Abstract
Purpose
We sought to develop and validate an Anticipated Surveillance Requirement Prediction Instrument (ASRI) for prediction of prolonged postanesthesia care unit length of stay (PACU-LOS, more than four hours) after ambulatory surgery.
Methods
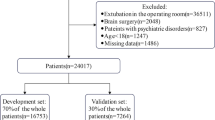
We analyzed hospital registry data from patients who received anesthesia care in ambulatory surgery centres (ASCs) of university-affiliated hospital networks in New York, USA (development and internal validation cohort [n = 183,711]) and Massachusetts, USA (validation cohort [n = 148,105]). We used stepwise backwards elimination to create ASRI.
Results
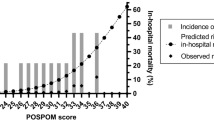
The model showed discriminatory ability in the development, internal, and external validation cohorts with areas under the receiver operating characteristic curve of 0.82 (95% confidence interval [CI], 0.82 to 0.83), 0.82 (95% CI, 0.81 to 0.83), and 0.80 (95% CI, 0.79 to 0.80), respectively. In cases started in the afternoon, ASRI scores ≥ 43 had a total predicted risk for PACU stay past 8 p.m. of 32% (95% CI, 31.1 to 33.3) vs 8% (95% CI, 7.9 to 8.5) compared with low score values (P-for-interaction < 0.001), which translated to a higher direct PACU cost of care of USD 207 (95% CI, 194 to 2,019; model estimate, 1.68; 95% CI, 1.64 to 1.73; P < 0.001) The effects of using the ASRI score on PACU use efficiency were greater in a free-standing ASC with no limitations on PACU bed availability.
Conclusion
We developed and validated a preoperative prediction tool for prolonged PACU-LOS after ambulatory surgery that can be used to guide scheduling in ambulatory surgery to optimize PACU use during normal work hours, particularly in settings without limitation of PACU bed availability.
Résumé
Objectif
Nous avons cherché à mettre au point et à valider un Instrument de prédiction anticipée des besoins de surveillance pour anticiper toute prolongation de la durée de séjour en salle de réveil (plus de quatre heures) après chirurgie ambulatoire.
Méthode
Nous avons analysé les données enregistrées dans le registre de l’hôpital des patient·es qui ont reçu des soins d’anesthésie dans des centres de chirurgie ambulatoire (CCA) des réseaux hospitaliers affiliés à une université à New York, aux États-Unis (cohorte de développement et de validation interne [n = 183 711]) et au Massachusetts, États-Unis (cohorte de validation [n = 148 105]). Nous avons utilisé un procédé d’élimination progressive régressive pour créer notre instrument de prédiction.
Résultats
Le modèle a montré une capacité discriminatoire dans les cohortes de développement, de validation interne et de validation externe, avec des surfaces sous la courbe de fonction d’efficacité de l’opérateur (ROC) de 0,82 (intervalle de confiance [IC] à 95 %, 0,82 à 0,83), 0,82 (IC 95 %, 0,81 à 0,83), et 0,80 (IC 95 %, 0,79 à 0,80), respectivement. Dans les cas commencés en après-midi, les scores sur notre instrument de prédiction ≥ 43 montraient un risque total prédit de séjour en salle de réveil après 20 h de 32 % (IC 95 %, 31,1 à 33,3) vs 8 % (IC 95 %, 7,9 à 8,5) comparativement aux valeurs de score faibles (P-pour-interaction < 0,001), ce qui s’est traduit par une augmentation de 207 USD du coût direct des soins en salle de réveil (IC 95 %, 194 à 2019; estimation du modèle, 1,68; IC 95 %, 1,64 à 1,73; P < 0,001). Les effets de l’utilisation du score de notre instrument de prédiction sur l’efficacité d’utilisation de la salle de réveil étaient plus importants dans un CCA autonome sans limitation dans la disponibilité des lits en salle de réveil.
Conclusion
Nous avons mis au point et validé un outil de prédiction préopératoire de la prolongation de la durée de séjour en salle de réveil après une chirurgie ambulatoire qui peut être utilisé pour guider la planification en chirurgie ambulatoire afin d’optimiser l’utilisation de la salle de réveil pendant les heures normales de travail, en particulier dans les milieux sans limitation de disponibilité des lits en salle de réveil.




Similar content being viewed by others
References
Hall MJ, Schwartzman A, Zhang J, Liu X. Ambulatory surgery data from hospitals and ambulatory surgery centers: United States, 2010. Natl Health Stat Report 2017; 1–15.
Munnich EL, Richards MR. Long-run growth of ambulatory surgery centers 1990–2015 and Medicare payment policy. Health Serv Res 2022; 57: 66–71. https://doi.org/10.1111/1475-6773.13707
Junger A, Klasen J, Benson M, et al. Factors determining length of stay of surgical day-case patients. Eur J Anaesthesiol 2001; 18: 314–21. https://doi.org/10.1046/J.0265-0215.2000.00837.X
Kent C, Metzner J, Bollag L. An analysis of risk factors and adverse events in ambulatory surgery. Ambul Anesth 2014; 1: 3–10. https://doi.org/10.2147/aa.s53280
Van Caelenberg E, De Regge M, Eeckloo K, Coppens M. Analysis of failed discharge after ambulatory surgery: unanticipated admission. Acta Chir Belg 2019; 119: 139–45. https://doi.org/10.1080/00015458.2018.1477488
Coley KC, Williams BA, DaPos SV, Chen C, Smith RB. Retrospective evaluation of unanticipated admissions and readmissions after same day surgery and associated costs. J Clin Anesth 2002; 14: 349–53. https://doi.org/10.1016/S0952-8180(02)00371-9
Dexter F, Tinker JH. Analysis of strategies to decrease postanesthesia care unit costs. Anesthesiology 1995; 82: 94–101. https://doi.org/10.1097/00000542-199501000-00013
Imasogie N, Chung F. Risk factors for prolonged stay after ambulatory surgery: economic considerations. Curr Opin Anaesthesiol 2002; 15: 245–9. https://doi.org/10.1097/00001503-200204000-00017
Azimaraghi O, Ahrens E, Wongtangman K, et al. Association of sugammadex reversal of neuromuscular block and postoperative length of stay in the ambulatory care facility: a multicentre hospital registry study. Br J Anaesth 2023; 130: 296–304. https://doi.org/10.1016/j.bja.2022.10.044
von Elm E, Altman DG, Egger M, et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol 2008; 61: 344–9. https://doi.org/10.1016/j.jclinepi.2007.11.008
Shaikh SI, Nagarekha D, Hegade G, Marutheesh M. Postoperative nausea and vomiting: a simple yet complex problem. Anesth Essays Res 2016; 10: 388–96. https://doi.org/10.4103/0259-1162.179310
Gabriel RA, Waterman RS, Kim J, Ohno-Machado L. A predictive model for extended postanesthesia care unit length of stay in outpatient surgeries. Anesth Analg 2017; 124: 1529–36. https://doi.org/10.1213/ane.0000000000001827
Whippey A, Kostandoff G, Paul J, Ma J, Thabane L, Ma HK. Predictors of unanticipated admission following ambulatory surgery: a retrospective case-control study. Can J Anesth 2013; 60: 675–83. https://doi.org/10.1007/s12630-013-9935-5
Ganter MT, Blumenthal S, Dübendorfer S, et al. The length of stay in the post-anaesthesia care unit correlates with pain intensity, nausea and vomiting on arrival. Perioper Med 2014; 3: 10. https://doi.org/10.1186/s13741-014-0010-8
Teja B, Raub D, Friedrich S, et al. Incidence, prediction, and causes of unplanned 30-day hospital admission after ambulatory procedures. Anesth Analg 2020; 131: 497–507. https://doi.org/10.1213/ANE.0000000000004852
Chung F, Mezei G. Factors contributing to a prolonged stay after ambulatory surgery. Anesth Analg 1999; 89: 1352–9. https://doi.org/10.1097/00000539-199912000-00004
Gan TJ, Diemunsch P, Habib AS, et al. Consensus guidelines for the management of postoperative nausea and vomiting. Anesth Analg 2014; 118: 85–113. https://doi.org/10.1213/ane.0000000000000002
Collins L, Halwani A, Vaghadia H. Impact of a regional anesthesia analgesia program for outpatient foot surgery. Can J Anesth 1999; 46: 840–5. https://doi.org/10.1007/bf03012972
Waddle JP, Evers AS, Piccirillo JF. Postanesthesia care unit length of stay: quantifying and assessing dependent factors. Anesth Analg 1998; 87: 628–33. https://doi.org/10.1213/00000539-199809000-00026
Chatterjee S, Rudra A, Sengupta S. Current concepts in the management of postoperative nausea and vomiting. Anesthesiol Res Pract 2011; 2011: 748031. https://doi.org/10.1155/2011/748031
Shin CH, Grabitz SD, Timm FP, et al. Development and validation of a Score for preoperative prediction of obstructive sleep apnea (SPOSA) and its perioperative outcomes. BMC Anesthesiol 2017; 17: 71. https://doi.org/10.1186/s12871-017-0361-z
Hosmer DW, Lemeshow S. Applied Logistic Regression. Hoboken: John Wiley & Sons, Inc.: 2005. https://doi.org/10.1002/0471722146
Daggy J, Lawley M, Willis D, et al. Using no-show modeling to improve clinic performance. Health Informatics J 2010; 16: 246–59. https://doi.org/10.1177/1460458210380521
Janssen KJ, Moons KG, Kalkman CJ, Grobbee DE, Vergouwe Y. Updating methods improved the performance of a clinical prediction model in new patients. J Clin Epidemiol 2008; 61: 76–86. https://doi.org/10.1016/j.jclinepi.2007.04.018
Awad IT, Chung F. Factors affecting recovery and discharge following ambulatory surgery. Can J Anesth 2006; 53: 858–72. https://doi.org/10.1007/BF03022828
Picard L, Duceau B, Cambriel A, et al. Risk factors for prolonged time to hospital discharge after ambulatory cholecystectomy under general anaesthesia. A retrospective cohort study. Int J Surg 2022; 104: 106706. https://doi.org/10.1016/j.ijsu.2022.106706
Williams BA, DeRiso BM, Figallo CM, et al. Benchmarking the perioperative process: III. Effects of regional anesthesia clinical pathway techniques on process efficiency and recovery profiles in ambulatory orthopedic surgery. J Clin Anesth 1998; 10: 570–8. https://doi.org/10.1016/s0952-8180(98)00083-x
Klein SM, Greengrass RA, Gleason DH, Nunley JA, Steele SM. Major ambulatory surgery with continuous regional anesthesia and a disposable infusion pump. Anesthesiology 1999; 91: 563–5. https://doi.org/10.1097/00000542-199908000-00034
Wachtendorf LJ, Tartler TM, Ahrens E, et al. Comparison of the effects of sugammadex versus neostigmine for reversal of neuromuscular block on hospital costs of care. Br J Anaesth 2023; 130: 133–41. https://doi.org/10.1016/j.bja.2022.10.015
Joshi GP. Efficiency in ambulatory surgery center. Curr Opin Anaesthesiol 2008; 21: 695–8. https://doi.org/10.1097/ACO.0b013e328311d1b2
Mingus ML, Bodian CA, Bradford CN, Eisenkraft JB. Prolonged surgery increases the likelihood of admission of scheduled ambulatory surgery patients. J Clin Anesth 1997; 9: 446–50. https://doi.org/10.1016/S0952-8180(97)00098-6
Wongtangman K, Aasman B, Garg S, et al. Development and validation of a machine learning ASA-score to identify candidates for comprehensive preoperative screening and risk stratification. J Clin Anesth 2023; 87: 111103. https://doi.org/10.1016/j.jclinane.2023.111103
Author contributions
Samuel Rupp, Elena Ahrens, and Omid Azimaraghi helped conceptualize the paper, performed the literature review, wrote the initial draft of manuscript, and read the final manuscript. Samuel Rupp, Elena Ahrens, as well as Maira Rudolph contributed equally to this manuscript. Maximilian S. Schaefer, Philipp Fassbender, Carina P. Himes, Preeti Anand, Parsa Mirhaji, Richard Smith, and Jeffrey Freda gave suggestions on data analysis, helped interpret the data, reviewed the manuscript, edited and modified drafts, and read the final manuscript. Matthias Eikermann is the guarantor of the study and takes full responsibility for the integrity of the work as a whole. Karuna Wongtangman helped conceptualize the paper, gave suggestions on data analysis, reviewed the manuscript, edited and modified drafts, and read the final manuscript.
Acknowledgements
We are grateful to Shweta Grag, Annika Witt, and Maya Doehne for their help with variable extraction and data cleaning.
Disclosures
Maximilian S. Schaefer received funding for investigator-initiated studies from Merck & Co., which do not pertain to this manuscript. He is an Associate Editor for BMC Anesthesiology. He received honoraria for presentations from Fisher & Paykel Healthcare and Mindray Medical International Ltd.
Funding statement
The study was funded by the Departments of Anesthesiology, Surgical Services, and Center for Health Data Innovations, Montefiore Health System (Bronx, NY, USA). Funds were allotted to support time and effort of study personnel. The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and the decision to submit the manuscript for publication.
Prior conference presentations
A conference abstract highlighting preliminary results of this research was presented at the 2022 Annual Meeting of the American Society of Anesthesiologists (21–25 October, New Orleans, LA, USA).
Data availability
Due to the sensitive nature of the data collected for this study, requests to access the data set from qualified researchers trained in human research and confidentiality may be sent to Matthias Eikermann at meikermann@montefiore.org.
Editorial responsibility
This submission was handled by Dr. Stephan K. W. Schwarz, Editor-in-Chief, Canadian Journal of Anesthesia/Journal canadien d’anesthésie.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rupp, S., Ahrens, E., Rudolph, M.I. et al. Development and validation of an instrument to predict prolonged length of stay in the postanesthesia care unit following ambulatory surgery. Can J Anesth/J Can Anesth 70, 1939–1949 (2023). https://doi.org/10.1007/s12630-023-02604-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-023-02604-1