S43–8.
Stellbrink HJ. Kommentar zur Neufassung der Deutsch-Österreichischen Leitlinien zur antiretroviralen Therapie. HIV&more. 2010 [cited 2024 Jan 4]. https://www.hivandmore.de/archiv/2010-2/HIV2_10_FoBiStellbrink.pdf.
Sauerbrei W, Schumacher M. A bootstrap resampling procedure for model building: application to the cox regression model. Stat Med. 1992;11:2093–109.
Daly ML, Fogo A, McDonald C, Morris-Jones R. Kaposi sarcoma: no longer an AIDS-defining illness? A retrospective study of Kaposi sarcoma cases with CD4 counts above 300/mm3 at presentation. Clin Exp Dermatol. 2014;39:7–12.
Rodrigues Costa C, Joaquina Cohen A, Lisboa Fernandes O, Carvalho Miranda K, Sena Passos X, Hasimoto Souza L, do Rosário Rodrigues Silva M. Asymptomatic oral carriage of Candida species in HIV-infected patients in the highly active antiretroviral therapy era. Rev Inst Med Trop Sao Paulo. 2006;48(5).
Sullivan A, Lanham T, Krol R, Zachariah S. Pneumocystis jirovecii Pneumonia in a HIV-infected patient with a CD4 count greater than 400 Cells/μL and atovaquone prophylaxis. Case Rep Infect Dis. 2020;2020:8532780.
Young J, Psichogiou M, Meyer L, Ayayi S, Grabar S, Raffi F, et al. CD4 cell count and the risk of AIDS or death in HIV-Infected adults on combination antiretroviral therapy with a suppressed viral load: a longitudinal cohort study from COHERE. PLoS Med. 2012;9: e1001194.
Freeman R, Gwadz M, Francis K, Hoffeld E. Forgetting to take HIV antiretroviral therapy: a qualitative exploration of medication adherence in the third decade of the HIV epidemic in the United States. SAHARA J. 2021;18:113–30.
Hansana V, Sanchaisuriya P, Durham J, Sychareun V, Chaleunvong K, Boonyaleepun S, Schelp FP. Adherence to antiretroviral therapy (ART) among people living with HIV (PLHIV): a cross-sectional survey to measure in Lao PDR. BMC Public Health. 2013;13:617.
World Health Organization. HIV drug resistance. 2022 [cited 2023 Aug 22]. https://www.who.int/news-room/fact-sheets/detail/hiv-drug-resistance.
Cross S, Önen N, Gase A, Overton ET, Ances BM. Identifying risk factors for HIV-associated neurocognitive disorders using the international HIV dementia scale. J Neuroimmune Pharmacol. 2013;8:1114–22.
World Health Organization. Noncommunicable diseases. 2022 [cited 2023 Aug 23]. https://www.who.int/news-room/fact-sheets/detail/noncommunicable-diseases.
King M, Lipsky MS. Clinical implications of aging. Dis Mon. 2015;61:467–74.
Hamada Y, Getahun H, Tadesse BT, Ford N. HIV-associated tuberculosis. Int J STD AIDS. 2021;32:780–90.
Kim YJ, Woo JH, Kim MJ, Park DW, Song JY, Kim SW, et al. Opportunistic diseases among HIV-infected patients: a multicenter-nationwide Korean HIV/AIDS cohort study, 2006 to 2013. Korean J Intern Med. 2016;31:953–60.
Uyei J, Coetzee D, Macinko J, Guttmacher S. Integrated delivery of HIV and tuberculosis services in sub-Saharan Africa: a systematic review. Lancet Infect Dis. 2011;11:855–67.
World Population Review. HIV Rates by Country 2023. 2023 [cited 2023 Aug 23]. https://worldpopulationreview.com/country-rankings/hiv-rates-by-country.
Greenberg AE, Kupka E. Tuberculosis transmission by waste waters: A review. Sewage and Industrial Wastes. 1957;29:524–37.
Narasimhan P, Wood J, Macintyre CR, Mathai D. Risk factors for tuberculosis Pulm Med. 2013;2013: 828939.
Sailo CV, Tonsing MV, Sanga Z, Chhakchhuak Z, Kharkongor F, Fela V, et al. Risk factors of tuberculosis in Mizoram: First report of the possible role of water source. Indian J Tuberc. 2022;69:675–81.
The Lancet HIV. Time to tackle late diagnosis. Lancet HIV. 2022;9: e139.
Farhadian N, Karami Matin B, Farnia V, Zamanian MH, Najafi F, Farhadian M. The prevalence of people who inject drugs among those with HIV late presentation: a meta-analysis. Subst Abuse Treat Prev Policy. 2022;17:11.
Friedman H, Pross S, Klein TW. Addictive drugs and their relationship with infectious deseases. FEMS Immunol Med Microbiol. 2006;47:330–42.
Roy S, Ninkovic J, Banerjee S, Charboneau RG, Das S, Dutta R, et al. Opioid drug abuse and modulation of immune function: consequences in the susceptibility to opportunistic infections. J Neuroimmune Pharmacol. 2011;6:442–65.
Werb D, Milloy MJ, Kerr T, Zhang R, Montaner J, Wood E. Injection drug use and HIV antiretroviral therapy discontinuation in a Canadian setting. AIDS Behav. 2013;17:68–73.
Palepu A, Milloy MJ, Kerr T, Zhang R, Wood E. Homelessness and adherence to antiretroviral therapy among a cohort of HIV-infected injection drug users. J Urban Health. 2011;88:545–55.
Adams M, Sionean C, Broz D, Lewis R, Wejnert C, Group NS. Serious mental illness among young people who inject drugs: An assessment of injection risks and healthcare use. J Infect Dis. 2020;222:S401-S9.
Altice FL, Kamarulzaman A, Soriano VV, Schechter M, Friedland GH. Treatment of medical, psychiatric, and substance-use comorbidities in people infected with HIV who use drugs. Lancet. 2010;376:367–87.
Acknowledgements
We are grateful to all patients who joined the HIV-1 Seroconverter and ClinSurv HIV cohorts and would like to thank the cooperating partners and participating practitioners. We also thank Kerstin Dehmel, Parvin Ghassim, Andrea Kühne, and Andrea Sailer (Department of Infectious Disease Epidemiology, Robert Koch-Institute, Berlin, Germany) for supporting the study and data management. The HIV-1 Seroconverter study centres: Aachen: Heribert Knechten, Petra Panstruga (Praxenzentrum Blondelstraße Aachen); Berlin: Keikawus Arasteh, Michael Rittweger (Vivantes Auguste Viktoria Klinikum); Hans Wesselmann, Nikolai Menner (Charité—Universitätsmedizin Berlin); Ulrich Bohr (Praxiszentrum Kaiserdamm); Heiko Jessen, Arne B. Jessen (Praxis Jessen und Kollegen); Hubert Schulbin, Sascha Brand, (Praxis Kreuzberg); Jan Gumprecht, Beate Weninger (MVZ Infektiologie Ärzteforum Seestrasse); Heribert Hillenbrand, Heiko Karcher (Praxis City Ost); Klaus Fischer, Dietmar Schranz (Praxis Wilmersdorfer); Mathias Vallée (Praxis Dres. Vallée); Jukka Hartikainen, Stephan Grunwald (Zibp—Zentrum für Infektiologie Berlin Prenzlauer Berg—GmbH); Jörg A. Claus, Claudia Thomas (Praxis Claus und Thomas); Roland Grimm, Sarah Schoor (Schwerpunktpraxis Mitte); Christiane Cordes, Reinhold Schröder (Praxis Dr. Cordes); Tobias Glaunsinger (Praxis Prenzlauer Berg); Michael Rausch, Thomas Reineke (Praxis Schöneberg); Gordon Weinberg, Manuel Bruhy (Infektiologisches Zentrum Steglitz); Siegfried Köppe, Peter Kreckel (Praxis Köppe u. Kreckel); Andreas Berger, Sinah Lindemann (UBN/Praxis); Bochum: Norbert H. Brockmeyer, Anja Potthoff (WIR “Walk In Ruhr”); Bonn: Kathrin van Bremen, Jürgen Rockstroh (Universitätsklinikum Bonn); Dortmund: Martin Hower, Claudia Bachmann (Klinikum Dortmund gGmbH); Dresden: Petra Spornraft-Ragaller (Universitätsklinikum Carl Gustav Carus); Dieter Teichmann (Städtisches Klinikum Dresden); Düsseldorf: Björn-Erik Ole Jensen, Falk Hüttig (Universitätsklinikum Düsseldorf); Essen: Stefan Esser; Pia Schenk-Westkamp (Universitätsklinikum Essen); Frankfurt: Annette Haberl, Christoph Stephan (Goethe Universität Frankfurt); Freiburg: Susanne Usadel, Matthias Müller (MVZ Clotten Freiburg, Universitätsklinikum Freiburg); Gießen: Janina Trauth, (Justus-Liebig-Universität Gießen); Hagen: Alan Chavez-Valladares, Gerd Deutschinoff (Agaplesion Allgemeines Krankenhaus Hagen Infektionsambulanz); Halle/Saale: Burkhard Kreft, Danica Lange (Martin-Luther-Universität Halle-Saale); Hamburg: Olaf Degen, Guido Schäfer (Universitätsklinik Hamburg–Eppendorf); Andreas Plettenberg, Frieder Kuhlendahl (Inst. für interdisziplinäre Medizin Hamburg); Dorothea Wiemer, Lavinia Biemann (Bundeswehrkrankenhaus Hamburg am Bernhard-Nocht Institut); Knud Schewe, Christian Hoffmann (ICH, Infektionsmedizinisches Centrum Hamburg); Hannover: Georg Behrens, Matthias Stoll, (Medizinische Hochschule Hannover); Jena: Benjamin T. Schleenvoigt, Mathias W. Pletz (Universitätsklinikum Jena); Koblenz: Ansgar Rieke (Gemeinschaftsklinikum Mittelrhein, Johannes Gutenberg-Universität Mainz); Köln: Stephan Schneeweiß, Stefan Scholten (Praxis Hohenstaufenring); Mark Oette (Krankenhaus der Augustinerinnen, Universität zu Köln); Krefeld: Peter A. Arbter, (Praxis Dr. Arbter); Leipzig: Thomas Grünewald, Jeannine Weidemann (Klinikum St. Georg gGmbH); Ines Ruck (Praxis Dr. Ruck); Ludwigshafen: Bernd Claus (Klinikum Ludwigshafen); Mainz: Martin Sprinzl, Peter R. Galle (Klinikum der Johannes-Gutenberg-Universität Mainz); Mannheim: Matthias P. Ebert, Roger Vogelmann (Medizinische Klinik Universität Heidelberg, Mannheimer Onkologie Praxis); München: Johannes Bogner, Ulrike Hellerer (Klinikum der Ludwig-Maximilians-Universität); Antoniya Todorova, Claudia Traidl-Hoffmann (Klinik und Poliklinik, TU Dermatologie); Birgit Mück (MVZ Karlsplatz); Ramona Pauli (Isarpraxis); Christoph D. Spinner, Jochen Schneider (TU München Klinikum r.d. Isar); Birgit Mück (prinzmed); Neuss: Robert Baumann (Gemeinschaftspraxis Dres. Baumann und Sels); Osnabrück: Niels Schübel, Christiane Berning (Infektiologisches Centrum, Klinikum Osnabrück); Regensburg: Franz Audebert (Praxiszentrum Alte Mälzerei); Stuttgart: Trein A., Schnaitmann E. (Infectomed GbR); Clemens Roll, Simone Marquardt (Praxis Dres Frietsch, Müller, Roll, Stuttgart); Ulm: Georg Härter, Beate Grüner (Universitätsklinikum Ulm) Wiesbaden: Cengiz Güler (Praxis Güler); Zwickau: Steve Rößler (Arztpraxis Rößler). The ClinSurv HIV cohort study centres: Berlin: Dirk Schürmann, Marianne Warncke (Charité, Universitätsmedizin Berlin); Bonn: Jürgen Rockstroh, Jan-Christian Wasmuth, Svetlana Hass (Universitätsklinikum Bonn); Düsseldorf: Björn-Erik Ole Jensen, Cecilie Feind (Universitätsklinik Düsseldorf); Essen: Stefan Esser, Pia Schenk-Westkamp (Universitätsklinikum Essen); Frankfurt: Christoph Stephan, Annette Haberl, Peter Schott (HIV Center Frankfurt); Hamburg: Andreas Plettenberg, Thore Lorenzen, Frieder Kuhlendahl (Inst. für interdisziplinäre Medizin Hamburg); Axel Adam, Thomas Buhk, Stephan Fenske, Stefan Hansen, Christian Hoffmann, Michael Sabranski, Knud Schewe, Hans-Jürgen Stellbrink, Dennis Radzuweit, Alexander Mainka, Constantin Rickassel (ICH, Infektionsmedizinisches Centrum Hamburg); Olaf Degen, Guido Schäfer, Robin Scheiter (Universitätsklinikum Hamburg Eppendorff); Hannover: Matthias Stoll, Steve Gerschmann, Renate Beider (Medizinsche Universität Hannover); Kiel: Heinz-August Horst, Silke Trautmann (Universitätsklinikum Schleswig–Holstein); Köln: Gerd Fätkenheuer, Jörg Janne Vehreschild, Laura Hamacher, Lennart Nicksch (Universitätsklinikum Köln); München: Johannes Bogner, Barbara Sonntag, Oliver Pullen (Ludwig-Maximilians Universität München); Rostock: Carlos Fritzsche (Universitätsklinikum Rostock).
Funding
Open Access funding enabled and organized by Projekt DEAL. This study received no specific grant from any funding agency.
Author information
Authors and Affiliations
Consortia
Contributions
AP, UK, BGB and VB designed the study. CK merged the cohort data and assisted in the preparation of the data. BEOJ, CS, OD and DS are HIV clinicians who contributed to the data collection and assisted in the conception of the study. TK provided guidance with regard to the methodology and supported the data analyses. AP and UK coordinated the study and performed the analyses. AP drafted the manuscript, tables, and figures and all authors contributed to discussing the results and revising the draft. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare that they have no competing interests with the specific matter of the manuscript. Outside of the submitted work, TK reports having received research grants from the Gemeinsamer Bundesausschuss (G-BA—Federal Joint Committee, Germany), and from the Bundesministerium für Gesundheit (BMG—Federal Ministry of Health, Germany). He further has received personal compensation from Eli Lilly, the BMJ, and Frontiers. Outside of the submitted work, UK reports having received research grants from the Bundesministerium für Gesundheit (BMG—Federal Ministry of Health, Germany), and from the Gemeinsamer Bundesausschuss (G-BA—Federal Joint Committee, Germany). Outside of the submitted work, BEOJ reports having received consulting fees and speaker's honoraria from GSK, ViiV Healthcare, Gilead Sciences, MSD, Pfizer, AstraZeneca, Janssen-Cilag, Fresenius Medical Care and Falk Foundation.
Additional information
List of authors in the HIV-1 Seroconverter Cohort and the ClinSurv HIV Cohort are listed in acknowledgements.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pantke, A., Kollan, C., Gunsenheimer-Bartmeyer, B. et al. AIDS-defining events among people living with HIV who have been under continuous antiretroviral therapy for more than one year, a German cohort study 1999–2018. Infection 52, 637–648 (2024). https://doi.org/10.1007/s15010-024-02188-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-024-02188-y