Abstract
Purpose
Radiomics of vertebral bone structure is a promising technique for identification of osteoporosis. We aimed at assessing the accuracy of machine learning in identifying physiological changes related to subjects’ sex and age through analysis of radiomics features from CT images of lumbar vertebrae, and define its generalizability across different scanners.
Materials and methods
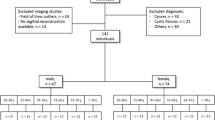
We annotated spherical volumes-of-interest (VOIs) in the center of the vertebral body for each lumbar vertebra in 233 subjects who had undergone lumbar CT for back pain on 3 different scanners, and we evaluated radiomics features from each VOI. Subjects with history of bone metabolism disorders, cancer, and vertebral fractures were excluded. We performed machine learning classification and regression models to identify subjects’ sex and age respectively, and we computed a voting model which combined predictions.
Results
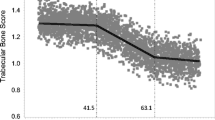
The model was trained on 173 subjects and tested on an internal validation dataset of 60. Radiomics was able to identify subjects’ sex within single CT scanner (ROC AUC: up to 0.9714), with lower performance on the combined dataset of the 3 scanners (ROC AUC: 0.5545). Higher consistency among different scanners was found in identification of subjects’ age (R2 0.568 on all scanners, MAD 7.232 years), with highest results on a single CT scanner (R2 0.667, MAD 3.296 years).
Conclusion
Radiomics features are able to extract biometric data from lumbar trabecular bone, and determine bone modifications related to subjects’ sex and age with great accuracy. However, acquisition from different CT scanners reduces the accuracy of the analysis.




Similar content being viewed by others
Abbreviations
- AUROC:
-
Area Under the Receiver Operating Characteristic curve
- BMD:
-
Bone Mineral Density
- CT:
-
Computed Tomography
- DXA:
-
Dual-Energy X-ray Absorptiometry
- FRAX:
-
Fracture Risk Assessment Tool
- ICC:
-
Intraclass Correlation Coefficient
- MAD:
-
Median Absolute Deviation
- VF:
-
Vertebral Fracture
References
Baum T, Gräbeldinger M, Räth C et al (2014) Trabecular bone structure analysis of the spine using clinical MDCT: can it predict vertebral bone strength? J Bone Miner Metab 32:56–64. https://doi.org/10.1007/s00774-013-0465-6
Hemmatian H, Bakker AD, Klein-Nulend J, Van Lenthe H, G, (1914) Aging, Osteocytes, and Mechanotransduction. Curr Osteoporos Rep. https://doi.org/10.1007/s11914-017-0402-z
Russo CR, Lauretani F, Bandinelli S et al (2003) Aging bone in men and women: beyond changes in bone mineral density. Osteoporos Int 14:531–538. https://doi.org/10.1007/s00198-002-1322-y
Yu A, Huang M, Wang L et al (2023) Age and gender differences in vertebral bone marrow adipose tissue and bone mineral density based on MRI and quantitative CT. Eur J Radiol 159:110669. https://doi.org/10.1016/j.ejrad.2022.110669
Kanis JA, Cooper C, Rizzoli R, Reginster J-Y (2019) European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int 30:3–44. https://doi.org/10.1007/s00198-018-4704-5
Tamimi I, Cortes ARG, Sánchez-Siles J-M et al (2020) Composition and characteristics of trabecular bone in osteoporosis and osteoarthritis. Bone 140:115558. https://doi.org/10.1016/j.bone.2020.115558
Lambin P, Leijenaar RTH, Deist TM et al (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14:749–762. https://doi.org/10.1038/nrclinonc.2017.141
Hong JH, Jung J-Y, Jo A et al (2021) Development and validation of a radiomics model for differentiating bone islands and osteoblastic bone metastases at abdominal CT. Radiology 299:626–632. https://doi.org/10.1148/radiol.2021203783
Staal FCR, van der Reijd DJ, Taghavi M et al (2021) Radiomics for the prediction of treatment outcome and survival in patients with colorectal cancer: a systematic review. Clin Colorectal Cancer 20:52–71. https://doi.org/10.1016/j.clcc.2020.11.001
Sun Q, Chen Y, Liang C et al (2021) Biologic pathways underlying prognostic radiomics phenotypes from paired MRI and RNA sequencing in glioblastoma. Radiology 301(3):203281. https://doi.org/10.1148/radiol.2021203281
Hinzpeter R, Baumann L, Guggenberger R et al (2021) Radiomics for detecting prostate cancer bone metastases invisible in CT: a proof-of-concept study. Eur Radiol. https://doi.org/10.1007/s00330-021-08245-6
He L, Liu Z, Liu C et al (2021) Radiomics based on lumbar spine magnetic resonance imaging to detect osteoporosis. Acad Radiol 28:e165–e171. https://doi.org/10.1016/j.acra.2020.03.046
Biamonte E, Levi R, Carrone F et al (2022) Artificial intelligence-based radiomics on computed tomography of lumbar spine in subjects with fragility vertebral fractures. J Endocrinol Invest 45:2007–2017. https://doi.org/10.1007/s40618-022-01837-z
Franke K, Gaser C (2019) Ten years of brainage as a neuroimaging biomarker of brain aging: what insights have we gained? Front Neurol 10:789. https://doi.org/10.3389/fneur.2019.00789
Han Y, Wang G (2020) Skeletal bone age prediction based on a deep residual network with spatial transformer. Comput Methods Programs Biomed 197:105754. https://doi.org/10.1016/j.cmpb.2020.105754
Zwanenburg A, Vallières M, Abdalah MA et al (2020) The image biomarker standardization initiative: standardized quantitative radiomics for high-throughput image-based phenotyping. Radiology 295:328–338. https://doi.org/10.1148/radiol.2020191145
Fedorov A, Beichel R, Kalpathy-Cramer J et al (2012) 3D Slicer as an image computing platform for the quantitative imaging network. Magn Reson Imaging 30:1323–1341. https://doi.org/10.1016/j.mri.2012.05.001
van Griethuysen JJM, Fedorov A, Parmar C et al (2017) Computational radiomics system to decode the radiographic phenotype. Cancer Res 77:e104–e107. https://doi.org/10.1158/0008-5472.CAN-17-0339
Fortin J-P, Cullen N, Sheline YI et al (2018) Harmonization of cortical thickness measurements across scanners and sites. Neuroimage 167:104–120. https://doi.org/10.1016/j.neuroimage.2017.11.024
Ozaki Y, Tanigaki Y, Watanabe S, Onishi M (2020) Multiobjective tree-structured parzen estimator for computationally expensive optimization problems. In: Proceedings of the 2020 Genetic and Evolutionary Computation Conference. ACM, New York, NY, USA, pp 533–541
Hsieh C-I, Zheng K, Lin C et al (2021) Automated bone mineral density prediction and fracture risk assessment using plain radiographs via deep learning. Nat Commun 12:5472. https://doi.org/10.1038/s41467-021-25779-x
Smets J, Shevroja E, Hügle T et al (2021) Machine Learning Solutions for Osteoporosis—A Review. J Bone Miner Res 36:833–851. https://doi.org/10.1002/jbmr.4292
Fang Y, Li W, Chen X et al (2021) Opportunistic osteoporosis screening in multi-detector CT images using deep convolutional neural networks. Eur Radiol 31:1831–1842. https://doi.org/10.1007/s00330-020-07312-8
Valentinitsch A, Trebeschi S, Kaesmacher J et al (2019) Opportunistic osteoporosis screening in multi-detector CT images via local classification of textures. Osteoporos Int 30:1275–1285. https://doi.org/10.1007/s00198-019-04910-1
Kawashima Y, Fujita A, Buch K et al (2019) Using texture analysis of head CT images to differentiate osteoporosis from normal bone density. Eur J Radiol 116:212–218. https://doi.org/10.1016/j.ejrad.2019.05.009
Sun W, Liu S, Guo J et al (2021) A CT-based radiomics nomogram for distinguishing between benign and malignant bone tumours. Cancer Imaging 21:20. https://doi.org/10.1186/s40644-021-00387-6
Dionísio FCF, Oliveira LS, Hernandes MA et al (2020) Manual and semiautomatic segmentation of bone sarcomas on MRI have high similarity. Brazilian J Med Biol Res. https://doi.org/10.1590/1414-431x20198962
Eweje FR, Bao B, Wu J et al (2021) Deep learning for classification of bone lesions on routine MRI. EBioMedicine 68:103402. https://doi.org/10.1016/j.ebiom.2021.103402
Geirhos R, Jacobsen J-H, Michaelis C et al (2020) Shortcut learning in deep neural networks. Nat Mach Intell 2:665–673. https://doi.org/10.1038/s42256-020-00257-z
DeGrave AJ, Janizek JD, Lee S-I (2021) AI for radiographic COVID-19 detection selects shortcuts over signal. Nat Mach Intell 3:610–619. https://doi.org/10.1038/s42256-021-00338-7
Berenguer R, del Pastor-Juan M, R, Canales-Vázquez J, et al (2018) Radiomics of CT features may be nonreproducible and redundant: influence of CT acquisition parameters. Radiology 288:407–415. https://doi.org/10.1148/radiol.2018172361
van Hamersvelt RW, Schilham AMR, Engelke K et al (2017) Accuracy of bone mineral density quantification using dual-layer spectral detector CT: a phantom study. Eur Radiol 27:4351–4359. https://doi.org/10.1007/s00330-017-4801-4
Euler A, Nowak T, Bucher B et al (2021) Assessment of bone mineral density from a computed tomography topogram of photon-counting detector computed tomography—effect of phantom size and tube voltage. Invest Radiol 56:614–620. https://doi.org/10.1097/RLI.0000000000000781
Niu YT, Olszewski ME, Zhang YX et al (2011) Experimental study and optimization of scan parameters that influence radiation dose in temporal bone high-resolution multidetector row CT. Am J Neuroradiol 32:1783–1788. https://doi.org/10.3174/ajnr.A2609
Topol EJ (2019) High-performance medicine: the convergence of human and artificial intelligence. Nat Med 25:44–56. https://doi.org/10.1038/s41591-018-0300-7
Bogowicz M, Jochems A, Deist TM et al (2020) Privacy-preserving distributed learning of radiomics to predict overall survival and HPV status in head and neck cancer. Sci Rep 10:4542. https://doi.org/10.1038/s41598-020-61297-4
Murray N, Le M, Ebrahimzadeh O et al (2017) Imaging the spine with dual-energy CT. Curr Radiol Rep 5:9. https://doi.org/10.1007/s40134-017-0236-6
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by RL, FG, MB, DR, MM, and GA. The first draft of the manuscript was written by RL, LSP and MG, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of IRCCS-Humanitas Research Hospital (March 22, 2022 / 2410).
Consent to participate
The collection of written informed consent was waived by Ethics Committee due to the retrospective nature of the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Levi, R., Garoli, F., Battaglia, M. et al. CT-based radiomics can identify physiological modifications of bone structure related to subjects’ age and sex. Radiol med 128, 744–754 (2023). https://doi.org/10.1007/s11547-023-01641-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-023-01641-6