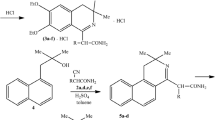
Series of 3,3-dimethyl- and 3,3-diethyl-substituted 2-R-(3,4-dihydroisoquinolin-1-yl)acetamides were synthesized via Ritter reactions of 1-(3,4-dimethoxyphenyl)-2-methylpropanol-2 and 1-phenyl-2-ethylbutanol-2 with 2-R-cyanoacetamides (R = Et, n-Pr, i-Pr, n-Bu, n-Am, and i-Am). The hydrochlorides of the obtained compounds were tested for anti-arrhythmic activity using the CaCl2 model. Three of ten compounds showed anti-arrhythmic activity with a maximum anti-arrhythmic index (AI) of 3.9.With respect to blood clotting, all compounds proved to be hemostatics. The most active compound increased the blood coagulation index up to 37.9%. Eight compounds exceeded pyrantel and two compounds exceeded levamisole with respect to anthelmintic activity. Larvicidal (insecticidal) effects were observed for nine compounds, two of which reduced the larvae life span to 10.6 and 10.4 min, thus exceeding the effect of diazinon (17.0 min).
Similar content being viewed by others
References
E. A. Markaryan, Zh. S. Arustamyan, S. V. Avetisyan, et al., Khim.-farm. Zh., 34(12), 5 – 7 (2000); Pharm. Chem. J., 34(12), 632 – 634 (2000).
S. V. Avetisyan, E. A. Markaryan, Zh. S. Arustamyan, and R. E. Markaryan, Khim.-farm. Zh., 40(7), 16 – 17 (2006); Pharm. Chem. J., 40(7), 360 – 362 (2006).
R. Z. Dautova, V. S. Shklyaev, B. Ya. Syropyatov, and A. G. Mikhailovskii, Khim.-farm. Zh., 23(2), 172 – 176 (1989); Pharm. Chem. J., 23(2), 133 – 136 (1989).
V. G. Kartsev and G. A. Tolstikov (eds.), Nitrogen-containing Heterocycles and Alkaloids, Iridium-Press, Moscow (2001).
B. Ya. Syropyatov, A. A. Gorbunov, V. S. Shklyaev, et al., Khim.-farm. Zh., 30(11), 13 – 14 (1996); Pharm. Chem. J., 30(11), 675 – 677 (1996).
A. G. Mikhailovskii, O. V. Gashkova, I. P. Rudakova, and D. A. Peretyagin, Khim.-farm. Zh., 51(7), 25 – 27 (2017); Pharm. Chem. J., 51(7), 546 – 549 (2017).
A. G. Mikhailovskii, E. S. Likhtenshtein, I. P. Rudakova, et al., Khim.-farm. Zh., 55(7), 8 – 13 (2021); Pharm. Chem. J., 55(7), 638 – 643 (2021).
D. A. Peretyagin, A. G. Mikhailovskii, I. P. Rudakova, et al., Khim.-farm. Zh., 55(12), 33 – 37 (2021); Pharm. Chem. J., 55(12), 1299 – 1303 (2022).
V. A. Glushkov, K. A. Arapov, O. V. Minova, et al., Khim.-farm. Zh., 40(7), 18 – 20 (2006); Pharm. Chem. J., 40(7), 363 – 366 (2006).
A. N. Mironov and N. D. Bunyatyan, Handbook for Preclinical Drug Studies. Part 1 [in Russian], Grif i K, Moscow (2012), 944 pp.
I. A. Arkhipov, Anthelmintics: Pharmacology and Use [in Russian], Meditsina, Moscow (2009).
M. M. Ganiev and V. D. Nedorezkov, Chemical Agents for Plant Protection [in Russian], Lan’, St. Petersburg (2013).
O. Yu. Eremina and Yu. V. Lopatina, Desinfekts. Delo, No. 2, 46 – 50 (2004).
V. V. Prozorovskii, M. P. Prozorovskaya, and V. M. Demchenko, Farmakol. Toksikol., 4, 497 – 502 (1978).
V. V. Gorbunova and N. P. Gorbunov, Farmakol. Toksikol., 3, 48 – 50 (1983).
M. P. Nikolaev, Experimental Bases of Pharmacology and Toxicology [in Russian], MEDGIZ, Moscow (1941).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 56, No. 6, pp. 23 – 28, June, 2022.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Peretyagin, D.A., Mikhailovskii, A.G., Rudakova, I.P. et al. Synthesis of 3,3-Dimethyl- and 3,3-Diethyl-Substituted 2-(3,4-Dihydroisoquinolin-1-yl)Carboxylic Acid Amide Hydrochlorides and Their Anti-Arrhythmic, Hemostatic, Anthelmintic, and Larvicidal Activity. Pharm Chem J 56, 748–753 (2022). https://doi.org/10.1007/s11094-022-02705-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-022-02705-z