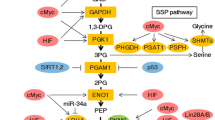
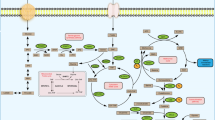
Abstract
As a multi-functional cellular organelle, mitochondrial metabolic reprogramming is well recognized as a hallmark of cancer. The center of mitochondrial metabolism is oxidative phosphorylation (OXPHOS), in which cells use enzymes to oxidize nutrients, thereby converting the chemical energy to the biological energy currency ATPs. OXPHOS also creates the mitochondrial membrane potential and serve as the driving force of other mitochondrial metabolic pathways and experiences significant reshape in the different stages of tumor progression. In this minireview, we reviewed the major mitochondrial pathways that are connected to OXPHOS and are affected in cancer cells. In addition, we summarized the function of novel bio-active molecules targeting mitochondrial metabolic processes such as OXPHOS, mitochondrial membrane potential and mitochondrial dynamics. These molecules exhibit intriguing preclinical and clinical results and have been proven to be promising antitumor candidates in recent studies.

Similar content being viewed by others
References
Bonnay, F., Veloso, A., Steinmann, V., Köcher, T., Deniz Abdusselamoglu, M., Bajaj, S., Rivelles, E., Landskron, L., Esterbauer, H., Zinzen, R. P., & Knoblich, J. A. (2020). Oxidative metabolism drives immortalization of neural stem cells during tumorigenesis. Cell, 182(6), 1490. https://doi.org/10.1016/j.cell.2020.07.039
Chinopoulos, C., & Seyfried, T. N. (2018). Mitochondrial substrate-level phosphorylation as energy source for glioblastoma: Review and hypothesis. The American Society of Neurochemistry. https://doi.org/10.1177/1759091418818261
Cooper, M. G. (2000). The mechanism of oxidative phosphorylation. Sinauer Associates.
DeBerardinis, R. J., Mancuso, A., Daikhin, E., Nissim, I., Yudkoff, M., Wehrli, S., & Thompson, C. B. (2007). Beyond aerobic glycolysis: Transformed cells can engage in glutamine metabolism that exceeds the requirement for protein and nucleotide synthesis. Proceedings of the National Academy of Sciences of the United States of America, 104(49), 19345. https://doi.org/10.1073/pnas.0709747104
Degli Esposti, M. (1998). Inhibitors of NADH-ubiquinone reductase: An overview. Biochimica Et Biophysica Acta (BBA) - Bioenergetics, 1364(2), 222. https://doi.org/10.1016/s0005-2728(98)00029-2
Esparza-Moltó, P. B., & Cuezva, J. M. (2018). The role of mitochondrial H +-ATP synthase in cancer. Frontiers in Oncology. https://doi.org/10.3389/fonc.2018.00053
Fang, Y., & Zhang, Z. (2020). Arsenic trioxide as a novel anti-glioma drug: A review. Cellular & Molecular Biology Letters. https://doi.org/10.1186/s11658-020-00236-7
Fiorillo, M., Lamb, R., Tanowitz, H. B., Cappello, A. R., Martinez-Outschoorn, U. E., Sotgia, F., & Lisanti, M. P. (2016a). Bedaquiline, an FDA-approved antibiotic, inhibits mitochondrial function and potently blocks the proliferative expansion of stem-like cancer cells (CSCs). Aging, 8(8), 1593. https://doi.org/10.18632/aging.100983
Fiorillo, M., Lamb, R., Tanowitz, H. B., Mutti, L., Krstic-Demonacos, M., Rita Cappello, A., Martinez-Outschoorn, U. E., Sotgia, F., & Lisanti, M. P. (2016b). Repurposing atovaquone: Targeting mitochondrial complex III and OXPHOS to eradicate cancer stem cells. Oncotarget, 7(23), 34084. https://doi.org/10.18632/oncotarget.9122
Guièze, R., Liu, V. M., Rosebrock, D., Jourdain, A. A., Hernández-Sánchez, M., Martinez Zurita, A., Sun, J., Ten Hacken, E., Baranowski, K., Thompson, P. A., Heo, J.-M., Cartun, Z., Aygün, O., BryanIorgulescu, J., Zhang, W., Notarangelo, G., Livitz, D., Li, S., Davids, M. S., … Wu, C. J. (2019). Mitochondrial reprogramming underlies resistance to BCL-2 inhibition in lymphoid malignancies. Cancer Cell, 36(4), 369. https://doi.org/10.1016/j.ccell.2019.08.005
Guntuku, L., Naidu, V. G., & Yerra, V. G. (2016). Mitochondrial dysfunction in gliomas: Pharmacotherapeutic potential of natural compounds. Current Neuropharmacology, 14(6), 567. https://doi.org/10.2174/1570159x14666160121115641
Janiszewska, M., Suvà, M. L., Riggi, N., Houtkooper, R. H., Auwerx, J., Clément-Schatlo, V., Radovanovic, I., Rheinbay, E., Provero, P., & Stamenkovic, I. (2012). Imp2 controls oxidative phosphorylation and is crucial for preserving glioblastoma cancer stem cells. Genes & Development, 26(17), 1926. https://doi.org/10.1101/gad.188292.112
Kanderi, T., & Gupta, V. (2020). Glioblastoma multiforme. Statpearls Publishing.
Kuramoto, K., Suzuki, S., Sakaki, H., Takeda, H., Sanomachi, T., Seino, S., Narita, Y., Kayama, T., Kitanaka, C., & Okada, M. (2017). Licochalcone A specifically induces cell death in glioma stem cells via mitochondrial dysfunction. FEBS Open Bio, 7(6), 835. https://doi.org/10.1002/2211-5463.12226
Kuramoto, K., Yamamoto, M., Suzuki, S., Sanomachi, T., Togashi, K., Seino, S., Kitanaka, C., & Okada, M. (2020). Verteporfin inhibits oxidative phosphorylation and induces cell death specifically in glioma stem cells. The FEBS Journal, 287(10), 2023. https://doi.org/10.1111/febs.15187
Lee, K. S., Wu, Z., Song, Y., Mitra, S. S., Feroze, A. H., Cheshier, S. H., & Lu, B. (2013). Roles of PINK1, mTORC2, and mitochondria in preserving brain tumor-forming stem cells in a noncanonical notch signaling pathway. Genes & Development, 27(24), 2642. https://doi.org/10.1101/gad.225169.113
Legros, F., Lombès, A., Frachon, P., & Rojo, M. (2002). Mitochondrial fusion in human cells is efficient, requires the inner membrane potential, and is mediated by mitofusins. Molecular Biology of the Cell, 13(12), 4343. https://doi.org/10.1091/mbc.e02-06-0330
Liesa, M., & Shirihai, O. S. (2013). Mitochondrial dynamics in the regulation of nutrient utilization and energy expenditure. Cell Metabolism, 17(4), 491. https://doi.org/10.1016/j.cmet.2013.03.002
Mi-Ichi, F., Miyadera, H., Kobayashi, T., Takamiya, S., Waki, S., Iwata, S., Shibata, S., & Kita, K. (2005). Parasite mitochondria as a target of chemotherapy: Inhibitory effect of licochalcone A on the Plasmodium falciparum respiratory chain. Annals of the New York Academy of Sciences. https://doi.org/10.1196/annals.1352.037
Mishra, P., & Chan, D. C. (2016). Metabolic regulation of mitochondrial dynamics. The Journal of Cell Biology, 212(4), 379. https://doi.org/10.1083/jcb.201511036
Muz, B., de la Puente, P., Azab, F., & Azab, A. K. (2015). The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy. Hypoxia (auckland). https://doi.org/10.2147/HP.S93413
Neupane, P., Bhuju, S., Thapa, N., & Bhattarai, H. K. (2019). ATP synthase: Structure function and inhibition. Biomolecular Concepts, 10(1), 1. https://doi.org/10.1515/bmc-2019-0001
Ralph, S. J., Moreno-Sánchez, R., Neuzil, J., & Rodríguez-Enríquez, S. (2011). Inhibitors of succinate: Quinone reductase/complex II regulate production of mitochondrial reactive oxygen species and protect normal cells from ischemic damage but induce specific cancer cell death. Pharmaceutical Research, 28(11), 2695. https://doi.org/10.1007/s11095-011-0566-7
Ramamoorthy, M. D., Kumar, A., Ayyavu, M., & Dhiraviam, K. N. (2018). Reserpine induces apoptosis and cell cycle arrest in hormone independent prostate cancer cells through mitochondrial membrane potential failure. Anti-Cancer Agents in Medicinal Chemistry, 18(9), 1313. https://doi.org/10.2174/1871520618666180209152215
Seyfried, T. N., & Mukherjee, P. (2005). Targeting energy metabolism in brain cancer: Review and hypothesis. Nutrition & Metabolism. https://doi.org/10.1186/1743-7075-2-30
Shi, Y., Lim, S. K., Liang, Q., Iyer, S. V., Wang, H. Y., Wang, Z., Xie, X., Sun, D., Chen, Y. J., Tabar, V., Gutin, P., Williams, N., De Brabander, J. K., & Parada, L. F. (2019). Gboxin is an oxidative phosphorylation inhibitor that targets glioblastoma. Nature, 567(7748), 341. https://doi.org/10.1038/s41586-019-0993-x
Strickland, M., & Stoll, E. A. (2017). Metabolic reprogramming in glioma. Frontiers in Cell and Developmental Biology. https://doi.org/10.3389/fcell.2017.00043
Vander Heiden, M. G., Cantley, L. C., & Thompson, C. B. (2009). Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science (new York), 324(5930), 1029. https://doi.org/10.1126/science.1160809
Voet, D., Voet, J. G., & Pratt, C. W. (2006). Fundamentals of biochemistry (2nd ed.). Wiley.
Wan, Y. Y., Zhang, J. F., Yang, Z. J., Jiang, L. P., Wei, Y. F., Lai, Q. N., Wang, J. B., Xin, H. B., & Han, X. J. (2014). Involvement of Drp1 in hypoxia-induced migration of human glioblastoma U251 cells. Oncology Reports, 32(2), 619. https://doi.org/10.3892/or.2014.3235
Warburg, O., Wind, F., & Negelein, E. (1927). The metabolism of tumors in the body. The Journal of General Physiology, 8(6), 519. https://doi.org/10.1085/jgp.8.6.519
Xie, Q., Wu, Q., Horbinski, C. M., Flavahan, W. A., Yang, K., Zhou, W., Dombrowski, S. M., Huang, Z., Fang, X., Shi, Y., Ferguson, A. N., Kashatus, D. F., Bao, S., & Rich, J. N. (2015). Mitochondrial control by DRP1 in brain tumor initiating cells. Nature Neuroscience, 18(4), 501. https://doi.org/10.1038/nn.3960
Author information
Authors and Affiliations
Contributions
ZW and RL: Conception and design, Literature search, ZW, WH, RL: Manuscript preparation and Final approval.
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, Z., Ho, W.S. & Lu, R. Targeting Mitochondrial Oxidative Phosphorylation in Glioblastoma Therapy. Neuromol Med 24, 18–22 (2022). https://doi.org/10.1007/s12017-021-08678-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-021-08678-8