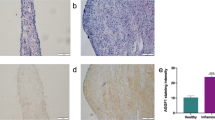
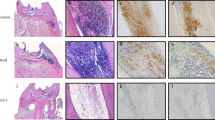
Abstract
Leukemia inhibitory factor (LIF) has been recognized as a novel inflammatory modulator in inflammation-associated diseases. This study aimed to investigate the modulation of LIF in dental pulp inflammation. Experimental pulpitis was established in wild-type (WT) and Lif-deficient (Lif−/−) mice. Histological and immunostaining analyses were conducted to assess the role of LIF in the progression of pulpitis. Mouse macrophage cell line (RAW264.7) was treated with LPS to simulate an inflammatory environment. Exogenous LIF was added to this system to examine its modulation in macrophage inflammatory response in vitro. Primary bone marrow-derived macrophages (BMDMs) from WT and Lif−/− mice were isolated and stimulated with LPS to confirm the effect of Lif deletion on macrophage inflammatory response. Supernatants from LIF and LPS-treated human dental pulp cells (hDPCs) were collected and added to macrophages. Macrophage chemotaxis was assessed using transwell assays. The results showed an increased expression of LIF and LIFR with the progression of pulpitis, and LIFR was highly expressed in macrophages. Lif deficiency alleviated experimental pulpitis with the reduction of pro-inflammatory cytokines and macrophage infiltration. Exogenous LIF promoted inflammatory response of LPS-induced macrophages through a STAT3/p65-dependent pathway. Consistently, Lif deletion inhibited macrophage inflammatory response in vitro. Supernatants of LIF-treated hDPCs enhanced macrophage migration in LPS-induced inflammatory environment. Our findings demonstrated that LIF aggravates pulpitis by promoting macrophage inflammatory response through a STAT3/p65-dependent pathway. Furthermore, LIF plays a crucial role in driving the recruitment of macrophages to inflamed pulp tissue by promoting chemokine secretion in DPCs.







Similar content being viewed by others
Availability of Data and Materials
The experimental data used to support this study can be provided upon request.
References
Van Hassel, H.J. 2021. Reprint of: Physiology of the human dental pulp. Journal of Endodontics 47 (5): 690–695. https://doi.org/10.1016/j.joen.2021.03.001.
Dal-Fabbro, R., W.B. Swanson, L.C. Capalbo, H. Sasaki, and M.C. Bottino. 2023. Next-generation biomaterials for dental pulp tissue immunomodulation. Dental Materials 39 (4): 333–349. https://doi.org/10.1016/j.dental.2023.03.013.
Nicola, N.A., and J.J. Babon. 2015. Leukemia inhibitory factor (LIF). Cytokine & Growth Factor Reviews 26 (5): 533–544. https://doi.org/10.1016/j.cytogfr.2015.07.001.
Poulton, I.J., N.E. McGregor, S. Pompolo, E.C. Walker, and N.A. Sims. 2012. Contrasting roles of leukemia inhibitory factor in murine bone development and remodeling involve region-specific changes in vascularization. Journal of Bone and Mineral Research 27 (3): 586–595. https://doi.org/10.1002/jbmr.1485.
Fitzgerald, J.S., T.G. Poehlmann, E. Schleussner, and U.R. Markert. 2008. Trophoblast invasion: The role of intracellular cytokine signalling via signal transducer and activator of transcription 3 (STAT3). Human Reproduction Update 14 (4): 335–344. https://doi.org/10.1093/humupd/dmn010.
Sendtner, M., R. Götz, B. Holtmann, J.L. Escary, Y. Masu, P. Carroll, et al. 1996. Cryptic physiological trophic support of motoneurons by LIF revealed by double gene targeting of CNTF and LIF. Current Biology 6 (6): 686–694. https://doi.org/10.1016/s0960-9822(09)00450-3.
Duluc, D., Y. Delneste, F. Tan, M.-P. Moles, L. Grimaud, J. Lenoir, et al. 2007. Tumor-associated leukemia inhibitory factor and IL-6 skew monocyte differentiation into tumor-associated macrophage-like cells. Blood 110 (13): 4319–4330. https://doi.org/10.1182/blood-2007-02-072587.
Nguyen, H.N., E.H. Noss, F. Mizoguchi, C. Huppertz, K.S. Wei, G.F.M. Watts, et al. 2017. Autocrine loop involving IL-6 family member LIF, LIF receptor, and STAT4 drives sustained fibroblast production of inflammatory mediators. Immunity 46 (2): 220–232. https://doi.org/10.1016/j.immuni.2017.01.004.
Sugiura, S., R. Lahav, J. Han, S.Y. Kou, L.R. Banner, F. de Pablo, et al. 2000. Leukaemia inhibitory factor is required for normal inflammatory responses to injury in the peripheral and central nervous systems in vivo and is chemotactic for macrophages in vitro. European Journal of Neuroscience 12 (2): 457–466. https://doi.org/10.1046/j.1460-9568.2000.00922.x.
Zhang, Y.S., D.E. Xin, Z. Wang, X. Song, Y. Sun, Q.C. Zou, et al. 2019. STAT4 activation by leukemia inhibitory factor confers a therapeutic effect on intestinal inflammation. EMBO Journal 38 (6):e99595. https://doi.org/10.15252/embj.201899595.
Dallagi, A., J. Girouard, J. Hamelin-Morrissette, R. Dadzie, L. Laurent, C. Vaillancourt, et al. 2015. The activating effect of IFN-γ on monocytes/macrophages is regulated by the LIF-trophoblast-IL-10 axis via Stat1 inhibition and Stat3 activation. Cellular & Molecular Immunology 12 (3): 326–341. https://doi.org/10.1038/cmi.2014.50.
Zhu, M., K. Oishi, S.C. Lee, and P.H. Patterson. 2001. Studies using leukemia inhibitory factor (LIF) knockout mice and a LIF adenoviral vector demonstrate a key anti-inflammatory role for this cytokine in cutaneous inflammation. Journal of Immunology 166 (3): 2049–2054. https://doi.org/10.4049/jimmunol.166.3.2049.
Davis, S.M., L.A. Collier, E.D. Winford, C.C. Leonardo, C.T. Ajmo, E.A. Foran, et al. 2018. Leukemia inhibitory factor modulates the peripheral immune response in a rat model of emergent large vessel occlusion. Journal of Neuroinflammation 15 (1): 288. https://doi.org/10.1186/s12974-018-1326-y.
Hendriks, J.J., H. Slaets, S. Carmans, H.E. de Vries, C.D. Dijkstra, P. Stinissen, et al. 2008. Leukemia inhibitory factor modulates production of inflammatory mediators and myelin phagocytosis by macrophages. Journal of Neuroimmunology 204 (1–2): 52–57. https://doi.org/10.1016/j.jneuroim.2008.07.015.
Bruno, K.F., J.A. Silva, T.A. Silva, A.C. Batista, A.H.G. Alencar, and C. Estrela. 2010. Characterization of inflammatory cell infiltrate in human dental pulpitis. International Endodontic Journal 43 (11): 1013–1021. https://doi.org/10.1111/j.1365-2591.2010.01757.x.
Le Fournis, C., C. Jeanneau, T. Giraud, I. El Karim, F.T. Lundy, and I. About. 2021. Fibroblasts control macrophage differentiation during pulp inflammation. Journal of Endodontics 47 (9): 1427–1434. https://doi.org/10.1016/j.joen.2021.06.015.
Zaky, S.H., M. Shehabeldin, H. Ray, C. Sfeir, 2021. The role of inflammation modulation in dental pulp regeneration. European Cells & Materials 4 : 184–193. https://doi.org/10.22203/eCM.v041a13.
He, Y., Y. Gan, J. Lu, Q. Feng, H. Wang, H. Guan, et al. 2017. Pulpal tissue inflammatory reactions after experimental pulpal exposure in mice. Journal of Endodontics 43 (1): 90–95. https://doi.org/10.1016/j.joen.2016.09.003.
Ye, L., L. Peng, H. Tan, and X. Zhou. 2006. HGF enhanced proliferation and differentiation of dental pulp cells. Journal of Endodontics 32 (8): 736–741. https://doi.org/10.1016/j.joen.2006.01.007.
Fan, Y., R. Mao, and J. Yang. 2013. NF-κB and STAT3 signaling pathways collaboratively link inflammation to cancer. Protein & Cell 4 (3): 176–185. https://doi.org/10.1007/s13238-013-2084-3.
Grivennikov, S.I., and M. Karin. 2010. Dangerous liaisons: STAT3 and NF-kappaB collaboration and crosstalk in cancer. Cytokine & Growth Factor Reviews 21 (1): 11–19. https://doi.org/10.1016/j.cytogfr.2009.11.005.
Franklin, R.A. 2021. Fibroblasts and macrophages: Collaborators in tissue homeostasis. Immunological Reviews 302 (1): 86–103. https://doi.org/10.1111/imr.12989.
Kerr, B.J., and P.H. Patterson. 2004. Potent pro-inflammatory actions of leukemia inhibitory factor in the spinal cord of the adult mouse. Experimental Neurology 188 (2): 391–407. https://doi.org/10.1016/j.expneurol.2004.04.012.
Upadhyay, A., D. Senyschyn, L. Santos, R. Gu, G.J. Carroll, and J.A. Jazayeri. 2012. K/B×N serum transfer arthritis is delayed and less severe in leukaemia inhibitory factor (LIF)-deficient mice. Clinical and Experimental Immunology 169 (2): 71–78. https://doi.org/10.1111/j.1365-2249.2012.04601.x.
Bell, M.C., and G.J. Carroll. 2000. Rheumatoid synovial fluid contains bioactive leukemia inhibitory factor with cartilage degrading activity–Another target for chondroprotective intervention. Journal of Rheumatology 27 (2): 332–338 (PMID:10685793).
Weber, M.A., S. Schnyder-Candrian, B. Schnyder, V. Quesniaux, V. Poli, C.L. Stewart, et al. 2005. Endogenous leukemia inhibitory factor attenuates endotoxin response. Laboratory Investigation 85 (2): 276–284. https://doi.org/10.1038/labinvest.3700216.
Nowell, M.A., A.S. Williams, S.A. Carty, J. Scheller, A.J. Hayes, G.W. Jones, et al. 2009. Therapeutic targeting of IL-6 trans signaling counteracts STAT3 control of experimental inflammatory arthritis. Journal of Immunology 182 (1): 613–622. https://doi.org/10.4049/jimmunol.182.1.613.
Atreya, R., J. Mudter, S. Finotto, J. Müllberg, T. Jostock, S. Wirtz, et al. 2000. Blockade of interleukin 6 trans signaling suppresses T-cell resistance against apoptosis in chronic intestinal inflammation: Evidence in crohn disease and experimental colitis in vivo. Nature Medicine 6 (5): 583–588. https://doi.org/10.1038/75068.
Xing, Z., J. Gauldie, G. Cox, H. Baumann, M. Jordana, X.F. Lei, et al. 1998. IL-6 is an antiinflammatory cytokine required for controlling local or systemic acute inflammatory responses. Journal of Clinical Investigation 101 (2): 311–320. https://doi.org/10.1172/JCI1368.
Yasukawa, H., M. Ohishi, H. Mori, M. Murakami, T. Chinen, D. Aki, et al. 2003. IL-6 induces an anti-inflammatory response in the absence of SOCS3 in macrophages. Nature Immunology 4 (6): 551–556. https://doi.org/10.1038/ni938.
Garbers, C., H.M. Hermanns, F. Schaper, G. Müller-Newen, J. Grötzinger, S. Rose-John, et al. 2012. Plasticity and cross-talk of interleukin 6-type cytokines. Cytokine & Growth Factor Reviews 23 (3): 85–97. https://doi.org/10.1016/j.cytogfr.2012.04.001.
Greenhill, C.J., S. Rose-John, R. Lissilaa, W. Ferlin, M. Ernst, P.J. Hertzog, et al. 2011. IL-6 trans-signaling modulates TLR4-dependent inflammatory responses via STAT3. Journal of Immunology 186 (2): 1199–1208. https://doi.org/10.4049/jimmunol.1002971.
Lee, H., A. Herrmann, J.H. Deng, M. Kujawski, G. Niu, Z. Li, et al. 2009. Persistently activated Stat3 maintains constitutive NF-kappaB activity in tumors. Cancer Cell 15 (4): 283–293. https://doi.org/10.1016/j.ccr.2009.02.015.
Yang, J., X. Liao, M.K. Agarwal, L. Barnes, P.E. Auron, and G.R. Stark. 2007. Unphosphorylated STAT3 accumulates in response to IL-6 and activates transcription by binding to NFkappaB. Genes & Development 21 (11): 1396–1408. https://doi.org/10.1101/gad.1553707.
Tofaris, G.K., P.H. Patterson, K.R. Jessen, and R. Mirsky. 2002. Denervated Schwann cells attract macrophages by secretion of leukemia inhibitory factor (LIF) and monocyte chemoattractant protein-1 in a process regulated by interleukin-6 and LIF. Journal of Neuroscience 22 (15): 6696–6703. https://doi.org/10.1523/JNEUROSCI.22-15-06696.2002.
Acknowledgements
Thanks to the Key Laboratory of Oral Biomedicine Ministry of Education and Hubei Key Laboratory of Stomatology for providing us with the laboratory.
Funding
This study was financially supported by the National Natural Science Foundation of China (Nos. 82071090 and 82001014), the Innovation and Development Fund of Hubei Province Natural Science Foundation (No. 2022CFD005), Hubei Provincial Natural Science Foundation (No. 2022CFB115), and Shenzhen Scientific Research Project (No. JCYJ20180302144621755).
Author information
Authors and Affiliations
Contributions
Donghua Guo contributed to conceptualization, methodology, writing—original draft, visualization, and data curation. Wei Dong participated in methodology, validation, and discussion. Jiali Zhang and Yi Zhou participated in writing—review and editing, supervision, and discussion. Yaqi Cong, Yi Liu, Youde Liang, and Zhou Ye participated in the implementation of some experiments and discussion. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
All animal experiment was approved by the Institutional Animal Care and Use Committee of Wuhan University (approval no: S07923020D).
Consent for Publication
All the authors have read the manuscript and agreed to submit the paper to the journal.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, D., Dong, W., Cong, Y. et al. LIF Aggravates Pulpitis by Promoting Inflammatory Response in Macrophages. Inflammation 47, 307–322 (2024). https://doi.org/10.1007/s10753-023-01910-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-023-01910-6