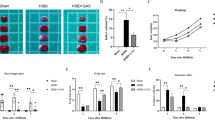
Abstract
In response to hypoxic-ischemic brain damage (HIBD), microglia activation and its mediated inflammation contribute to neuronal damage. Inhibition of over-activated microglia is deemed to be a potential therapeutic strategy. Our previous studies showed that gastrodin efficiently depressed the neuroinflammation mediated by activated microglia in HIBD neonatal rats. The underlying mechanisms through which gastrodin acts on activated microglia have not been fully elucidated. This study is designed to determine whether gastrodin would regulate the Notch signaling pathway and Sirtuin3 (Sirt3), which are implicated in regulating microglia activation. The present results showed that gastrodin markedly suppressed the expression of members of Notch signaling pathway (Notch-1, NICD, RBP-JK and Hes-1) in activated microglia both in vivo and in vitro. Conversely, Sirt3 expression was enhanced. In BV-2 microglia treated with a γ-secretase inhibitor of Notch pathway- DAPT, the expression of RBP-JK, Hes-1, and NICD was suppressed in activated microglia. Treatment with DAPT and gastrodin further decreased NICD and Hes-1 expression. Sirt3 expression was also decreased after DAPT treatment. However, Sirt3 expression in activated BV-2 microglia given a combined DAPT and gastrodin treatment was not further increased. In addition, combination of DAPT and Gastrodin cumulatively decreased tumor necrosis factor-α (TNF-α) expression. The results suggest that gastrodin regulates microglia activation via the Notch signaling pathway and Sirt3. More importantly, interference of the Notch signaling pathway inhibited Sirt3 expression, indicating that Sirt3 is a downstream gene of the Notch signaling pathway. It is suggested that Notch and Sirt3 synergistically regulate microglia activation such as in TNF-α production.








Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- AT1 :
-
Angiotensin II receptor 1
- CNS:
-
Central nervous system
- DAPT:
-
N-[N-(3,5-Difluorophenacetyl)-1-alany1]-S-phenyglycine t-butylester
- FCS:
-
Fetal calf serum
- HIBD:
-
Hypoxic-ischemic brain damage
- Hes-1:
-
Transcription factor hairy and enhancer of split-1
- iNOS:
-
Inducible nitric oxid synthase
- IL-1β:
-
Interleukin-1β
- LPS:
-
Lipopolysaccharide
- Lectin:
-
Lycopersicon esculentum
- MCAO:
-
Middle cerebral artery occlusion
- NO:
-
Nitric oxid
- NICD:
-
Intracellular Notch receptor domain
- PBS:
-
Phosphate buffered saline
- ROS:
-
Reactive oxygen species
- RBP-JK:
-
Recombining binding protein suppressor of hairless
- Sirt3:
-
Sirtuin3
- SD:
-
Sprague–Dawley
- TNF-α:
-
Tumor necrosis factor-α
References
Alberi, L., Hoey, S. E., Brai, E., Scotti, A. L., & Marathe, S. (2013). Notch signaling in the brain: In good and bad times. Ageing Research Reviews, 12(3), 801–814. https://doi.org/10.1016/j.arr.2013.03.004
Ashwell, K. (1991). The distribution of microglia and cell death in the fetal rat forebrain. Brain Research Developmental Brain Research, 58(1), 1–12.
Cao, Q., Karthikeyan, A., Dheen, S. T., Kaur, C., & Ling, E. A. (2017). Production of proinflammatory mediators in activated microglia is synergistically regulated by Notch-1, glycogen synthase kinase (GSK-3beta) and NF-kappaB/p65 signalling. PLoS ONE, 12(10), e0186764. https://doi.org/10.1371/journal.pone.0186764
Cao, Q., Li, P., Lu, J., Dheen, S. T., Kaur, C., & Ling, E. A. (2010). Nuclear factor-kappaB/p65 responds to changes in the Notch signaling pathway in murine BV-2 cells and in amoeboid microglia in postnatal rats treated with the gamma-secretase complex blocker DAPT. Journal of Neuroscience Research, 88(12), 2701–2714. https://doi.org/10.1002/jnr.22429
Cao, Q., Lu, J., Kaur, C., Sivakumar, V., Li, F., Cheah, P. S., et al. (2008). Expression of Notch-1 receptor and its ligands Jagged-1 and Delta-1 in amoeboid microglia in postnatal rat brain and murine BV-2 cells. Glia, 56(11), 1224–1237. https://doi.org/10.1002/glia.20692
Cao, R., Li, S., Yin, J., Guo, L., & Shi, J. (2019). Sirtuin 3 promotes microglia migration by upregulating CX3CR1. Cell Adhesion & Migration, 13(1), 229–235. https://doi.org/10.1080/19336918.2019.1629224
Cheng, Z., Yang, Y., Duan, F., Lou, B., Zeng, J., Huang, Y., et al. (2019). Inhibition of notch1 signaling alleviates endotoxin-induced inflammation through modulating retinal microglia polarization. Frontiers in Immunology, 10, 389. https://doi.org/10.3389/fimmu.2019.00389
Dheen, S. T., Kaur, C., & Ling, E. A. (2007). Microglial activation and its implications in the brain diseases. Current Medicinal Chemistry, 14(11), 1189–1197.
Douglas-Escobar, M., & Weiss, M. D. (2015). Hypoxic-ischemic encephalopathy: A review for the clinician. JAMA Pediatrics, 169(4), 397–403. https://doi.org/10.1001/jamapediatrics.2014.3269
Dumont, U., Sanchez, S., Olivier, B., Chateil, J. F., Pellerin, L., Beauvieux, M. C., et al. (2019). Maternal consumption of piceatannol: A nutritional neuroprotective strategy against hypoxia-ischemia in rat neonates. Brain Research, 1717, 86–94. https://doi.org/10.1016/j.brainres.2019.04.012
Furukawa, S., Sameshima, H., Yang, L., & Ikenoue, T. (2013). Activation of acetylcholine receptors and microglia in hypoxic-ischemic brain damage in newborn rats. Brain and Development, 35(7), 607–613. https://doi.org/10.1016/j.braindev.2012.10.006
Hao, X. Z., Tian, J. Q., Yin, L. K., Zhang, X. X., Li, C. C., Feng, X. Y., et al. (2017). MRI detects protective effects of DAPT treatment with modulation of microglia/macrophages at subacute and chronic stages following cerebral ischemia. Molecular Medicine Reports, 16(4), 4493–4500. https://doi.org/10.3892/mmr.2017.7200
Huang, J. Z., Ren, Y., Jiang, Y., Shen, S. Y., Ding, J., & Hua, F. (2018). GluR1 protects hypoxic ischemic brain damage via activating Akt signaling pathway in neonatal rats. European Review for Medical and Pharmacological Sciences, 22(24), 8857–8865. https://doi.org/10.26355/eurrev_201812_16654
Jiang, D. Q., Ma, Y. J., Wang, Y., Lu, H. X., Mao, S. H., & Zhao, S. H. (2019). Microglia activation induces oxidative injury and decreases SIRT3 expression in dopaminergic neuronal cells. Journal of Neural Transmission (Vienna), 126(5), 559–568. https://doi.org/10.1007/s00702-019-02005-z
Jiang, D. Q., Wang, Y., Li, M. X., Ma, Y. J., & Wang, Y. (2017). SIRT3 in neural stem cells attenuates microglia activation-induced oxidative stress injury through mitochondrial pathway. Frontiers in Cellular Neuroscience, 11, 7. https://doi.org/10.3389/fncel.2017.00007
Kao, T. K., Chang, C. Y., Ou, Y. C., Chen, W. Y., Kuan, Y. H., Pan, H. C., et al. (2013). Tetramethylpyrazine reduces cellular inflammatory response following permanent focal cerebral ischemia in rats. Experimental Neurology, 247, 188–201. https://doi.org/10.1016/j.expneurol.2013.04.010
Kaur, C., Rathnasamy, G., & Ling, E. A. (2013). Roles of activated microglia in hypoxia induced neuroinflammation in the developing brain and the retina. Journal of Neuroimmune Pharmacology, 8(1), 66–78. https://doi.org/10.1007/s11481-012-9347-2
Kaur, C., Rathnasamy, G., & Ling, E. A. (2017). Biology of microglia in the developing brain. Journal of Neuropathology and Experimental Neurology, 76(9), 736–753. https://doi.org/10.1093/jnen/nlx056
Kim, B. W., Koppula, S., Kim, J. W., Lim, H. W., Hwang, J. W., Kim, I. S., et al. (2012). Modulation of LPS-stimulated neuroinflammation in BV-2 microglia by Gastrodia elata: 4-hydroxybenzyl alcohol is the bioactive candidate. Journal of Ethnopharmacology, 139(2), 549–557. https://doi.org/10.1016/j.jep.2011.11.048
Koshizaka, M., Takemoto, M., Sato, S., Tokuyama, H., Fujimoto, M., Okabe, E., et al. (2012). An angiotensin II type 1 receptor blocker prevents renal injury via inhibition of the Notch pathway in Ins2 Akita diabetic mice. Exp Diabetes Res, 2012, 159874. https://doi.org/10.1155/2012/159874
Laptook, A. R., Shankaran, S., Tyson, J. E., Munoz, B., Bell, E. F., Goldberg, R. N., et al. (2017). Effect of therapeutic hypothermia initiated after 6 hours of age on death or disability among newborns with hypoxic-ischemic encephalopathy: A randomized clinical trial. JAMA, 318(16), 1550–1560. https://doi.org/10.1001/jama.2017.14972
Li, J. J., Lu, J., Kaur, C., Sivakumar, V., Wu, C. Y., & Ling, E. A. (2009). Expression of angiotensin II and its receptors in the normal and hypoxic amoeboid microglial cells and murine BV-2 cells. Neuroscience, 158(4), 1488–1499. https://doi.org/10.1016/j.neuroscience.2008.11.046
Li, S. J., Liu, W., Wang, J. L., Zhang, Y., Zhao, D. J., Wang, T. J., et al. (2014). The role of TNF-alpha, IL-6, IL-10, and GDNF in neuronal apoptosis in neonatal rat with hypoxic-ischemic encephalopathy. European Review for Medical and Pharmacological Sciences, 18(6), 905–909.
Li, X. H., Liu, S. J., Liu, X. Y., Zhao, H. Y., Yang, M. G., Xu, D. X., et al. (2018). Expression of SIRT3 in various glial cell types in the periventricular white matter in the neonatal rat brain after hypoxia. Tissue and Cell, 52, 1–8. https://doi.org/10.1016/j.tice.2018.03.004
Ling, E. A., & Wong, W. C. (1993). The origin and nature of ramified and amoeboid microglia: a historical review and current concepts. Glia, 7(1), 9–18. https://doi.org/10.1002/glia.440070105
Liu, S. J., Liu, X. Y., Li, J. H., Guo, J., Li, F., Gui, Y., et al. (2018). Gastrodin attenuates microglia activation through renin-angiotensin system and Sirtuin3 pathway. Neurochemistry International, 120, 49–63. https://doi.org/10.1016/j.neuint.2018.07.012
Longa, E. Z., Weinstein, P. R., Carlson, S., & Cummins, R. (1989). Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke, 20(1), 84–91. https://doi.org/10.1161/01.str.20.1.84
Lutolf, S., Radtke, F., Aguet, M., Suter, U., & Taylor, V. (2002). Notch1 is required for neuronal and glial differentiation in the cerebellum. Development, 129(2), 373–385.
Neumann, H., Kotter, M. R., & Franklin, R. J. (2009). Debris clearance by microglia: An essential link between degeneration and regeneration. Brain, 132(Pt 2), 288–295. https://doi.org/10.1093/brain/awn109
Peng, Z., Wang, S., Chen, G., Cai, M., Liu, R., Deng, J., et al. (2015). Gastrodin alleviates cerebral ischemic damage in mice by improving anti-oxidant and anti-inflammation activities and inhibiting apoptosis pathway. Neurochemical Research, 40(4), 661–673. https://doi.org/10.1007/s11064-015-1513-5
Perry, V. H., Hume, D. A., & Gordon, S. (1985). Immunohistochemical localization of macrophages and microglia in the adult and developing mouse brain. Neuroscience, 15(2), 313–326.
Rangarajan, P., Karthikeyan, A., Lu, J., Ling, E. A., & Dheen, S. T. (2015). Sirtuin 3 regulates Foxo3a-mediated antioxidant pathway in microglia. Neuroscience, 311, 398–414. https://doi.org/10.1016/j.neuroscience.2015.10.048
Sundaresan, N. R., Gupta, M., Kim, G., Rajamohan, S. B., Isbatan, A., & Gupta, M. P. (2009). Sirt3 blocks the cardiac hypertrophic response by augmenting Foxo3a-dependent antioxidant defense mechanisms in mice. Journal of Clinical Investigation, 119(9), 2758–2771. https://doi.org/10.1172/JCI39162
Sundaresan, N. R., Samant, S. A., Pillai, V. B., Rajamohan, S. B., & Gupta, M. P. (2008). SIRT3 is a stress-responsive deacetylase in cardiomyocytes that protects cells from stress-mediated cell death by deacetylation of Ku70. Molecular and Cellular Biology, 28(20), 6384–6401. https://doi.org/10.1128/MCB.00426-08
Wang, J. W., Liu, Y. M., Zhao, X. F., & Zhang, H. (2017). Gastrodin protects retinal ganglion cells through inhibiting microglial-mediated neuroinflammation in an acute ocular hypertension model. International Journal of Ophthalmology, 10(10), 1483–1489. https://doi.org/10.18240/ijo.2017.10.01
Wei, Z., Chigurupati, S., Arumugam, T. V., Jo, D. G., Li, H., & Chan, S. L. (2011). Notch activation enhances the microglia-mediated inflammatory response associated with focal cerebral ischemia. Stroke, 42(9), 2589–2594. https://doi.org/10.1161/STROKEAHA.111.614834
Yao, L., Cao, Q., Wu, C., Kaur, C., Hao, A., & Ling, E. A. (2013). Notch signaling in the central nervous system with special reference to its expression in microglia. CNS & Neurological Disorders: Drug Targets, 12(6), 807–814.
Yao, L., Kan, E. M., Kaur, C., Dheen, S. T., Hao, A., Lu, J., et al. (2013). Notch-1 signaling regulates microglia activation via NF-kappaB pathway after hypoxic exposure in vivo and in vitro. PLoS ONE, 8(11), e78439. https://doi.org/10.1371/journal.pone.0078439
Yao, M., Wang, X., Wang, X., Zhang, T., Chi, Y., & Gao, F. (2015). The Notch pathway mediates the angiotensin II-induced synthesis of extracellular matrix components in podocytes. International Journal of Molecular Medicine, 36(1), 294–300. https://doi.org/10.3892/ijmm.2015.2193
Yao, Y. Y., Bian, L. G., Yang, P., Sui, Y., Li, R., Chen, Y. L., et al. (2019). Gastrodin attenuates proliferation and inflammatory responses in activated microglia through Wnt/beta-catenin signaling pathway. Brain Research, 1717, 190–203. https://doi.org/10.1016/j.brainres.2019.04.025
Yuan, Y., Rangarajan, P., Kan, E., Wu, Y., Wu, C., & Ling, E. A. (2015). Scutellarin regulates the Notch pathway and affects the migration and morphological transformation of activated microglia in experimentally induced cerebral ischemia in rats and in activated BV-2 microglia. Journal of Neuroinflammation, 12(1), 11. https://doi.org/10.1186/s12974-014-0226-z
Zeng, W. X., Han, Y. L., Zhu, G. F., Huang, L. Q., Deng, Y. Y., Wang, Q. S., et al. (2017). Hypertonic saline attenuates expression of Notch signaling and proinflammatory mediators in activated microglia in experimentally induced cerebral ischemia and hypoxic BV-2 microglia. BMC Neuroscience, 18(1), 32. https://doi.org/10.1186/s12868-017-0351-6
Zhou, X., Chen, M., Zeng, X., Yang, J., Deng, H., Yi, L., et al. (2014). Resveratrol regulates mitochondrial reactive oxygen species homeostasis through Sirt3 signaling pathway in human vascular endothelial cells. Cell Death Dis, 5, e1576. https://doi.org/10.1038/cddis.2014.530
Zong, Y., Ai, Q. L., Zhong, L. M., Dai, J. N., Yang, P., He, Y., et al. (2012). Ginsenoside Rg1 attenuates lipopolysaccharide-induced inflammatory responses via the phospholipase C-gamma1 signaling pathway in murine BV-2 microglial cells. Current Medicinal Chemistry, 19(5), 770–779. https://doi.org/10.2174/092986712798992066
Acknowledgements
We appreciate the help of Emeritus Professor Eng-Ang Ling, Department of Anatomy, National University of Singapore for his revision of the manuscript.
Funding
This study was supported by National Natural Science Foundation of China (Project Number 31760297, Y Yuan), Applied Basic Research Projects of Yunnan Province (Project Number 2018FE001(-189), Y Yuan). It was also supported by National Natural Science Foundation of China (No. 31960194 and 31460274, J-J Li; No. 81760280, F Li) and Applied Basic Research Projects of Yunnan Province of China (No.2019FE001(-003), J-J Li; No. 2018FE001 (-002), F Li).
Author information
Authors and Affiliations
Contributions
YY conceptualized and designed this study. YY and J-JL supervised the whole project. JG, X-L-NZ, Z-RB, X-KY, L-SL, YZ, FL, and C-YW carried out the experiments and prepared the manuscript. All authors participated in discussion, analysis of data, and editing the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12017_2020_8627_MOESM1_ESM.jpg
Electronic supplementary material 1 (JPG 3681 kb). Supplementary Fig 1 Gastrodin decreased Notch-1 and NICD expression in activated microglia at 7 d. Immunofluorescence images show the expression of Notch-1 and NICD (red) in lectin+ microglia (green) in the corpus callosum of HIBD rats was increased compared with the corresponding cells in sham group rats at 7 d. Notch-1 and NICD expression was observed to decrease following treatment with gastrodin. DAPI (blue) shows nuclei. Significant differences are expressed as *P <0.05 between the sham and HIBD group, # P <0.05 between HIBD and HIBD + G group. Scale bars: 20 μm. The values represent the mean ± SD in triplicate
12017_2020_8627_MOESM2_ESM.jpg
Electronic supplementary material 2 (JPG 4075 kb). Supplementary Fig 2 Gastrodin reduced RBP-JK and Hes-1 expression in activated microglia at 7 d. Immunofluorescence images show the expression of RBP-JK and Hes-1 (red) in lectin+ microglia (green) in the corpus callosum of HIBD rats was increased compared with the corresponding cells in sham group rats at 7 d. RBP-JK and Hes-1 expression was reduced following treatment with gastrodin. DAPI (blue) shows nuclei. Significant differences are expressed as * P <0.05 between the sham and HIBD group, # P <0.05 between HIBD and HIBD + G group. Scale bars: 20 μm. The values represent the mean ± SD in triplicate
12017_2020_8627_MOESM3_ESM.jpg
Electronic supplementary material 3 (JPG 1904 kb). Supplementary Fig 3 Gastrodin increased Sirt3 expression in activated microglia at 7 d. Protein expression of Notch-1, NICD, RBP-JK and Hes-1 was decreased, while Sirt3 expression was increased in HIBD rat following treatment with gastrodin at 7 d. (A) Immunofluorescence images show the expression of Sirt3 (red) in lectin+ microglia (green) in the corpus callosum of HIBD rats was increased compared with the corresponding cells in sham group rats at 7 d. Sirt3 expression was further increased following treatment with gastrodin. Scale bars: 20 μm. (B) The expression levels of Notch-1, NICD, RBP-JK and Hes-1 in the corpus callosum in HIBD rat were depressed significantly at 7 d following treatment with gastrodin when compared with the HIBD rats, while the expression level of Sirt3 was upregulated at 7 d after pretreatment with gastrodin when compared with the HIBD rats. Significant differences in protein levels between the sham and HIBD groups are expressed as * p< 0.05; differences between HIBD and HIBD + G are expressed as # p< 0.05. The values represent the mean ± SD in triplicate
Rights and permissions
About this article
Cite this article
Guo, J., Zhang, XLN., Bao, ZR. et al. Gastrodin Regulates the Notch Signaling Pathway and Sirt3 in Activated Microglia in Cerebral Hypoxic-Ischemia Neonatal Rats and in Activated BV-2 Microglia. Neuromol Med 23, 348–362 (2021). https://doi.org/10.1007/s12017-020-08627-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-020-08627-x