Abstract
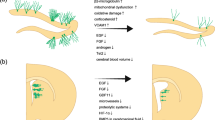
Traditionally, the primary role of the meninges is thought to be structural, i.e., to act as a surrounding membrane that contains and cushions the brain with cerebrospinal fluid. During development, the meninges is formed by both mesenchymal and neural crest cells. There is now emerging evidence that subsets of undifferentiated stem cells might persist in the adult meninges. In this mini-review, we survey representative studies of brain-meningeal interactions and discuss the hypothesis that the meninges are not just protective membranes, but instead contain multiplex stem cell subsets that may contribute to central nervous system (CNS) homeostasis. Further investigations into meningeal multipotent cells may reveal a “hidden” target for promoting neurovascular remodeling and repair after CNS injury and disease.


Similar content being viewed by others
References
Achilleos, A., & Trainor, P. A. (2012). Neural crest stem cells: discovery, properties and potential for therapy. Cell Research, 22, 288–304
Arac, A., Grimbaldeston, M. A., Nepomuceno, A. R., Olayiwola, O., Pereira, M. P., Nishiyama, Y., Tsykin, A., Goodall, G.J., Schlecht, U., Vogel, H., & Tsai, M. (2014). Evidence that meningeal mast cells can worsen stroke pathology in mice. American Journal of Pathology, 184, 2493–2504
Biernaskie, J., & Miller, F. D. (2010). White matter repair: skin-derived precursors as a source of myelinating cells. Canadian Journal of Neurological Sciences, 37(Suppl 2), S34-41
Bifari, F., Decimo, I., Pino, A., Llorens-Bobadilla, E., Zhao, S., Lange, C., Panuccio, G., Boeckx, B., Thienpont, B., Vinckier, S., & Wyns, S. (2017). Neurogenic radial glia-like cells in meninges migrate and differentiate into functionally integrated neurons in the neonatal cortex. Cell Stem Cell, 20(360–73), e7
Cai, R., Pan, C., Ghasemigharagoz, A., Todorov, M. I., Forstera, B., Zhao, S., Bhatia, H.S., Parra-Damas, A., Mrowka, L., Theodorou, D., & Rempfler, M. (2019). Panoptic imaging of transparent mice reveals whole-body neuronal projections and skull-meninges connections. Nature Neuroscience, 22, 317–327
Carmichael, S. T. (2010). Targets for neural repair therapies after stroke. Stroke, 41, S124–S126
Chai, Y., Ito, Y., & Han, J. (2003). TGF-beta signaling and its functional significance in regulating the fate of cranial neural crest cells. Critical Reviews in Oral Biology and Medicine, 14, 78–88
Decimo, I., Bifari, F., Rodriguez, F. J., Malpeli, G., Dolci, S., Lavarini, V., Pretto, S., Vasquez, S., Sciancalepore, M., Montalbano, A., & Berton, V. (2011). Nestin- and doublecortin-positive cells reside in adult spinal cord meninges and participate in injury-induced parenchymal reaction. Stem Cells., 29, 2062–2076
Decimo, I., Fumagalli, G., Berton, V., Krampera, M., & Bifari, F. (2012). Meninges: from protective membrane to stem cell niche. American Journal of Stem Cells, 1, 92–105
Dupin, E., & Coelho-Aguiar, J. M. (2013). Isolation and differentiation properties of neural crest stem cells. Cytometry. Part A, 83, 38–47
Etchevers, H. C., Couly, G., Vincent, C., & Le Douarin, N. M. (1999). Anterior cephalic neural crest is required for forebrain viability. Development, 126, 3533–3543
Etchevers, H. C., Vincent, C., Le Douarin, N. M., & Couly, G. F. (2001). The cephalic neural crest provides pericytes and smooth muscle cells to all blood vessels of the face and forebrain. Development, 128, 1059–1068
Filiano, A. J., Gadani, S. P., & Kipnis, J. (2015). Interactions of innate and adaptive immunity in brain development and function. Brain Research, 1617, 18–27
Hallmann, R., Horn, N., Selg, M., Wendler, O., Pausch, F., & Sorokin, L. M. (2005). Expression and function of laminins in the embryonic and mature vasculature. Physiological Reviews, 85, 979–1000
Hartmann, D., Sievers, J., Pehlemann, F. W., & Berry, M. (1992). Destruction of meningeal cells over the medial cerebral hemisphere of newborn hamsters prevents the formation of the infrapyramidal blade of the dentate gyrus. The Journal of Comparative Neurology, 320, 33–61
Iadecola, C., & Anrather, J. (2011). The immunology of stroke: from mechanisms to translation. Nature Medicine, 17, 796–808
Jacobson, S., & Marcus, E. (2008). Neuroanatomy for the neuroscientist. (pp. 325–331). New York: Springer.
Kelly, K. K., MacPherson, A. M., Grewal, H., Strnad, F., Jones, J. W., Yu, J., Pierzchalski, K., Kane, M. A., Herson, P. S., & Siegenthaler, J. A. (2016). Col1a1+ perivascular cells in the brain are a source of retinoic acid following stroke. BMC Neuroscience, 17, 49
Lee, G., Kim, H., Elkabetz, Y., Al Shamy, G., Panagiotakos, G., Barberi, T., Tabar, V., & Studer, L. (2007). Isolation and directed differentiation of neural crest stem cells derived from human embryonic stem cells. Nature Biotechnology, 25, 1468–1475
Mercier, F., & Arikawa-Hirasawa, E. (2012). Heparan sulfate niche for cell proliferation in the adult brain. Neuroscience Letters, 510, 67–72
Morikawa, S., Mabuchi, Y., Niibe, K., Suzuki, S., Nagoshi, N., Sunabori, T., Shimmura, S., Nagai, Y., Nakagawa, T., Okano, H., & Matsuzaki, Y. (2009). Development of mesenchymal stem cells partially originate from the neural crest. Biochemical and Biophysical Research Communications, 379, 1114–1119
Muller, J., Ossig, C., Greiner, J. F., Hauser, S., Fauser, M., Widera, D., Kaltschmidt, C., Storch, A., & Kaltschmidt, B. (2015). Intrastriatal transplantation of adult human neural crest-derived stem cells improves functional outcome in parkinsonian rats. Stem Cells Translational Medicine, 4, 31–43
Nakagomi, T., Molnar, Z., Nakano-Doi, A., Taguchi, A., Saino, O., Kubo, S., Clausen, M., Yoshikawa, H., Nakagomi, N., & Matsuyama, T. (2011). Ischemia-induced neural stem/progenitor cells in the pia mater following cortical infarction. Stem Cells Development., 20, 2037–2051
O’Rahilly, R., & Muller, F. (1986). The meninges in human development. Journal of Neuropathology and Experimental Neurology, 45, 588–608
Saadai, P., Wang, A., Nout, Y. S., Downing, T. L., Lofberg, K., Beattie, M. S., Bresnahan, J. C., Li, S., & Farmer, D. L. (2013). Human induced pluripotent stem cell-derived neural crest stem cells integrate into the injured spinal cord in the fetal lamb model of myelomeningocele. Journal of Pediatric Surgery, 48, 158–163
Acknowledgements
This work was supported by grants from NIH.
Author information
Authors and Affiliations
Contributions
EHL, EYS, and KH contributed to manuscript preparation.
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hayakawa, K., Snyder, E.Y. & Lo, E.H. Meningeal Multipotent Cells: A Hidden Target for CNS Repair?. Neuromol Med 23, 339–343 (2021). https://doi.org/10.1007/s12017-021-08663-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-021-08663-1