Abstract
Background
Patients with a penicillin allergy label are at risk of an associated increase in adverse antibiotic events and hospitalization costs.
Aim
We aimed to study the economic savings derived from the correct diagnosis and delabeling inpatients with suspected beta-lactam allergy, considering the acquisition cost of antimicrobials prescribed during a patient's hospital stay.
Method
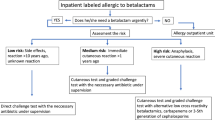
We prospectively evaluated patients admitted to the University Hospital of Salamanca who had been labeled as allergic to beta-lactams and performed a delabeling study. Subsequently, cost differences between antibiotics administered before and after the allergy study and those derived from those patients who received alternative antibiotics during admission and those who switched to beta-lactams after the allergy study were calculated.
Results
One hundred seventy-seven inpatients labeled as allergic to beta-lactams underwent a delabeling study; 34 (19.2%) were confirmed to have allergy to beta-lactams. Of the total number of patients, 136 (76.8%) received antibiotics during their hospitalization, involving a mean (SD) cost of €203.07 (318.42) and a median (IQR) cost of €88.97 (48.86–233.56). After delabeling in 85 (62.5%) patients, the antibiotic treatment was changed to beta-lactams. In this group of patients, the mean cost (SD) decreased from €188.91 (351.09) before the change to 91.31 (136.07) afterward, and the median cost (IQR) decreased from €72.92 (45.82–211.99) to €19.24 (11.66–168). The reduction was significant compared to the median cost of patients whose treatment was not changed to beta-lactams (p<0.001).
Conclusion
Delabeling hospitalized patients represents a cost-saving measure for treating patients labeled as allergic to beta-lactams.




Similar content being viewed by others
References
Shenoy ES, Macy E, Rowe T, et al. Evaluation and management of penicillin allergy: a review. JAMA. 2019;321:188–99.
Trubiano JA, Adkinson NF, Phillips EJ. Penicillin allergy is not necessarily forever. JAMA. 2017;318:82–3.
Sacco KA, Bates A, Brigham TJ, et al. Clinical outcomes following inpatient penicillin allergy testing: a systematic review and meta-analysis. Allergy. 2017;72:1288–96.
Huang KG, Cluzet V, Hamilton K, et al. The impact of reported beta-lactam allergy in hospitalized patients with hematologic malignancies requiring antibiotics [Erratum in Clin Infect Dis 2018;67:1151]. Clin Infect Dis. 2018;67:27–33.
U.S. Centers for Disease Control and Prevention (CDC). Antibiotic Use in the United States, 2017: Progress and Opportunities; 2017. US Department of Health and Human Services. Accessed 22 June 2021. https://www.cdc.gov/antibiotic-use/stewardship-report/pdf/stewardship-report.pdf.
Marston HD, Dixon DM, Knisely JM, et al. Antimicrobial resistance. JAMA. 2016;316:1193–204.
Mancini CM, Wimmer M, Schulz LT, et al. Association of penicillin or cephalosporin allergy documentation and antibiotic use in hospitalized patients with pneumonia. J Allergy Clin Immunol Pract. 2021;9:3060–8.
Nelson J, Carrillo-Martin I, Bosch W, et al. Outcomes in hospitalized kidney transplant patients with a penicillin allergy label in the United States, 2005–2014. J Allergy Clin Immunol Pract. 2022;10:867–9.
Blumenthal KG, Peter JG, Trubiano JA, et al. Antibiotic allergy. Lancet. 2019;393:183–98.
Blumenthal KG, Lu N, Zhang Y, et al. Risk of meticillin-resistant Staphylococcus aureus and Clostridium difficile in patients with a documented penicillin allergy: population based matched cohort study. BMJ. 2018;361: k2400.
Jeffres MN, Narayanan PP, Shuster JE, et al. Consequences of avoiding beta-lactams in patients with beta-lactam allergies. J Allergy Clin Immunol. 2016;137:1148–53.
Blumenthal KG, Lu N, Zhang Y, et al. Recorded penicillin allergy and risk of mortality: a population-based matched cohort study. J Gen Intern Med. 2019;34:1685–7.
Staicu ML, Vyles D, Shenoy ES, et al. Penicillin allergy delabeling: a multidisciplinary opportunity. J Allergy Clin Immunol Pract. 2020;8:2858–68.
Stone CA, Trubiano J, Coleman DT, et al. The challenge of delabeling penicillin allergy. Allergy. 2020;75:273–88.
Graham JK, Yang C, Vyles D, et al. Barriers to penicillin allergy delabeling in a pediatric emergency department. Ann Allergy Asthma Immunol. 2022;128:107–8.
McDanel D, Hosch L, Pham KD, et al. Relabeling of penicillin drug allergy after evaluation in a drug allergy clinic. J Allergy Clin Immunol Pract. 2022;10:346–8.
Romano A, Atanaskovic-Markovic M, Barbaud A, et al. Towards a more precise diagnosis of hypersensitivity to beta-lactams—an EAACI position paper. Allergy. 2020;75:1300–15.
Moreno E, Laffond E, Muñoz-Bellido F, et al. Performance in real life of the European Network on Drug Allergy algorithm in immediate reactions to beta-lactam antibiotics. Allergy. 2016;71:1787–90.
McDanel JS, Perencevich EN, Diekema DJ, et al. Comparative effectiveness of betalactams versus vancomycin for treatment of methicillin-susceptible Staphylococcus aureus bloodstream infections among 122 hospitals. Clin Infect Dis. 2015;61:361–7.
Picard M, Begin P, Bouchard H, et al. Treatment of patients with a history of penicillin allergy in a large tertiary-care academic Hospital. J Allergy Clin Immunol Pract. 2013;1:252–7.
Thong BY, Leong KP, Tang CY, et al. Drug allergy in a general hospital: Results of a novel prospective inpatient reporting system. Ann Allergy Asthma Immunol. 2003;90:342–7.
Fransson S, Mosbech H, Kappel M, et al. The importance of prolonged provocation in drug allergy—results from a Danish Allergy Clinic. J Allergy Clin Immunol Pract. 2017;5:1394–401.
Moreno EM, Moreno V, Laffond E, et al. Usefulness of an artificial neural network in the prediction of β-lactam allergy. J Allergy Clin Immunol Pract. 2020;8:2974–82.
Sobrino-García M, Muñoz-Bellido FJ, Moreno E, et al. A Comprehensive prospective study of the costs associated with evaluation of ß-lactam allergy. J Investig Allergol Clin Immunol. 2021;31:52–7.
Chiriac AM, Wang Y, Schrijvers R, et al. Designing predictive models for beta-lactam allergy using the drug allergy and hypersensitivity database. J Allergy Clin Immunol Pract. 2018;6:139–48.
Sousa-Pinto B, Blumenthal KG, Macy E, et al. Penicillin allergy testing is cost-saving: an economic evaluation study. Clin Infect Dis. 2021;72:924–38.
Koo G, Yu R, Phillips E, et al. Retrospective stratification of cephalosporin allergy label risk using validated penicillin allergy frameworks. J Allergy Clin Immunol Pract. 2022;10:2472–5.
Chen JR, Khan DA. Evaluation of penicillin allergy in the hospitalized patient: opportunities for antimicrobial stewardship. Curr Allergy Asthma Rep. 2017;17:40.
Álvarez-Cuesta E, Madrigal-Burgaleta R, Broyles AD, et al. Standards for practical intravenous rapid drug desensitization & delabeling: a WAO committee statement. World Allergy Organ J. 2022;15: 100640.
Schrüfer P, Stoevesandt J, Trautmann A. Outcome of a de-labelling algorithm compared with results of penicillin (β-lactam) allergy testing. Allergy Asthma Clin Immunol. 2022;18:26.
Brockow K, Wurpts G, Trautmann A. Patients with questionable penicillin (beta-lactam) allergy: causes and solutions. Allergol Select. 2022;6:33–41.
Anstey KM, Tsao L, Otani IM. Drug allergy delabeling programs: recent strategies and targeted populations. Clin Rev Allergy Immunol. 2022;62:484–504.
Khan DA. Proactive management of penicillin and other antibiotic allergies. Allergy Asthma Proc. 2020;41:82–9.
Berger RE, Singh HK, Loo AS, et al. Improving antibiotic stewardship for inpatients with reported beta-lactam allergies and limited access to penicillin skin testing. Jt Comm J Qual Patient Saf. 2022;48:147–53.
Macy E, Contreras R. Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: a cohort study. J Allergy Clin Immunol. 2014;133:790–6.
Sobrino M, Muñoz-Bellido FJ, Macías E, et al. A prospective study of costs associated with the evaluation of β-lactam allergy in children. J Pediatr. 2020;223:108–13.
Acknowledgements
We would like to thank the nursing staff of the Allergy Service of the University Hospital of Salamanca for their work in carrying out the allergic study and the i2e3 Biomedical Research Institute for providing medical writing assistance on behalf of Allergy Therapeutics (BEC-AT).
Funding
This study has been funded by the project RICORS-REI RD21/0002/0054, funded by Instituto de Salud Carlos III (ISCIII) and European Union -NextGeneration EU. Miriam Sobrino-García declares the following financial interests/personal relationships which may be considered as potential competing interests: “Juan Rodés” Clinical Research Contract (JR22/00050) for this research project, financed by the Carlos III Health Institute, and writing assistance provided by i2e3 Biomedical Research Institute, financed by Allergy Therapeutics Ibérica®.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Ignacio Dávila, In the last three years, has received payment for lectures, including service on speaker’s bureaus from Allergy Therapeutics, Astra-Zeneca, Chiesi, Diater, GSK, Leti, Novartis, Sanofi; for a consultancy from Allergy Therapeutics, ALK-Abello, Astra-Zeneca, GSK, Merck, MSD, Novartis, Sanofi; and grants for Thermofisher Diagnostics, ISCIII and Junta de Castilla y León. Francisco J. Muñoz-Bellido, Esther Moreno-Rodilla, Rita Martín-Muñoz and Aránzazu García-Iglesias have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sobrino-García, M., Muñoz-Bellido, F.J., Moreno-Rodilla, E. et al. Delabeling of allergy to beta-lactam antibiotics in hospitalized patients: a prospective study evaluating cost savings. Int J Clin Pharm (2024). https://doi.org/10.1007/s11096-024-01737-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11096-024-01737-7