Abstract
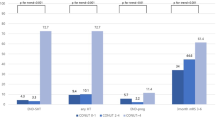
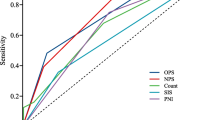
Although the assessment of nutrition is essential for stroke patients, detailed associations between nutritional status at admission, subsequent complications, and clinical outcomes in patients with acute intracerebral hemorrhage (ICH) are unclear. We aimed to elucidate these associations using the Controlling Nutritional Status (CONUT) score. Consecutive patients with acute ICH were investigated. Nutritional status was evaluated using the CONUT score, calculated from the serum albumin level, lymphocyte count, and total cholesterol level. Subsequent complications, such as hemorrhage expansion (HE) during the acute stage and aspiration pneumonia during hospitalization, were evaluated. Poor outcome was defined as a modified Rankin Scale score of ≥ 3 at 3 months. Of the 721 patients, 49 had HE, 111 had aspiration pneumonia, and 409 had poor outcomes. Patients with HE had significantly lower total cholesterol levels than those without HE. Patients with aspiration pneumonia had significantly lower albumin levels, lower lymphocyte counts, and higher CONUT scores than those without aspiration pneumonia. Patients with poor outcomes had significantly lower albumin levels, lower lymphocyte counts, lower total cholesterol levels, and higher CONUT scores than those with good outcomes. Multivariable logistic analysis showed that higher CONUT scores were independently associated with poor outcome (odds ratio, 1.28; 95% confidence interval, 1.09–1.49; P = 0.002) after adjusting for baseline characteristics, HE, and aspiration pneumonia. Each component of CONUT was a useful predictor of subsequent complications. Malnutrition, determined using the CONUT score, was independently associated with poor outcomes in patients with ICH after adjusting for these complications.


Similar content being viewed by others
References
Foley NC, Salter KL, Robertson J, Teasell RW, Woodbury MG (2009) Which reported estimate of the prevalence of malnutrition after stroke is valid? Stroke 40:e66–e74. https://doi.org/10.1161/STROKEAHA.108.518910
Chen N, Li Y, Fang J, Lu Q, He L (2019) Risk factors for malnutrition in stroke patients: a meta-analysis. Clin Nutr 38:127–135. https://doi.org/10.1016/j.clnu.2017.12.014
Yoo SH, Kim JS, Kwon SU, Yun SC, Koh JY, Kang DW (2008) Undernutrition as a predictor of poor clinical outcomes in acute ischemic stroke patients. Arch Neurol 65:39–43. https://doi.org/10.1001/archneurol.2007.12
FOOD Trial Collaboration (2003) Poor nutritional status on admission predicts poor outcomes after stroke: observational data from the FOOD trial. Stroke 34:1450–1456. https://doi.org/10.1161/01.STR.0000074037.49197.8C
Cai ZM, Wu YZ, Chen HM, Feng RQ, Liao CW, Ye SL, Liu ZP, Zhang MM, Zhu BL (2020) Being at risk of malnutrition predicts poor outcomes at 3 months in acute ischemic stroke patients. Eur J Clin Nutr 74:796–805. https://doi.org/10.1038/s41430-020-0605-8
Zhang M, Ye S, Huang X, Sun L, Liu Z, Liao C, Feng R, Chen H, Wu Y, Cai Z, Lin Q (2020) Comparing the prognostic significance of nutritional screening tools and ESPEN-DCM on 3-month and 12-month outcomes in stroke patients. Clin Nutr. https://doi.org/10.1016/j.clnu.2020.11.001
De Ulíbarri JI, González-Madroño A, De Villar NG, González P, González B, Mancha A, Rodriguez F, Fernández G et al (2005) CONUT: a tool for controlling nutritional status. First validation in a hospital population. Nutr Hosp 20:38–45
Naito H, Nezu T, Hosomi N, Aoki S, Kinoshita N, Kuga J, Shimomura R, Araki M, Ueno H, Ochi K, Maruyama H (2018) Controlling nutritional status score for predicting 3-mo functional outcome in acute ischemic stroke. Nutrition 55–56:1–6. https://doi.org/10.1016/j.nut.2018.02.025
Naito H, Hosomi N, Nezu T, Kuzume D, Aoki S, Morimoto Y, Yoshida T, Shiga Y, Kinoshita N, Ueno H, Maruyama H (2020) Prognostic role of the controlling nutritional status score in acute ischemic stroke among stroke subtypes. J Neurol Sci 416:116984. https://doi.org/10.1016/j.jns.2020.116984
Xiang W, Chen X, Ye W, Li J, Zhang X, Xie D (2020) Prognostic nutritional index for predicting 3-month outcomes in ischemic stroke patients undergoing thrombolysis. Front Neurol 11:599. https://doi.org/10.3389/fneur.2020.00599
Qi H, Yang X, Hao C, Zhang F, Pang X, Zhou Z, Dai J (2019) Clinical value of controlling nutritional status score in patients with aneurysmal subarachnoid hemorrhage. World Neurosurg 126:e1352–e1358. https://doi.org/10.1016/j.wneu.2019.03.100
Dziedzic T, Pera J, Klimkowicz A, Turaj W, Slowik A, Rog TM, Szczudlik A (2006) Serum albumin level and nosocomial pneumonia in stroke patients. Eur J Neurol 13:299–301. https://doi.org/10.1111/j.1468-1331.2006.01210.x
Dziedzic T, Slowik A, Szczudlik A (2004) Serum albumin level as a predictor of ischemic stroke outcome. Stroke 35:e156–e158. https://doi.org/10.1161/01.STR.0000126609.18735.be
Morotti A, Marini S, Jessel MJ et al (2017) Lymphopenia, infectious complications, and outcome in spontaneous intracerebral hemorrhage. Neurocrit Care 26:160–166. https://doi.org/10.1007/s12028-016-0367-2
Mann G, Hankey GJ, Cameron D (1999) Swallowing function after stroke: prognosis and prognostic factors at 6 months. Stroke 30:744–748. https://doi.org/10.1161/01.str.30.4.744
Jauch EC, Saver JL, Adams HP et al (2013) Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 44:870–947. https://doi.org/10.1161/STR.0b013e318284056a
Noda H, Iso H, Irie F et al (2009) Low-density lipoprotein cholesterol concentrations and death due to intraparenchymal hemorrhage: The Ibaraki Prefectural Health Study. Circulation 119:2136–2145. https://doi.org/10.1161/CIRCULATIONAHA.108.795666
Ma C, Gurol ME, Huang Z, Lichtenstein AH, Wang X, Wang Y, Neumann S, Wu S, Gao X (2019) Low-density lipoprotein cholesterol and risk of intracerebral hemorrhage: a prospective study. Neurology 93:e445–e457. https://doi.org/10.1212/WNL.0000000000007853
Elkhatib THM, Shehta N, Bessar AA (2019) Hematoma expansion predictors: laboratory and radiological risk factors in patients with acute intracerebral hemorrhage: a prospective observational study. J Stroke Cerebrovasc Dis 28:2177–2186. https://doi.org/10.1016/j.jstrokecerebrovasdis.2019.04.038
Brott T, Adams HP, Olinger CP et al (1989) Measurements of acute cerebral infarction: a clinical examination scale. Stroke 20:864–870. https://doi.org/10.1161/01.str.20.7.864
Salman RAS, Frantzias J, Lee RJ et al (2018) Absolute risk and predictors of the growth of acute spontaneous intracerebral haemorrhage: a systematic review and meta-analysis of individual patient data. Lancet Neurol 17:885–894. https://doi.org/10.1016/S1474-4422(18)30253-9
Harms H, Hoffmann S, Malzahn U, Ohlraun S, Heuschmann P, Meisel A (2012) Decision-making in the diagnosis and treatment of stroke-associated pneumonia. J Neurol Neurosurg Psychiatry 83:1225–1230. https://doi.org/10.1136/jnnp-2012-302194
Li Q, Zhang G, Huang YJ et al (2015) Blend sign on computed tomography: novel and reliable predictor for early hematoma growth in patients with intracerebral hemorrhage. Stroke 46:2119–2123. https://doi.org/10.1161/STROKEAHA.115.009185
González-Madroño A, Mancha A, Rodríguez FJ, Culebras J, de Ulibarri JI (2012) Confirming the validity of the CONUT system for early detection and monitoring of clinical undernutrition: comparison with two logistic regression models developed using SGA as the gold standard. Nutr Hosp 27:564–571. https://doi.org/10.1590/S0212-16112012000200033
Nakagomi A, Kohashi K, Morisawa T, Kosugi M, Endoh I, Kusama Y, Atarashi H, Shimizu W (2016) Nutritional status is associated with inflammation and predicts a poor outcome in patients with chronic heart failure. J Atheroscler Thromb 23:713–727. https://doi.org/10.5551/jat.31526
Iseki Y, Shibutani M, Maeda K et al (2015) Impact of the preoperative controlling nutritional status (CONUT) score on the survival after curative surgery for colorectal cancer. PLoS ONE 10:e0132488. https://doi.org/10.1371/journal.pone.0132488
Morotti A, Marini S, Lena UK et al (2017) Significance of admission hypoalbuminemia in acute intracerebral hemorrhage. J Neurol 264:905–911. https://doi.org/10.1007/s00415-017-8451-x
Di Napoli M, Behrouz R, Topel CH et al (2017) Hypoalbuminemia, systemic inflammatory response syndrome, and functional outcome in intracerebral hemorrhage. J Crit Care 41:247–253. https://doi.org/10.1016/j.jcrc.2017.06.002
Chen Q, Liu J, Xu H et al (2019) Association between eosinophilic leukocyte count and hematoma expansion in acute spontaneous intracerebral hemorrhage. Front Neurol 10:1164. https://doi.org/10.3389/fneur.2019.01164
Tirschwell DL, Smith NL, Heckbert SR, Lemaitre RN, Longstreth WT, Psaty BM (2004) Association of cholesterol with stroke risk varies in stroke subtypes and patient subgroups. Neurology 63:1868–1875. https://doi.org/10.1212/01.wnl.0000144282.42222.da
Ooneda G, Yoshida Y, Suzuki K, Shinkai H, Hori S, Kobori K, Takayama Y, Sekiguchi M (1978) Smooth muscle cells in the development of plasmatic arterionecrosis, arteriosclerosis, and arterial contraction. Blood Vessels 15:148–156. https://doi.org/10.1159/000158160
Tandon N, Harmon JT, Rodbard D, Jamieson GA (1983) Thrombin receptors define responsiveness of cholesterol-modified platelets. J Biol Chem 258:11840–11845. https://doi.org/10.1016/S0021-9258(17)44308-0
Sato M, Ido Y, Yoshimura Y, Mutai H (2019) Relationship of malnutrition during hospitalization with functional recovery and postdischarge destination in elderly stroke patients. J Stroke Cerebrovasc Dis 28:1866–1872. https://doi.org/10.1016/j.jstrokecerebrovasdis.2019.04.012
Nii M, Maeda K, Wakabayashi H, Nishioka S, Tanaka A (2016) Nutritional improvement and energy intake are associated with functional recovery in patients after cerebrovascular disorders. J Stroke Cerebrovasc Dis 25:57–62. https://doi.org/10.1016/j.jstrokecerebrovasdis.2015.08.033
Kokura Y, Maeda K, Wakabayashi H, Nishioka S, Higashi S (2016) High nutritional-related risk on admission predicts less improvement of functional independence measure in geriatric stroke patients: a retrospective cohort study. J Stroke Cerebrovasc Dis 25:1335–1341. https://doi.org/10.1016/j.jstrokecerebrovasdis.2016.01.048
Nozoe M, Kanai M, Kubo H, Yamamoto M, Shimada S, Mase K (2019) Prestroke sarcopenia and functional outcomes in elderly patients who have had an acute stroke: a prospective cohort study. Nutrition 66:44–47. https://doi.org/10.1016/j.nut.2019.04.011
Welch AA (2014) Nutritional influences on age-related skeletal muscle loss. Proc Nutr Soc 73:16–33. https://doi.org/10.1017/S0029665113003698
Millward DJ (2012) Nutrition and sarcopenia: evidence for an interaction. Proc Nutr Soc 71:566–575. https://doi.org/10.1017/S0029665112000201
Jönsson AC, Lindgren I, Norrving B, Lindgren A (2008) Weight loss after stroke: a population-based study from the Lund Stroke Register. Stroke 39:918–923. https://doi.org/10.1161/STROKEAHA.107.497602
Koga M, Toyoda K, Yamagami H et al (2012) Systolic blood pressure lowering to 160 mmHg or less using nicardipine in acute intracerebral hemorrhage: a prospective, multicenter, observational study (the Stroke Acute Management with Urgent Risk-factor Assessment and Improvement-Intracerebral hemorrhage study). J Hypertens 30:2357–2364. https://doi.org/10.1097/HJH.0b013e328359311b
Funding
This study was supported by research grants from the Japan Society for the Promotion of Science KAKENHI (Grant numbers: 20K16579).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: YS, TN, RS, KS, TH, YT, SA and TK. Performed the experiments: YS, TN, RS, KS, TH, YT, SA and TK. Analyzed the data: YS and TN. Contributed reagents/materials/analysis tools: TN, RS, KS, TH, YT, SA, and TK. Wrote the paper: YS, TN, NH, TK, and H Maruyama.
Corresponding author
Ethics declarations
Conflict of interest
Dr. Maruyama received Grants-in-Aid for Scientific Research (20K07887) and research support from Eisai, Pfizer, Takeda Pharmaceutical, Otsuka Pharmaceutical, Nihon Pharmaceutical, Shionogi, Teijin Pharma, Fuji Film, Sumitomo Dainippon Pharma, Nihon Medi-Physics, Bayer, MSD, Daiichi Sankyo, Kyowa Hakko Kirin, Sanofi, Novartis, Kowa Pharmaceutical, Astellas Pharma, Tsumura, and Japan Blood Products Organization, which are unrelated to the submitted work. All other authors declare that they have no conflict of interest.
Ethical approval
This study complied with the Declaration of Helsinki for investigations involving humans, and the study protocol was approved by the Ethics Committee of the Brain Attack Center Ota Memorial Hospital (217).
Informed consent
The requirement for informed consent was waived by the ethics committee considering the retrospective, noninterventional nature of the study.
Data availability
The data that support the findings of this study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shiga, Y., Nezu, T., Shimomura, R. et al. Various effects of nutritional status on clinical outcomes after intracerebral hemorrhage. Intern Emerg Med 17, 1043–1052 (2022). https://doi.org/10.1007/s11739-021-02901-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-021-02901-x