Abstract
Purpose of Review
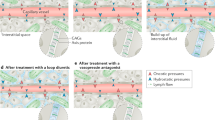
The lymphatic system plays a major but overlooked role in maintaining fluid homeostasis. Given the unique fluid homeostasis functions of the kidneys, dysregulation of the renal lymphatic system underlies the development of self-propagating congestive pathomechanisms. In this review, we outline the roles of the renal lymphatic system in heart failure (HF).
Recent Findings
Studies have uncovered several pathomechanisms involving the renal lymphatic system in congestive states, such as impaired interstitial draining by the renal lymphatic system, impaired structure and valves of renal lymphatics, lymphatic-induced increase in renal reabsorption of water and sodium, and development of albuminuria with proteinuria-induced renal lymphangiogenesis. These self-propagating mechanisms result in “renal tamponade” with manifestations of cardiorenal syndrome and inappropriate renal response to diuretics.
Summary
Dysregulation of the renal lymphatic system is integral to the development and progression of congestion in HF. Targeting renal lymphatics may provide a novel pathway to treat intractable congestion.




Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
• Fudim M, Salah HM, Sathananthan J, et al. Lymphatic dysregulation in patients with heart failure: JACC Review Topic of the Week. J Am Coll Cardiol. 2021;78:66–76. In this paper, lymphatic dysregulation was suggested as a driving pathomechanism for the manifestations of congestion in heart failure, and targeting the lymphatic system was proposed as a potenital novel treatment pathway to address congestion in heart failure.
Breslin JW, Yang Y, Scallan JP, Sweat RS, Adderley SP, Murfee WL. Lymphatic vessel network structure and physiology. Compr Physiol. 2018;9:207–99.
Schwager S, Detmar M. Inflammation and lymphatic function. Front Immunol. 2019;10:308.
Wang Y, Oliver G. Current views on the function of the lymphatic vasculature in health and disease. Genes Dev. 2010;24:2115–26.
Hall JE, Hall ME. Guyton and Hall Textbook of Medical Physiology, Elsevier, 2020.
Jones D, Min W. An overview of lymphatic vessels and their emerging role in cardiovascular disease. J Cardiovasc Dis Res. 2011;2:141–52.
Tammela T, Alitalo K. Lymphangiogenesis: molecular mechanisms and future promise. Cell. 2010;140:460–76.
Santambrogio L. The lymphatic fluid. Int Rev Cell Mol Biol. 2018;337:111–33.
Null M, Agarwal M. Anatomy, lymphatic system. StatPearls. Treasure Island (FL): StatPearls Publishing Copyright © 2022, Stat Pearls Publishing LLC, 2022.
Choi I, Lee S, Hong YK. The new era of the lymphatic system: no longer secondary to the blood vascular system. Cold Spring Harb Perspect Med. 2012;2:a006445.
Donnan MD, Kenig-Kozlovsky Y, Quaggin SE. The lymphatics in kidney health and disease. Nat Rev Nephrol. 2021;17:655–75.
LeBrie SJ, Mayerson HS. Influence of elevated venous pressure on flow and composition of renal lymph. American Journal of Physiology-Legacy Content. 1960;198:1037–40.
Cockett ATK. Lymphatic network of kidney I. anatomic and physiologic considerations. Urology. 1977;9:125–9.
Heney NM, O’Morchoe PJ, O’Morchoe CCC. The renal lymphatic system during obstructed urine flow. J Urol. 1971;106:455–62.
Ishikawa Y, Akasaka Y, Kiguchi H, et al. The human renal lymphatics under normal and pathological conditions. Histopathology. 2006;49:265–73.
Cuttino JT, Clark RL, Charles JJ. Microradiographic demonstration of human intrarenal microlymphatic pathways. Urol Radiol. 1989;11:83–7.
Holmes MJ, O’Morchoe PJ, O’Morchoe CCC. Morphology of the intrarenal lymphatic system. Capsular and hilar communications. Am J Anat. 1977;149:333–51.
Holmes MJ, O’Morchoe PJ, O’Morchoe CC. Morphology of the intrarenal lymphatic system. Capsular and hilar communications. Am J Anat. 1977;149:333–51.
Russell PS, Hong J, Windsor JA, Itkin M, Phillips ARJ. Renal lymphatics: anatomy, physiology, and clinical implications. Front Physiol. 2019;10:251.
Boorsma EM, ter Maaten JM, Damman K et al. Albuminuria as a marker of systemic congestion in patients with heart failure. Eur Heart J 2022;44:368–380.
Sugarman J, Friedman M, Barrett E, Addis T. The distribution, flow, protein and urea content of renal lymph. American Journal of Physiology-Legacy Content. 1942;138:108–12.
Swann HG, Ormsby AA, Delashaw JB, Tharp WW. Relation of lymph to distending fluids of the kidney. Proc Soc Exp Biol Med. 1958;97:517–22.
LeBrie S, Mayerson H. Composition of renal lymph and its significance. Proc Soc Exp Biol Med. 1959;100:378–80.
Hargens AR, Tucker BJ, Blantz RC. Renal lymph protein in the rat. Am J Physiol-Renal Physiol. 1977;233:F269–73.
Lever AF, Peart WS. Renin and angiotensin-like activity in renal lymph. J Physiol. 1962;160:548–63.
Bailie MD, Rector FC Jr, Seldin DW. Angiotensin II in arterial and renal venous plasma and renal lymph in the dog. J Clin Invest. 1971;50:119–26.
Ott CE, Knox FG. Tissue pressures and fluid dynamics in the kidney. Fed Proc. 1976;35:1872–5.
The lymphatic system in body homeostasis. physiological conditions. Lymphat Res Biol. 2003;1:11–24.
Fonarow GC, Heywood JT, Heidenreich PA, Lopatin M, Yancy CW. Temporal trends in clinical characteristics, treatments, and outcomes for heart failure hospitalizations, 2002 to 2004: findings from Acute Decompensated Heart Failure National Registry (ADHERE). Am Heart J. 2007;153:1021–8.
Damman K, Deursen VMV, Navis G, Voors AA, Veldhuisen DJV, Hillege HL. Increased central venous pressure is associated with impaired renal function and mortality in a broad spectrum of patients with cardiovascular disease. J Am Coll Cardiol. 2009;53:582–8.
Ganda A, Onat D, Demmer RT, et al. Venous congestion and endothelial cell activation in acute decompensated heart failure. Curr Heart Fail Rep. 2010;7:66–74.
Tansey EA, Montgomery LEA, Quinn JG, Roe SM, Johnson CD. Understanding basic vein physiology and venous blood pressure through simple physical assessments. Adv Physiol Educ. 2019;43:423–9.
Joris I, Cuénoud HF, Doern GV, Underwood JM, Majno G. Capillary leakage in inflammation A study by vascular labeling. Am J Pathol. 1990;137:1353–63.
• Boorsma EM, ter Maaten JM, Voors AA, van Veldhuisen DJ. Renal compression in heart failure: the renal tamponade hypothesis. JACC: Heart Failure. 2022;10:175–83. In this paper, the concept of "renal tamponade" (i.e., compression of renal structures due limited space for expansion within the rigid renal capsule) was proposed as a pathomechanism for the disproportionate impairment in kidney function in the setting of elevated central venous pressure.
Colombo PC, Ganda A, Lin J, et al. Inflammatory activation: cardiac, renal, and cardio-renal interactions in patients with the cardiorenal syndrome. Heart Fail Rev. 2012;17:177–90.
Afsar B, Ortiz A, Covic A, Solak Y, Goldsmith D, Kanbay M. Focus on renal congestion in heart failure. Clin Kidney J. 2016;9:39–47.
Katz YJ, Cockett ATK, Moor RS. Elevation of inferior vena cava pressure and thoracic lymph and urine flow. Circ Res. 1959;7:118–22.
Mullens W, Verbrugge FH, Nijst P, Tang WHW. Renal sodium avidity in heart failure: from pathophysiology to treatment strategies. Eur Heart J. 2017;38:1872–82.
Fountain JH, Lappin SL. Physiology, renin angiotensin system. StatPearls. Treasure Island (FL): StatPearls Publishing, Copyright © 2022, StatPearls Publishing LLC, 2022.
Threefoot SA, Pearson JE, Georgiardis A. Albumin uptake by renal lymphatics with and without obstruction of the renal vein. Am J Cardiol. 1989;64:C51–6.
Zhang T, Guan G, Liu G, et al. Disturbance of lymph circulation develops renal fibrosis in rats with or without contralateral nephrectomy. Nephrology (Carlton). 2008;13:128–38.
Yazdani S, Poosti F, Kramer AB, et al. Proteinuria triggers renal lymphangiogenesis prior to the development of interstitial fibrosis. PLoS ONE. 2012;7:e50209.
Rangaswami J, Bhalla V, Blair JEA, et al. Cardiorenal syndrome: classification, pathophysiology, diagnosis, and treatment strategies: a scientific statement from the American Heart Association. Circulation. 2019;139:e840–78.
Ronco C, Haapio M, House AA, Anavekar N, Bellomo R. Cardiorenal syndrome. J Am Coll Cardiol. 2008;52:1527–39.
Zymliński R, Biegus J, Sokolski M, et al. Increased blood lactate is prevalent and identifies poor prognosis in patients with acute heart failure without overt peripheral hypoperfusion. Eur J Heart Fail. 2018;20:1011–8.
Adams KF, Fonarow GC, Emerman CL, et al. Characteristics and outcomes of patients hospitalized for heart failure in the United States: rationale, design, and preliminary observations from the first 100,000 cases in the Acute Decompensated Heart Failure National Registry (ADHERE). Am Heart J. 2005;149:209–16.
Damman K, Navis G, Smilde TDJ, et al. Decreased cardiac output, venous congestion and the association with renal impairment in patients with cardiac dysfunction. Eur J Heart Fail. 2007;9:872–8.
Mullens W, Abrahams Z, Francis GS, et al. Importance of venous congestion for worsening of renal function in advanced decompensated heart failure. J Am Coll Cardiol. 2009;53:589–96.
Bart BA, Goldsmith SR, Lee KL, et al. Ultrafiltration in decompensated heart failure with cardiorenal syndrome. N Engl J Med. 2012;367:2296–304.
Cole WR, Witte MH, Kash SL, Rodger M, Bleisch VR, Muelheims GH. Thoracic duct-to-pulmonary vein shunt in the treatment of experimental right heart failure. Circulation. 1967;36:539–43.
• Abraham WT, Jonas M, Dongaonkar RM, et al. Direct interstitial decongestion in an animal model of acute-on-chronic ischemic heart failure. JACC Basic to Transl Sci. 2021;6:872–81. In this work, targeting the lymphatic system using a device-based approach to create a low-pressure zone at the thoracic duct outlet significantly increased urine output in animals and human.
Witte MH, Dumont AE, Clauss RH, Rader B, Levine N, Breed ES. Lymph circulation in congestive heart failure. Circulation. 1969;39:723–33.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Fudim was supported by the National Heart, Lung, and Blood Institute (NHLBI) (K23HL151744), the American Heart Association (20IPA35310955), Mario Family Award, Duke Chair’s Award, Translating Duke Health Award, Bayer, Bodyport, BTG Specialty Pharmaceuticals, and Verily. He receives consulting fees from Abbott, Alleviant, Audicor, AxonTherapies, Bayer, Bodyguide, Bodyport, Boston Scientific, CVRx, Daxor, Deerfield Catalyst, Edwards LifeSciences, Feldschuh Foundation, Fire1, Gradient, Intershunt, NXT Biomedical, Pharmacosmos, PreHealth, Shifamed, Splendo, Vironix, Viscardia, and Zoll. All other authors declare no disclosures.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Salah, H.M., Biegus, J. & Fudim, M. Role of the Renal Lymphatic System in Heart Failure. Curr Heart Fail Rep 20, 113–120 (2023). https://doi.org/10.1007/s11897-023-00595-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11897-023-00595-0