Abstract
Background
BRAT1 (BRCA1-associated ataxia telangiectasia mutated activator 1) is involved in many important biological processes, including DNA damage response and maintenance of mitochondrial homeostasis. Dysfunctional BRAT1 causes variable clinical phenotypes, which hinders BRAT1-related disease from recognition and diagnosis.
Methods
Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement was the guideline for this systematic review. MEDLINE was searched by terms (“BAAT1” and “BRAT1”) from inception until June 21, 2022.
Results
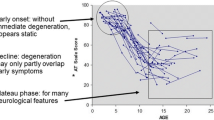
Twenty-eight studies, screened out of 49 records, were included for data extraction. The data from fifty patients with mutated BRAT1 were collected. There are 3 high relevant phenotypes, 4 medium relevant phenotypes and 3 low relevant phenotypes. Eye-related abnormal features were most frequently reported: 27 abnormal features were observed. Thirty-nine kinds of pathogenic nucleotide change in BRAT1 were reported. Top three common mutations of BRAT1 were c.638_639insA (16 cases), c.1395G > A (5 cases) and c.294dupA (4 cases). Homozygous mutations in BRAT1 presented a more severe phenotype than those who are compound heterozygotes.
Conclusions
This is the first comprehensive systematic review to present quantitative data about clinical characteristics of BRAT1-related disease, which helps doctors to recognize and diagnose it easier.




Similar content being viewed by others
Availability of data and materials
Data from patients can be made available from the corresponding author after discussion with the Institutional Review Board.
References
Aglipay JA, Martin SA, Tawara H, Lee SW, Ouchi T (2006) ATM activation by ionizing radiation requires BRCA1-associated BAAT1. J Biol Chem 281(14):9710–9718. https://doi.org/10.1074/jbc.M510332200
Puffenberger EG, Jinks RN, Sougnez C, Cibulskis K, Willert RA, Achilly NP, Cassidy RP, Fiorentini CJ, Heiken KF, Lawrence JJ, Mahoney MH, Miller CJ, Nair DT, Politi KA, Worcester KN, Setton RA, Dipiazza R, Sherman EA, Eastman JT, Francklyn C, Robey-Bond S, Rider NL, Gabriel S, Morton DH, Strauss KA (2012) Genetic mapping and exome sequencing identify variants associated with five novel diseases. PLoS ONE 7(1):e28936. https://doi.org/10.1371/journal.pone.0028936
Saunders CJ, Miller NA, Soden SE, Dinwiddie DL, Noll A, Alnadi NA, Andraws N, Patterson ML, Krivohlavek LA, Fellis J, Humphray S, Saffrey P, Kingsbury Z, Weir JC, Betley J, Grocock RJ, Margulies EH, Farrow EG, Artman M, Safina NP, Petrikin JE, Hall KP, Kingsmore SF (2012) Rapid whole-genome sequencing for genetic disease diagnosis in neonatal intensive care units. Sci Transl Med. 4(154):154ra135. https://doi.org/10.1126/scitranslmed.3004041
Saitsu H, Yamashita S, Tanaka Y, Tsurusaki Y, Nakashima M, Miyake N, Matsumoto N (2014) Compound heterozygous BRAT1 mutations cause familial Ohtahara syndrome with hypertonia and microcephaly. J Hum Genet 59(12):687–690. https://doi.org/10.1038/jhg.2014.91
Hanes I, Kozenko M, Callen DJ (2015) Lethal neonatal rigidity and multifocal seizure syndrome–a misnamed disorder? Pediatr Neurol 53(6):535–540. https://doi.org/10.1016/j.pediatrneurol.2015.09.002
Mundy SA, Krock BL, Mao R, Shen JJ (2016) BRAT1-related disease–identification of a patient without early lethality. Am J Med Genet A 170(3):699–702. https://doi.org/10.1002/ajmg.a.37434
Srivastava S, Olson HE, Cohen JS, Gubbels CS, Lincoln S, Davis BT, Shahmirzadi L, Gupta S, Picker J, Yu TW, Miller DT, Soul JS, Poretti A, Naidu S (2016) BRAT1 mutations present with a spectrum of clinical severity. Am J Med Genet A 170(9):2265–2273. https://doi.org/10.1002/ajmg.a.37783
Scheffer IE, Boysen KE, Schneider AL, Myers CT, Mehaffey MG, Rochtus AM, Yuen YP, Ronen GM, Chak WK, Gill D, Poduri A, Mefford HC (2020) BRAT1 encephalopathy: a recessive cause of epilepsy of infancy with migrating focal seizures. Dev Med Child Neurol 62(9):1096–1099. https://doi.org/10.1111/dmcn.14428
Qi Y, Ji X, Ding H, Liu L, Zhang Y, Yin A (2022) Novel biallelic variant in the BRAT1 gene caused nonprogressive cerebellar ataxia syndrome. Front Genet 13:821587. https://doi.org/10.3389/fgene.2022.821587
Colak FK, Guleray N, Azapagasi E, Yazıcı MU, Aksoy E, Ceylan N (2020) An intronic variant in BRAT1 creates a cryptic splice site, causing epileptic encephalopathy without prominent rigidity. Acta Neurol Belg 120(6):1425–1432. https://doi.org/10.1007/s13760-020-01513-0
Moher D, Liberati A, Tetzlaff J, Altman DG, P. Group (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. J Clin Epidemiol 62(10):1006–1012. https://doi.org/10.1016/j.jclinepi.2009.06.005
Rim JH, Kim SH, Hwang IS, Kwon SS, Kim J, Kim HW, Cho MJ, Ko A, Youn SE, Kim J, Lee YM, Chung HJ, Lee JS, Kim HD, Choi JR, Lee ST, Kang HC (2018) Efficient strategy for the molecular diagnosis of intractable early-onset epilepsy using targeted gene sequencing. BMC Med Genom 11(1):6. https://doi.org/10.1186/s12920-018-0320-7
Horn D, Weschke B, Knierim E, Fischer-Zirnsak B, Stenzel W, Schuelke M, Zemojtel T (2016) BRAT1 mutations are associated with infantile epileptic encephalopathy, mitochondrial dysfunction, and survival into childhood. Am J Med Genet A 170(9):2274–2281. https://doi.org/10.1002/ajmg.a.37798
Mahjoub A, Cihlarova Z, Tétreault M, MacNeil L, Sondheimer N, Caldecott KW, Hanzlikova H, Yoon G (2019) Homozygous pathogenic variant in BRAT1 associated with nonprogressive cerebellar ataxia. Neurol Genet 5(5):e359. https://doi.org/10.1212/nxg.0000000000000359
Smith NJ, Lipsett J, Dibbens LM, Heron SE (2016) BRAT1-associated neurodegeneration: Intra-familial phenotypic differences in siblings. Am J Med Genet A 170(11):3033–3038. https://doi.org/10.1002/ajmg.a.37853
So EY, Ouchi T (2014) BRAT1 deficiency causes increased glucose metabolism and mitochondrial malfunction. BMC Cancer 14:548. https://doi.org/10.1186/1471-2407-14-548
Nesta AV, Tafur D, Beck CR (2021) Hotspots of human mutation. Trends Genet 37(8):717–729. https://doi.org/10.1016/j.tig.2020.10.003
Montgomery SB, Goode DL, Kvikstad E, Albers CA, Zhang ZD, Xinmeng Jasmine M, Ananda G, Howie B, Karczewski KJ, Smith KS, Anaya V, Richardson R, Davis J, C. Genomes Project, MacArthur DG, Sidow A, Duret L, Gerstein M, Makova KD, Marchini J, McVean G, Lunter G (2013) The origin, evolution, and functional impact of short insertion-deletion variants identified in 179 human genomes. Genome Res 23(5):749–761. https://doi.org/10.1101/gr.148718.112
Low LH, Chow YL, Li Y, Goh CP, Putz U, Silke J, Ouchi T, Howitt J, Tan SS (2015) Nedd4 family interacting protein 1 (Ndfip1) is required for ubiquitination and nuclear trafficking of BRCA1-associated ATM activator 1 (BRAT1) during the DNA damage response. J Biol Chem 290(11):7141–7150. https://doi.org/10.1074/jbc.M114.613687
Straussberg R, Ganelin-Cohen E, Goldberg-Stern H, Tzur S, Behar DM, Smirin-Yosef P, Salmon-Divon M, Basel-Vanagaite L (2015) Lethal neonatal rigidity and multifocal seizure syndrome–report of another family with a BRAT1 mutation. Eur J Paediatr Neurol 19(2):240–242. https://doi.org/10.1016/j.ejpn.2014.11.004
van de Pol LA, Wolf NI, van Weissenbruch MM, Stam CJ, Weiss JM, Waisfisz Q, Kevelam SH, Bugiani M, van de Kamp JM, van der Knaap MS (2015) Early-onset severe encephalopathy with epilepsy: the BRAT1 gene should be added to the list of causes. Neuropediatrics 46(6):392–400. https://doi.org/10.1055/s-0035-1564791
Fernández-Jaén A, Álvarez S, So EY, Ouchi T, Jiménez de la Peña M, Duat A, Fernández-Mayoralas DM, Fernández-Perrone AL, Albert J, Calleja-Pérez B (2016) Mutations in BRAT1 cause autosomal recessive progressive encephalopathy: report of a Spanish patient. Eur J Paediatr Neurol 20(3):421–425. https://doi.org/10.1016/j.ejpn.2016.02.009
Celik Y, Okuyaz C, Arslankoylu AE, Ceylaner S (2017) Lethal neonatal rigidity and multifocal seizure syndrome with a new mutation in BRAT1. Epilepsy Behav Case Rep 8:31–32. https://doi.org/10.1016/j.ebcr.2017.05.003
Au H, Kp S, Pk K, Ab J (2017) BRCA1-associated ataxia telangiectasia mutated activation-1 mutation: an addition to the early infantile epileptic encephalopathy panel. J Clin Neonatol 6:200–204. https://doi.org/10.4103/jcn.JCN_32_17
Oatts JT, Duncan JL, Hoyt CS, Slavotinek AM, Moore AT (2017) Inner retinal dystrophy in a patient with biallelic sequence variants in BRAT1. Ophthalmic Genet 38(6):559–561. https://doi.org/10.1080/13816810.2017.1290118
Szymańska K, Laure-Kamionowska M, Szczałuba K, Koppolu A, Furmanek M, Kuśmierska K, Boniel S, Płoski R, Rydzanicz M (2018) Clinico-pathological correlation in case of BRAT1 mutation. Folia Neuropathol 56(4):362–371. https://doi.org/10.5114/fn.2018.80870
Skafi O, Fawaz A, Merhi BA, Jouni H, Dabaja H (2018) Rigidity with multifocal seizure syndrome, lethal neonatal in a lebanese neonate. a rare case report. J Pediatr Disord Neonatal Care. https://doi.org/10.18875/2638-3608.1.106
Van Ommeren RH, Gao AF, Blaser SI, Chitayat DA, Hazrati LN (2018) BRAT1 mutation: the first reported case of chinese origin and review of the literature. J Neuropathol Exp Neurol 77(12):1071–1078. https://doi.org/10.1093/jnen/nly093
Valence S, Cochet E, Rougeot C, Garel C, Chantot-Bastaraud S, Lainey E, Afenjar A, Barthez MA, Bednarek N, Doummar D, Faivre L, Goizet C, Haye D, Heron B, Kemlin I, Lacombe D, Milh M, Moutard ML, Riant F, Robin S, Roubertie A, Sarda P, Toutain A, Villard L, Ville D, Billette de Villemeur T, Rodriguez D, Burglen L (2019) Exome sequencing in congenital ataxia identifies two new candidate genes and highlights a pathophysiological link between some congenital ataxias and early infantile epileptic encephalopathies. Genet Med 21(3):553–563. https://doi.org/10.1038/s41436-018-0089-2
Pourahmadiyan A, Heidari M, Shojaaldini Ardakani H, Noorian S, Savad S (2021) A novel pathogenic variant of BRAT1 gene causes rigidity and multifocal seizure syndrome, lethal neonatal. Int J Neurosci 131(9):875–878. https://doi.org/10.1080/00207454.2020.1759589
Balasundaram P, Fijas M, Nafday S (2021) A rare case of lethal neonatal rigidity and multi-focal seizure syndrome. Cureus 13(2):e13600. https://doi.org/10.7759/cureus.13600
Cornet MC, Morabito V, Lederer D, Glass HC, Ferrao Santos S, Numis AL, Ferriero DM, Sands TT, Cilio MR (2021) Neonatal presentation of genetic epilepsies: Early differentiation from acute provoked seizures. Epilepsia 62(8):1907–1920. https://doi.org/10.1111/epi.16957
Li W, Wu S, Xu H, Zhao X, Pan Y, Huang H, Lv H, Zhu X, Liu Y (2022) Novel variant in BRAT1 with the lethal neonatal rigidity and multifocal seizure syndrome. Pediatr Res 91(3):565–571. https://doi.org/10.1038/s41390-021-01468-9
Nuovo S, Baglioni V, De Mori R, Tardivo S, Caputi C, Ginevrino M, Micalizzi A, Masuelli L, Federici G, Casella A, Lorefice E, Anello D, Tolve M, Farini D, Bertini E, Zanni G, Travaglini L, Vasco G, Sette C, Carducci C, Valente EM, Leuzzi V (2022) Clinical variability at the mild end of BRAT1-related spectrum: evidence from two families with genotype-phenotype discordance. Hum Mutat 43(1):67–73. https://doi.org/10.1002/humu.24293
Fowkes R, Elwan M, Akay E, Mitchell CJ, Thomas RH, Lewis-Smith D (2022) A review of the clinical spectrum of BRAT1 disorders and case of developmental and epileptic encephalopathy surviving into adulthood. Epilepsy Behav Rep 19:100549. https://doi.org/10.1016/j.ebr.2022.100549
Funding
This work was supported by a grant from the National Natural Science Foundation of China (82202059).
Author information
Authors and Affiliations
Contributions
WJ K wrote the manuscript, supervised data and manuscript. XY C and C L collected and analyzed related articles and data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kong, W., Cao, X. & Lu, C. Clinical characteristics of BRAT1-related disease: a systematic literature review. Acta Neurol Belg (2024). https://doi.org/10.1007/s13760-024-02507-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13760-024-02507-y