Abstract
Background
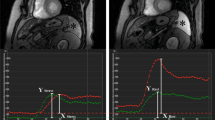
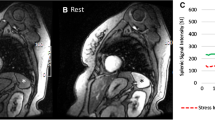
Splenic switch-off (SSO) is a phenomenon describing a decrease in splenic radiotracer uptake after vasodilatory stress. We aimed to assess the diagnostic utility of regadenoson-induced SSO.
Methods
We included consecutive patients who had clinically indicated Regadenoson Rb-82 PET-MPI for suspected CAD. This derivation cohort (no perfusion defects and myocardial flow reserves (MFR) ≥ 2) was used to calculate the splenic response ratio (SRR). The validation cohort was defined as patients who underwent both PET-MPI studies and invasive coronary angiography (ICA).
Results
The derivation cohort (n = 100, 57.4 ± 11.6 years, 77% female) showed a decrease in splenic uptake from rest to stress (79.9 ± 16.8 kBq⋅mL vs 69.1 ± 16.2 kBq⋅mL, P < .001). From the validation cohort (n = 315, 66.3 ± 10.4 years, 67% male), 28% (via SRR = 0.88) and 15% (visually) were classified as splenic non-responders. MFR was lower in non-responders (SRR; 1.55 ± 0.65 vs 1.76 ± 0.78, P = .02 and visually; 1.18 ± 0.33 vs 1.79 ± 0.77, P < .001). Based on ICA, non-responders were more likely to note obstructive epicardial disease with normal PET scans especially in patients with MFR < 1.5 (SRR; 61% vs 34% P = .05 and visually; 68% vs 33%, P = .01).
Conclusion
Lack of splenic response based on visual or quantitative assessment of SSO may be used to identify an inadequate vasodilatory response.





Similar content being viewed by others
Abbreviations
- CAD:
-
Coronary artery disease
- PET:
-
Positron Emission Tomography
- MBF:
-
Myocardial blood flow
- MFR:
-
Myocardial flow reserve
- MPI:
-
Myocardial perfusion imaging
- SSO:
-
Splenic switch-off
- SSR:
-
Splenic response ratio
- Rb:
-
Rubidium
- HD:
-
Hemodynamics
- ICA:
-
Invasive coronary angiography
References
Reeves RA, Halpern EJ, Rao VM. Cardiac imaging trends from 2010 to 2019 in the medicare population. Radiology 2021;3:e210156. https://doi.org/10.1148/ryct.2021210156.
Driessen RS, Danad I, Stuijfzand WJ, et al. Comparison of coronary computed tomography angiography, Fractional Flow reserve, and Perfusion Imaging for ischemia diagnosis. J Am Coll Cardiol 2019;73:161‐73. https://doi.org/10.1016/j.jacc.2018.10.056.
Zoghbi G, Iskandrian A. Selective adenosine agonists and myocardial perfusion imaging. J Nucl Cardiol 2012;19:126‐41. https://doi.org/10.1007/s12350-011-9474-9.
Al Jaroudi W, Iskandrian AE. Regadenoson: A new myocardial stress agent. J Am Coll Cardiol 2009;54:1123‐30. https://doi.org/10.1016/j.jacc.2009.04.089.
Johnson SG, Peters S. Advances in pharmacologic stress agents: Focus on regadenoson. J Nucl Med Technol 2010;38:163‐71. https://doi.org/10.2967/jnmt.109.065581.
Symphony Health Solutions. Lexiscan—symphony health solutions. https://lexiscan.com/OperationalFlexibility/Overview. Updated 2022.
Elkholy KO, Hegazy O, Okunade A, Aktas S, Ajibawo T. Regadenoson stress testing: A comprehensive review with a focused update. Curēus 2021;13:e12940. https://doi.org/10.7759/cureus.12940.
Patel KK, Spertus JA, Chan PS, et al. Myocardial blood flow reserve assessed by positron emission tomography myocardial perfusion imaging identifies patients with a survival benefit from early revascularization. Eur Heart J 2020;41:759‐68. https://doi.org/10.1093/eurheartj/ehz389.
Mc Ardle BA, Dowsley TF, deKemp RA, Wells GA, Beanlands RS. Does rubidium-82 PET have superior accuracy to SPECT perfusion imaging for the diagnosis of obstructive coronary disease? A systematic review and meta-analysis. J Am Coll Cardiol 2012;60:1828‐37. https://doi.org/10.1016/j.jacc.2012.07.038.
Go RT, Marwick TH, MacIntyre WJ, et al. A prospective comparison of rubidium-82 PET and thallium-201 SPECT myocardial perfusion imaging utilizing a single dipyridamole stress in the diagnosis of coronary artery disease. J Nucl Med 1990;31:1899‐905.
Hage FG, Dean P, Bhatia V, Iqbal F, Heo J, Iskandrian AE. The prognostic value of the heart rate response to adenosine in relation to diabetes mellitus and chronic kidney disease. Am Heart J 2011;162:356‐62. https://doi.org/10.1016/j.ahj.2011.05.014.
Mishra RK, Dorbala S, Logsetty G, et al. Quantitative relation between hemodynamic changes during intravenous adenosine infusion and the magnitude of coronary hyperemia: Implications for myocardial perfusion imaging. J Am Coll Cardiol 2005;45:553‐8. https://doi.org/10.1016/j.jacc.2004.10.064.
Norlén K. Central and regional haemodynamics during controlled hypotension produced by adenosine, sodium nitroprusside and nitroglycerin. Studies in the pig. Br J Anaesth 1988;61:186‐93. https://doi.org/10.1093/bja/61.2.186.
Hunter CR, Hill J, Ziadi MC, Beanlands RS, deKemp RA. Biodistribution and radiation dosimetry of (82)rb at rest and during peak pharmacological stress in patients referred for myocardial perfusion imaging. Eur J Nucl Med Mol Imaging 2015;42:1032‐42. https://doi.org/10.1007/s00259-015-3028-3.
Sandler MP, Kronenberg MW, Forman MB, Wolfe OH, Clanton JA, Partain CL. Dynamic fluctuations in blood and spleen radioactivity: Splenic contraction and relation to clinical radionuclide volume calculations. J Am Coll Cardiol 1984;3:1205‐11. https://doi.org/10.1016/s0735-1097(84)80178-3.
Bakula A, Patriki D, von Felten E, et al. Splenic switch-off as a novel marker for adenosine response in nitrogen-13 ammonia PET myocardial perfusion imaging: Cross-validation against CMR using a hybrid PET/MR device. J Nucl Cardiol 2020. https://doi.org/10.1007/s12350-020-02448-y.
Patriki D, von Felten E, Bakula A, et al. Splenic switch-off as a predictor for coronary adenosine response: Validation against 13N-ammonia during co-injection myocardial perfusion imaging on a hybrid PET/CMR scanner. J Cardiovasc Magn Reson 2021;23:196062. https://doi.org/10.5167/uzh-196062.
Manisty C, Ripley DP, Herrey AS, et al. Splenic switch-off: A tool to assess stress adequacy in adenosine perfusion cardiac MR imaging. Radiology 2015;276:732‐40. https://doi.org/10.1148/radiol.2015142059.
Hosking A, Koulouroudias M, Zemrak F, et al. Evaluation of splenic switch off in a tertiary imaging centre: Validation and assessment of utility. Eur Heart J Cardiovasc Imaging 2017;18:1216‐21. https://doi.org/10.1093/ehjci/jew205.
Bami K, Tewari S, Guirguis F, et al. Prognostic utility of splenic response ratio in dipyridamole PET myocardial perfusion imaging. J Nucl Cardiol 2018;26:1888‐97. https://doi.org/10.1007/s12350-018-1269-9.
Dilsizian V, Bacharach SL, Beanlands RS, et al. ASNC imaging guidelines/SNMMI procedure standard for positron emission tomography (PET) nuclear cardiology procedures. J Nucl Cardiol 2016;23:1187‐226. https://doi.org/10.1007/s12350-016-0522-3.
Nesterov SV, Deshayes E, Sciagrà R, et al. Quantification of myocardial blood flow in absolute terms using (82)rb PET imaging: The RUBY-10 study. JACC Cardiovasc Imaging 2014;7:1119‐27. https://doi.org/10.1016/j.jcmg.2014.08.003.
El Fakhri G, Sitek A, Guérin B, Kijewski MF, Di Carli MF, Moore SC. Quantitative dynamic cardiac 82Rb PET using generalized factor and compartment analyses. J Nucl Med 2005;46:1264‐71.
Youden WJ. Index for rating diagnostic tests. Cancer 1950;3:32‐5. https://doi.org/10.1002/1097-0142(1950)3:13.0.co;2-3.
2013 american society of nuclear cardiology/MedAxiom nuclear survey. J Nucl Cardiol 2014;21:5-88. https://doi.org/10.1007/s12350-014-9862-z
Walkden M, Bryant J, Abbas A, Harden S, Shambrook J, Peebles C. Turning up the adenosine turns off the spleen. J Cardiovasc Magn Reson 2015. https://doi.org/10.1186/1532-429X-17-S1-T1.
Mustafa SJ, Morrison RR, Teng B, Pelleg A. Adenosine receptors and the heart: Role in regulation of coronary blood flow and cardiac electrophysiology. Handb Exp Pharmacol 2009. https://doi.org/10.1007/978-3-540-89615-9_6.
Fozard JR, Milavec-Krizman M. Contraction of the rat isolated spleen mediated by adenosine A1 receptor activation. Br J Pharmacol 1993;109:1059‐63. https://doi.org/10.1111/j.1476-5381.1993.tb13729.x.
Kuijpers D, van Dijk R, van Assen M, et al. Disagreement between splenic switch-off and myocardial T1-mapping after caffeine intake. Int J Cardiovasc Imaging 2018;34:625‐32. https://doi.org/10.1007/s10554-017-1274-0.
Disclosures
Dr Al-mallah receives research support from Siemense unrelated to this work. All other authors declare no relevant financial or non-financial interests to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors of this article have provided a PowerPoint file, available for download at SpringerLink, which summarises the contents of the paper and is free for re-use at meetings and presentations. Search for the article DOI on SpringerLink.com.
The authors have also provided an audio summary of the article, which is available to download as ESM, or to listen to via the JNC/ASNC Podcast.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saad, J.M., Ahmed, A.I., Han, Y. et al. Splenic switch-off in regadenoson 82Rb-PET myocardial perfusion imaging: assessment of clinical utility. J. Nucl. Cardiol. 30, 1484–1496 (2023). https://doi.org/10.1007/s12350-022-03158-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-022-03158-3