Abstract
Purpose of Review
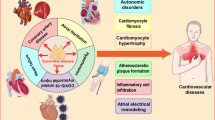
The obesity epidemic is on the rise, and while it is well known that obesity is associated with an increase in cardiovascular risk factors such as type 2 diabetes mellitus, hypertension, and obstructive sleep apnea, recent data has highlighted that the degree and type of fat distribution may play a bigger role in the pathogenesis of cardiovascular disease (CVD) than body mass index (BMI) alone. We aim to review updated data on adipose tissue inflammation and distribution and CVD.
Recent Findings
We review the pathophysiology of inflammation secondary to adipose tissue, the association of obesity-related adipokines and CVD, and the differences and significance of brown versus white adipose tissue. We delve into the clinical manifestations of obesity-related inflammation in CVD. We discuss the available data on heterogeneity of adipose tissue-related inflammation with a focus on subcutaneous versus visceral adipose tissue, the differential pathophysiology, and clinical CVD manifestations of adipose tissue across sex, race, and ethnicity. Finally, we present the available data on lifestyle modification, medical, and surgical therapeutics on reduction of obesity-related inflammation.
Summary
Obesity leads to a state of chronic inflammation which significantly increases the risk for CVD. More research is needed to develop non-invasive VAT quantification indices such as risk calculators which include variables such as sex, age, race, ethnicity, and VAT concentration, along with other well-known CVD risk factors in order to comprehensively determine risk of CVD in obese patients. Finally, pre-clinical biomarkers such as pro-inflammatory adipokines should be validated to estimate risk of CVD in obese patients.

Similar content being viewed by others
Abbreviations
- CVD:
-
Cardiovascular disease
- VAT:
-
Visceral adipose tissue
- SAT:
-
Subcutaneous adipose tissue
- TNF-α:
-
Tumor necrosis factor-α
- IL-6:
-
Interleukin-6
- CRP:
-
C-reactive protein
- LDL:
-
Low-density lipoprotein
- BMI:
-
Body mass index
- CT:
-
Computed tomography
- MRI:
-
Magnetic resonance imaging
- HFpEF:
-
Heart failure with preserved ejection fraction
- AF:
-
Atrial fibrillation
- CI:
-
Confidence interval
- MACE:
-
Major adverse cardiovascular events
- SGLT-2:
-
Sodium-glucose cotransporter-2
- GLP-1:
-
Glucagon-like peptide-1
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Girón-Ulloa A, González-Domínguez E, Klimek RS, et al. Specific macrophage subsets accumulate in human subcutaneous and omental fat depots during obesity. Immunol Cell Biol. 2020;98(10):868–82. https://doi.org/10.1111/imcb.12380.
Weisberg S, McCann D, Desai M, Rosenbaum M, Leibel R, Ferrante A. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112:1796–808. https://doi.org/10.1172/JCI19246.
Aron-Wisnewsky J, Tordjman J, Poitou C, et al. Human adipose tissue macrophages: M1 and M2 cell surface markers in subcutaneous and omental depots and after weight loss. J Clin Endocrinol Metab. 2009;94:4619–23.
Cancello R, Henegar C, Viguerie N, et al. Reduction of macrophage infiltration and chemoattractant gene expression changes in white adipose tissue of morbidly obese subjects after surgery-induced weight loss. Diabetes. 2005;54:2277–86.
Zhu F, Wang A, Li Y, Liang R, Li D, Li B. Adipose Tissue-resident regulatory T cells. Adv Exp Med Biol. 2017;1011(153–162). https://doi.org/10.1007/978-94-024-1170-6_4
Fuster JJ, Ouchi N, Gokce N, Walsh K. Obesity-Induced changes in adipose tissue microenvironment and their impact on cardiovascular disease. Circ Res. 2017;118(11):1786–807. https://doi.org/10.1161/CIRCRESAHA.115.306885.Obesity-induced.
Khan T, Muise E, Iyengar P, et al. Metabolic dysregulation and adipose tissue fibrosis: role of collagen VI. Mol Cell Biol. 2009;29:1575–91. https://doi.org/10.1128/MCB.01300-08.
Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman J. Positional cloning of the mouse obese gene and its human homologue. Nature. 1994;372:425–32. https://doi.org/10.1038/372425a0.
Friedman J, Halaas J. Leptin and the regulation of body weight in mammals. Nature. 1998;395:763–70. https://doi.org/10.1038/27376.
Li L, Mamputu J, Wiernsperger N, Renier G. Signaling pathways involved in human vascular smooth muscle cell proliferation and matrix metalloproteinase-2 expression induced by leptin: inhibitory effect of metformin. Diabetes. 2005;54:2227–34.
Hongo S, Watanabe T, Arita S, et al. Leptin modulates ACAT1 expression and cholesterol efflux from human macrophages. Am J Physiol Endocrinol Metab. 2009;297:E474–82. https://doi.org/10.1152/ajpendo.90369.2008.
O’Rourke L, Gronning L, Yeaman S, Shepherd P. Glucose-dependent regulation of cholesterol ester metabolism in macrophages by insulin and leptin. J Biol Chem. 2002;277:42557–62. https://doi.org/10.1074/jbc.M202151200.
Bodary P, Gu S, Shen Y, Hasty A, Buckler J, Eitzman D. Recombinant leptin promotes atherosclerosis and thrombosis in apolipoprotein E-deficient mice. Arter Thromb Vasc Biol. 2005;25:e119–22. https://doi.org/10.1161/01.ATV.0000173306.47722.ec.
Zeadin M, Butcher M, Werstuck G, Khan M, Yee C, Shaughnessy S. Effect of leptin on vascular calcification in apolipoprotein E-deficient mice. Arter Thromb Vasc Biol. 2009;29:2069–75. https://doi.org/10.1161/ATVBAHA.109.195255.
Fain J, Madan A, Hiler M, Cheema P, Bahouth S. Comparison of the release of adipokines by adipose tissue, adipose tissue matrix, and adipocytes from visceral and subcutaneous abdominal adipose tissues of obese humans. Endocrinology. 2004;145:2273–82. https://doi.org/10.1210/en.2003-1336.
Schieffer B, Selle T, Hilfiker A, et al. Impact of interleukin-6 on plaque development and morphology in experimental atherosclerosis. Circulation. 2004;110:3493–500. https://doi.org/10.1161/01.CIR.0000148135.08582.97.
Steppan C, Bailey S, Bhat S, et al. The hormone resistin links obesity to diabetes. Nature. 2001;409:307–12. https://doi.org/10.1038/35053000.
Reilly M, Lehrke M, Wolfe M, Rohatgi A, Lazar M, Rader D. Resistin is an inflammatory marker of atherosclerosis in humans. Circulation. 2005;111:932–9. https://doi.org/10.1161/01.CIR.0000155620.10387.43.
Muse E, Feldman D, Blaha M, et al. The association of resistin with cardiovascular disease in the Multi-Ethnic Study of Atherosclerosis. Atherosclerosis. 2015;239:101–8.
Ryo M, Nakamura T, Kihara S, et al. Adiponectin as a biomarker of the metabolic syndrome. Circ J. 2004;68:975–81.
Nakamura Y, Shimada K, Fukuda D, et al. Implications of plasma concentrations of adiponectin in patients with coronary artery disease. Heart. 2004;90:528–33.
Pischon T, Girman C, Hotamisligil G, Rifai N, Hu F, Rimm E. Plasma adiponectin levels and risk of myocardial infarction in men. JAMA. 2004;291(1730–1737). https://doi.org/10.1001/jama.291.14.1730.
Schulze M, Shai I, Rimm E, Li T, Rifai N, Hu F. Adiponectin and future coronary heart disease events among men with type 2 diabetes. Diabetes. 2005;54:534–9.
Yamawaki H, Kuramoto J, Kameshima S, Usui T, Okada M, Hara Y. Omentin, a novel adipocytokine inhibits TNF-induced vascular inflammation in human endothelial cells. Biochem Biophys Res Commun. 2011;408:339–43. https://doi.org/10.1016/j.bbrc.2011.04.039.
Yousuf O, Mohanty BD, Martin SS, et al. High-sensitivity C-reactive protein and cardiovascular disease: a resolute belief or an elusive link? J Am Coll Cardiol. 2013;62(5):397–408. https://doi.org/10.1016/j.jacc.2013.05.016.
Ridker PM, Everett BM, Thuren T, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377(12):1119–31. https://doi.org/10.1056/nejmoa1707914. Results of this study showed that therapies that lower inflammation lead to a decrease in CVD events independent of lipid lowering therapy
Shin J, Syme C, Wang D, et al. Novel genetic locus of visceral fat and systemic inflammation. J Clin Endocrinol Metab. 2019;104(9):3735–42. https://doi.org/10.1210/jc.2018-02656.
Neeland I, Marso S, Ayers C, et al. Effects of liraglutide on visceral and ectopic fat in adults with overweight and obesity at high cardiovascular risk: a randomised, double-blind, placebo-controlled, clinical trial. Lancet Diabetes Endocrinol. 2021;9(9):595–605. https://doi.org/10.1016/S2213-8587(21)00179-0. GLP-1 agonists lead to favorable remodeling of adipose tissue composition with decreases in VAT concentration along with inflammatory markers such as CRP
Powell-Wiley TM, Poirier P, Burke LE, et al. Obesity and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2021;143(21). https://doi.org/10.1161/CIR.0000000000000973
Bogers R, Bemelmans W, Hoogenveen R, et al. BMI-CHD Collaboration Investigators. Association of overweight with increased risk of coronary heart disease partly independent of blood pressure and cholesterol levels: a meta-analysis of 21 cohort studies including more than 300 000 persons. Arch Intern Med. 2007;167:1720–1728. https://doi.org/10.1001/archinte.167.16.1720
McGill HJ. Fatty streaks in the coronary arteries and aorta. Lab Invest. Published online 1968:560–564.
Couillard C, Ruel G, Archer W, et al. Circulating levels of oxidative stress markers and endothelial adhesion molecules in men with abdominal obesity. J Clin Endocrinol Metab. 2005;90:6454–9. https://doi.org/10.1210/jc.2004-2438.
Engin A. Endothelial dysfunction in obesity. Adv Exp Med Biol. 2017;960:345–79. https://doi.org/10.1007/978-3-319-48382-5_15.
Rocha V, Libby P. Obesity, inflammation, and atherosclerosis. Nat Rev Cardiol. 2009;6:399–409. https://doi.org/10.1038/nrcardio.2009.55.
Ross R. Atherosclerosis is an inflammatory disease. Am Hear J. 1999;138(pt 2):S419–20. https://doi.org/10.1016/s0002-8703(99)70266-8.
Schindler T, Cardenas J, Prior J, et al. Relationship between increasing body weight, insulin resistance, inflammation, adipocytokine leptin, and coronary circulatory function. J Am Coll Cardiol. 2006;47:1188–95. https://doi.org/10.1016/j.jacc.2005.10.062.
Zieske A, Malcom G, Strong J. Natural history and risk factors of ath- erosclerosis in children and youth: the PDAY study. Pediatr Pathol Mol Med. 2002;21:213–37. https://doi.org/10.1080/15227950252852104.
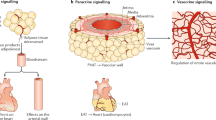
Mancio J, Oikonomou EK, Antoniades C. Perivascular adipose tissue and coronary atherosclerosis. Heart. 2018;104(20):1654–62. https://doi.org/10.1136/heartjnl-2017-312324.
Alpert M, Lavie C, Agrawal H, Aggarwal K, Kumar S. Obesity and heart failure: epidemiology, pathophysiology, clinical manifestations, and management. Transl Res. 2014;164:345–56. https://doi.org/10.1016/j.trsl.2014.04.010.
Kenchaiah S, Evans J, Levy D, et al. Obesity and the risk of heart failure. N Engl J Med. 2002;347(305–313). https://doi.org/10.1056/NEJMoa020245
Pandey A, Patel K, Vaduganathan M, et al. Physical activity, fitness, and obesity in heart failure with preserved ejection fraction. JACC Hear Fail. 2018;6:975–82. https://doi.org/10.1016/j.jchf.2018.09.006.
Pandey A, LaMonte M, Klein L, et al. Relationship between physical activity, body mass index, and risk of heart failure. J Am Coll Cardiol. 2017;69:1129–42. https://doi.org/10.1016/j.jacc.2016.11.081.
Harada T, Obokata M. Obesity-related heart failure with preserved ejection fraction: pathophysiology, diagnosis, and potential therapies. Hear Fail Clin. 16(3):357–368. https://doi.org/10.1016/j.hfc.2020.02.004
Csige I, Ujvárosy D, Szabó Z, et al. The impact of obesity on the cardiovascular system. J Diabetes Res. Published online 2018:3407306. https://doi.org/10.1155/2018/3407306
Mouton AJ, Li X, Hall ME, Hall JE. Obesity, hypertension, and cardiac dysfunction novel roles of immunometabolism in macrophage activation and inflammation. Circ Res. Published online 2020:789–806. https://doi.org/10.1161/CIRCRESAHA.119.312321
Obokata M, Reddy Y, Pislaru S, Melenovsky V, Borlaug B. Evidence supporting the existence of a distinct obese phenotype of heart failure with preserved ejection fraction. Circulation. 2017;136:6–19. https://doi.org/10.1161/CIRCULATIONAHA.116.026807.
Kitzman D, Lam C. Obese heart failure with preserved ejection fraction phenotype: from pariah to central player. Circulation. 2017;136:20–3. https://doi.org/10.1161/CIRCULATIONAHA.117.028365.
Wong C, Sullivan T, Sun M, et al. Obesity and the risk of incident, post-operative, and post-ablation atrial fibrillation: a meta-analysis of 626,603 individuals in 51 studies. JACC Clin Electrophysiol. 2015;1(139–152). https://doi.org/10.1016/j.jacep.2015.04.004
Huxley R, Lopez F, Folsom A, et al. Absolute and attributable risks of atrial fibrillation in relation to optimal and borderline risk factors: the Atherosclerosis Risk in Communities (ARIC) study. Circulation. 2011;123:1501–8.
Tsang T, Barnes M, Miyasaka Y, et al. Obesity as a risk factor for the progression of paroxysmal to permanent atrial fibrillation: a longitudinal cohort study of 21 years. Eur Hear J. 2008;29:2227–33. https://doi.org/10.1093/eurheartj/ehn324.
Gami A, Hodge D, Herges R, et al. Obstructive sleep apnea, obesity, and the risk of incident atrial fibrillation. J Am Coll Cardiol. 2007;49:565–71.
Mangiafico V, Saberwal B, Lavalle C, et al. Impact of obesity on atrial fibrillation ablation. Arch Cardiovasc Dis. 2020;113(8–9):551–63. https://doi.org/10.1016/j.acvd.2020.03.023.
Lavie CJ, Pandey A, Lau DH, Alpert MA, Sanders P. Obesity and atrial fibrillation prevalence, pathogenesis, and prognosis: effects of weight loss and exercise. J Am Coll Cardiol. 2017;70(16):2022–35. https://doi.org/10.1016/j.jacc.2017.09.002.
Mahajan R, Nelson A, Pathak R, et al. Electroanatomical remodeling of the atria in obesity: impact of adjacent epicardial fat. JACC Clin Electrophysiol. 2018;4:1529–40. https://doi.org/10.1016/j.jacep.2018.08.014.
Hatem S, Redheuil A, Grandjbakhch E. Cardiac adipose tissue and atrial fibrillation. Cardiovasc Res. 2016;109:502–9.
Al-Rawahi M, Proletti R, Thansoulis G. Pericardial fat and atrial fibrillation: epidemiology, mechanisms and interventions. Int J Cardiol. 2015;195:98–103.
Thanasoullis G, Massaro J, O’Donnel C. Pericardial fat is associated with prevalent atrial fibrillation: the Framingham Study. Circ Arrhythmia Electrophysiol. 2010;3:345–50.
Ibrahim M. Subcutaneous and visceral adipose tissue: structural and functional differences. Obes Rev. 2010;11(1):11–8.
Liu J, Fox CS, Hickson DA, et al. Impact of abdominal visceral and subcutaneous adipose tissue on cardiometabolic risk factors : the Jackson Heart Study. 2010;95(December):5419-5426https://doi.org/10.1210/jc.2010-1378
Neeland I, Turer A, Ayers C, Al. E. Body fat distribution and incident cardiovascular disease in obese adults. J Am Coll Cardiol. 2015;65(19):2150–2151. https://doi.org/10.1016/j.jacc.2015.01.061
Neeland I, Hughes C, Ayers C, Malloy C, Jin E. Effects of visceral adiposity on glycerol pathways in gluconeogenesis. Metabolism. 2017;67:80–9.
Smith U, Kahn B. Adipose tissue regulates insulin sensitivity: role of adipogenesis, de novo lipogenesis and novel lipids. J Intern Med. 2016;280:465–75.
Vasan S, Karpe F. Fat, yet fit. Nat Rev Endocrinol. 2016;12:375–6.
Chang E, Varghese M, Singer K. Gender and sex differences in adipose tissue. Curr Diab Rep. 2018;18(9):69. https://doi.org/10.1007/s11892-018-1031-3.
Fried S, Lee M, Karastergiou K. Shaping fat distribution: new insights into the molecular determinants of depot and sex dependent adipose biology. Obes (Silver Spring). 2015;23(7):1345–52.
Schwartz R, Shuman W, Larson V, Cain K, Fellingham G, Beard J, et al. The effect of intensive endurance exercise training on body fat distribution in young and older men. Metabolism. 1991;40(5):545–51.
Link J, Hasin-Brumshtein Y, Cantor R, Chen X, Arnold A, Lusis A, et al. Diet, gonadal sex, and sex chromosome complement influence white adipose tissue miRNA expression. BMC Genomics. 2017;18(1):89.
Shulman G. Ectopic fat in insulin resistance, dyslipidemia, and cardiometabolic disease. N Engl J Med. 2014;371(23):2237–8.
Lu S, McKenna S, Cologer-Clifford A, Nau E, Simon N. Androgen receptor in mouse brain: sex differences and similarities in autoregulation. Endocrinology. 1998;139(4):1594–601.
Locke A, Kahali B, Berndt S, Justice A, Pers T, Day F, et al. Genetic studies of body mass index yield new insights for obesity biology. Nature. 2015;518(7538):197–206.
Macotela Y, Boucher J, Tran T, Kahn C. Sex and depot differences in adipocyte insulin sensitivity and glucose metabolism. Diabetes. 2009;58(4):803–12.
Ornoy A, Giron S, Aner R, Goldstein M, Boyan B, Schwartz Z. Gender dependent effects of testosterone and 17 beta-estradiol on bone growth and modelling in young mice. Bone Min. 1994;24(1):43–58.
Chen X, McClusky R, Chen J, Beaven S, Tontonoz P, Arnold A et al. The number of x chromosomes causes sex differences in adiposity in mice. PLoS Genet. 2012;8(5):e1002709.
Xie H, Lim B, Lodish H. MicroRNAs induced during adipogenesis that accelerate fat cell development are downregulated in obesity. Diabetes. 2009;58(5):1050–7.
Trajkovski M, Ahmed K, Esau C, Stoffel M. MyomiR-133 regulates brown fat differentiation through Prdm16. Nat Cell Biol. 2012;14(12):1330–5.
White U, Tchoukalova Y. Sex dimorphism and depot differences in adipose tissue function. Biochim Biophys Acta. 2014;1842(3):377–92.
Tchoukalova Y, Koutsari C, Votruba S, Tchkonia T, Giorgadze N, Thomou T, et al. Sex and depot-dependent differences in adipogenesis in normal-weight humans. Obes (Silver Spring). 2010;18(10):1875–80.
Tchoukalova Y, Koutsari C, Karpyak M, Votruba S, Wendland E, Jensen M. Subcutaneous adipocyte size and body fat distribution. Am J Clin Nutr. 2008;87(1):56–63.
Shadid S, Koutsari C, Jensen M. Direct free fatty acid uptake into human adipocytes in vivo: relation to body fat distribution. Diabetes. 2007;56(5):1369–75.
Basu S, Haghiac M, Surace P, Challier J, Guerre-Millo M, Singh K, et al. Pregravid obesity associates with increased maternal endotoxemia and metabolic inflammation. Obes (Silver Spring). 2011;19(3):476–82.
Wells JCK. Ethnic variability in adiposity, thrifty phenotypes and cardiometabolic risk: addressing the full range of ethnicity, including those of mixed ethnicity. Obes Rev. 2012;13(SUPPL.2):14–29. https://doi.org/10.1111/j.1467-789X.2012.01034.x
Rønn PF, Andersen GS, Lauritzen T, et al. Abdominal visceral and subcutaneous adipose tissue and associations with cardiometabolic risk in Inuit, Africans and Europeans: a cross-sectional study. BMJ Open. 2020;10(9):e038071. https://doi.org/10.1136/bmjopen-2020-038071.
Sutton-Tyrrell K, Wildman R, Matthews K, Chae C, Lasley B, Brockwell S, et al. Sex- hormone-binding globulin and the free androgen index are related to cardiovascular risk factors in multiethnic premenopausal and perimenopausal women enrolled in the Study of Women Across the Nation (SWAN). Circulation. 2005;111(10):1242–9.
Perry A, Martin L. Race differences in obesity and its relationship to the sex hormone milieu. Horm Mol Biol Clin Investig. 2014;19(3):151–61.
Nazare JA, Smith JD, Borel AL, et al. Ethnic influences on the relations between abdominal subcutaneous and visceral adiposity, liver fat, and cardiometabolic risk profile: the international study of prediction of intra-abdominal adiposity and its relationship with cardiometabolic risk/intra-. Am J Clin Nutr. 2012;96(4):714–26. https://doi.org/10.3945/ajcn.112.035758.
Mongraw-chaffin M, Gujral UP, Kanaya AM, Namratha R. Relation of ectopic fat with atherosclerotic cardiovascular disease risk score in South Asians living in the USA (from the Mediators of Atherosclerosis in South Asians Living in America [MASALA] Study). Am J Cardiol. 2018;121(3):315–21. https://doi.org/10.1016/j.amjcard.2017.10.026.Relation.
Liu J, Coady S, Carr JJ, Hoffmann U, Taylor HA, Fox CS. Differential associations of abdominal visceral, subcutaneous adipose tissue with cardiometabolic risk factors between African and European Americans. Obesity. 2014;22(3):811–8. https://doi.org/10.1002/oby.20307.
Cooper RS. Social inequality, ethnicity and cardiovascular disease. Int J Epidemiol. 2001;30(SUPPL. 1):48–52. https://doi.org/10.1093/ije/30.suppl_1.S48.
Gill C, Lee M, Vatcheva KP, et al. Association of visceral adipose tissue and subclinical atherosclerosis in US-born immigrants. Published online. 2020. https://doi.org/10.1161/JAHA.120.017373.
Neeland IJ, Ross R, Després J, et al. Visceral and ectopic fat , atherosclerosis , and cardiometabolic disease : a position statement. :715–725. https://doi.org/10.1016/S2213-8587(19)30084-1
Wing R, Bolin P, Brancati F, et al. Cardiovascular effects of intensive lifestyle intervention in type 2 diabetes. N Engl J Med. 2013;369:145–54.
Moreno-Navarrete J, Catalan V, Ortega F, et al. Circulating omentin concentration increases after weight loss. 2010, 7, 27. Nutr Metab. 2010;7:27
Lancha A, Moncada R, Valenti V, et al. Effect of sleeve gastrectomy on osteopontin circulating levels and expression in adipose tissue and liver in rats. Obes Surg. 2014;24:1702–8.
Roager HM, Vogt JK, Kristensen M, et al. Whole grain-rich diet reduces body weight and systemic low-grade inflammation without inducing major changes of the gut microbiome : a randomised. Published online 2019:83–93. https://doi.org/10.1136/gutjnl-2017-314786
Gepner Y, Shelef I, Schwarzfuchs D, Al E. Effect of distinct lifestyle interventions on mobilization of fat storage pools: CENTRAL magnetic resonance imaging randomized controlled trial. Circulation. 2018;137:1143–57.
Myette-côté É, Durrer C, Neudorf H, et al. The effect of a short-term low-carbohydrate, high-fat diet with or without postmeal walks on glycemic control and inflammation in type 2 diabetes : a randomized trial. 2021;(47):1210–1219. https://doi.org/10.1152/ajpregu.00240.2018
Tran HQ, Bretin A, Adeshirlarijaney A, et al. “Western Diet’’’-induced adipose inflammation requires a complex gut microbiota.” Cell Mol Gastroenterol Hepatol. 9(2):313–333. https://doi.org/10.1016/j.jcmgh.2019.09.009
Rosenbaum M, Hall KD, Guo J, et al. Glucose and lipid homeostasis and inflammation in humans following an isocaloric ketogenic diet. 2019;27(6):971-981https://doi.org/10.1002/oby.22468.GLUCOSE
Ross R, Bradshaw A. The future of obesity reduction: beyond weight loss. Nat Rev Endocrinol. 2009;5:319–25.
Despres J. Obesity and cardiovascular disease: weight loss is not the only target. Can J Cardiol. 2015;21:216–22.
Janiszewski P, Ross R. Physical activity in the treatment of obesity: beyond body weight reduction. Appl Physiol Nutr Metab. 2007;32:512–22.
Frodermann V, Rohde D, Courties G, et al. Exercise reduces inflammatory cell production and cardiovascular inflammation via instruction of hematopoietic progenitor cells. Nat Med. 2019;25(11):1761–71. https://doi.org/10.1038/s41591-019-0633-x.Exercise.
Rao S, Pandey A, Garg S, et al. Effect of exercise and pharmacological interventions on visceral adiposity: a systematic review and meta-analysis of long-term randomized controlled trials. Mayo Clin Proc. 94(2):211–224. https://doi.org/10.1016/j.mayocp.2018.09.019
Pedersen LR, Olsen RH, Anholm C, et al. Effects of 1 year of exercise training versus combined exercise training and weight loss on body composition, low-grade inflammation and lipids in overweight patients with coronary artery disease: a randomized trial. Cardiovasc Diabetol. 2019;18(1):1–13. https://doi.org/10.1186/s12933-019-0934-x.
Khera R, Pandey A, Chandar AK, et al. Effects of weight-loss medications on cardiometabolic risk profiles: a systematic review and network meta-analysis. Gastroenterology. 2018;154(5):1309-1319.e7. https://doi.org/10.1053/j.gastro.2017.12.024.
Xu L, Ota T. Emerging roles of SGLT2 inhibitors in obesity and insulin resistance: focus on fat browning and macrophage polarization. Adipocyte. 2018;7(2):121–8. https://doi.org/10.1080/21623945.2017.1413516.
Xu L, Nagata N, Nagashimada M, et al. SGLT2 inhibition by empagliflozin promotes fat utilization and browning and attenuates inflammation and insulin resistance by polarizing M2 macrophages in diet induced obese mice. EBioMedicine. 2017;20:137–49. https://doi.org/10.1016/j.ebiom.2017.05.028.
Heffron S, Parham J, Pendse J, Alemán J. Treatment of obesity in mitigating metabolic risk. Circ Res. 126(11):1646–1665. https://doi.org/10.1161/CIRCRESAHA.119.315897. Epub 2020 May 21. Erratum in: Circ Res. 2020 Jul 3;127(2):e79.
Labrecque J, Laforest S, Michaud A, Biertho L, Tchernof A. Impact of bariatric surgery on white adipose tissue inflammation. Can J Diabetes. 2017;41(4):407–17. https://doi.org/10.1016/j.jcjd.2016.12.003.
Min T, Prior SL, Dunseath G, Churm R, Barry JD, Stephens JW. Temporal effects of bariatric surgery on adipokines, inflammation and oxidative stress in subjects with impaired glucose homeostasis at 4 years of follow-up. Obes Surg. 2020;30(5):1712–8. https://doi.org/10.1007/s11695-019-04377-3.
Stephens J, Min T, Dunseath G, Churm R, Barry J, Prior S. Temporal effects of laparoscopic sleeve gastrectomy on adipokines, inflammation, and oxidative stress in patients with impaired glucose homeostasis. Surg Obes Relat Dis. 15(12):2011–2017. 1https://doi.org/10.1016/j.soard.2019.04.006
Schauer P, Bhatt D, Kirwan J, et al. Bariatric Surgery versus intensive medical therapy for diabetes - 5-year outcomes. New Engl J Med. 2017;376:641–51.
Sjostrom C, Peltonen M, Wedel H, Sjostrom L. Differentiated long-term effects of intentional weight loss on diabetes and hypertension. Hypertension. 2000;36:20–5.
Maciejewski M, Arterburn D, Van Scoyoc L, et al. Bariatric surgery and long-term durability of weight loss. JAMA Surg. 2016;151:1046–55.
Lehmann S, Linder N, Retschlag U, et al. MRI assessment of changes in adipose tissue parameters after bariatric surgery. PLoS One. 13(11):e0206735. https://doi.org/10.1371/journal.pone.0206735
Adami G, Carbone F, Montecucco F, Camerini G, Cordera R. Adipose tissue composition in obesity and after bariatric surgery. Obes Surg. 29(9):3030–3038.https://doi.org/10.1007/s11695-019-04030-z
Rossi I, Omotosho P, Poirier J, Spagnoli A, Torquati A. Roux-en-Y gastric bypass decreases serum inflammatory markers and cardiovascular risk factors in obese diabetics. Surgery. 169(3):539–542. https://doi.org/10.1016/j.surg.2020.09.039
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interests
Dr. Neeland reports receiving consulting fees/honoraria from Nestle Health Sciences, Boehringer Ingelheim/Lilly Alliance, and Merck & Co. The other authors declare no competing interests.
Human and Animal Rights
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Macrovascular Complications in Diabetes
Rights and permissions
About this article
Cite this article
Rana, M.N., Neeland, I.J. Adipose Tissue Inflammation and Cardiovascular Disease: An Update. Curr Diab Rep 22, 27–37 (2022). https://doi.org/10.1007/s11892-021-01446-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11892-021-01446-9