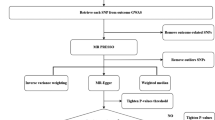
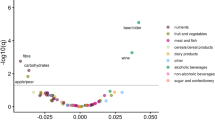
Abstract
Dietary factors have been extensively investigated as possible risk factors for liver cancer, but the evidence is inconclusive. Our study systematically assessed the association between 142 foods and nutrients and liver cancer risk in a Chinese population using a diet-wide association study. Based on data from 59,844 men in the Shanghai Men’s Health Study (SMHS), we assessed the diet intake by dietary questionnaires. Cox regression was used to quantify the association between each food and nutrient and liver cancer risk. A false discovery rate (FDR) of 0.05 was used to select the foods and nutrients for validation. In the cohort, 431 liver cancer cases were identified during 712,373 person-years of follow-up. Retinol (HR per 1 SD increment = 1.09, 95% CI: 1.03–1.14) was associated with a higher risk of liver cancer, whereas onions (HR per 1 SD increment = 0.67, 95% CI: 0.54–0.84) and manganese (HR per 1 SD increment = 0.85, 95% CI: 0.78–0.94) were inversely associated with liver cancer risk. In the replication analysis, estimates for these foods and nutrients were similar in magnitude and direction. Our findings confirm that retinol, onions and manganese were associated with liver cancer risk, which provides reliable evidence between diet and liver cancer development.


Similar content being viewed by others
Data availability
The data will be available on request pending approval by the scientific committee of the relevant institutes.
Abbreviations
- PLC:
-
Primary liver cancer
- HCC:
-
Hepatocellular carcinoma
- HBV:
-
Hepatitis B virus
- HCV:
-
Hepatitis C virus
- T2DM:
-
Type 2 diabetes mellitus
- MAFLD:
-
Metabolic associated fatty liver disease
- WCRF:
-
World Cancer Research Fund
- HPFS:
-
Health Professionals Follow-up Study
- NHS:
-
Nurses’ Health Study
- EPIC:
-
European Prospective Investigation into Cancer and Nutrition
- SMHS:
-
Shanghai Men’s Health Study
- DWAS:
-
Diet-wide association study
- GWAS:
-
Genome-wide association study
- FDR:
-
False discovery rate
- FFQ:
-
Food frequency questionnaire
- HR:
-
Hazard ratio
- CI:
-
Confidence interval
- PYs:
-
Person-years
- BMI:
-
Body mass index
- MET:
-
Metabolic equivalent
- IQR:
-
Inter quartile range
References
Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49. https://doi.org/10.3322/caac.21660.
Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66(2):115–32. https://doi.org/10.3322/caac.21338.
Martel C, Maucort-Boulch D, Plummer M, et al. World-wide relative contribution of Hepatitis B and C viruses in hepatocellular carcinoma. Hepatology. 2015;62(4):1190–200. https://doi.org/10.1002/hep.27969.
Chu YJ, Yang HI, Wu HC, et al. Aflatoxin B(1) exposure increases the risk of hepatocellular carcinoma associated with Hepatitis C virus Infection or alcohol consumption. Eur J Cancer. 2018;94:37–46. https://doi.org/10.1016/j.ejca.2018.02.010.
Saracco G. Primary Liver cancer is of multifactorial origin: importance of Hepatitis B virus Infection and dietary aflatoxin. J Gastroenterol Hepatol. 1995;10(5):604–8. https://doi.org/10.1111/j.1440-1746.1995.tb01354.x.
Fan JG, Farrell GC. Prevention of hepatocellular carcinoma in nonviral-related Liver Diseases. J Gastroenterol Hepatol. 2009;24(5):712–9. https://doi.org/10.1111/j.1440-1746.2009.05776.x.
Bosetti C, Turati F, Vecchia CL. Hepatocellular carcinoma epidemiology. Best Pract Res Clin Gastroenterol. 2014; 28(2014): 753 – 70. https://doi.org/10.1016/j.bpg.2014.08.007.
Yang WS, Zeng XF, Liu ZN, et al. Diet and Liver cancer risk: a narrative review of epidemiological evidence. Br J Nutr. 2020;124(3):330–40. https://doi.org/10.1017/s0007114520001208.
George ES, Sood S, Broughton A, et al. The association between diet and hepatocellular carcinoma: a systematic review. Nutrients. 2021;13(1):172. https://doi.org/10.3390/nu13010172.
World Cancer Research Fund, American Institute for Cancer Research. Diet, nutrition, physical activity, and cancer: a global perspective-The third report; 2018.
Ma Y, Yang W, Li T, et al. Meat intake and risk of hepatocellular carcinoma in two large US prospective cohorts of women and men. Int J Epidemiol. 2019;48(6):1863–71. https://doi.org/10.1093/ije/dyz146.
Duarte-Salles T, Fedirko V, Stepien M, et al. Dietary fat, fat subtypes and hepatocellular carcinoma in a large European cohort. Int J Cancer. 2015;137(11):2715–28. https://doi.org/10.1002/ijc.29643.
Ji XW, Wang J, Shen QM, et al. Dietary fat intake and Liver cancer incidence: a population-based cohort study in Chinese men. Int J Cancer. 2021;148(12):2982–96. https://doi.org/10.1002/ijc.33507.
Papadimitriou N, Bouras E, van den Brandt PA, et al. A prospective diet-wide association study for risk of Colorectal cancer in EPIC. Clin Gastroenterol Hepatol. 2022;20(4):864–73e13. https://doi.org/10.1016/j.cgh.2021.04.028.
Heath AK, Muller DC, van den Brandt PA, et al. Nutrient-wide association study of 92 foods and nutrients and Breast cancer risk. Breast Cancer Res. 2020;22(1):5. https://doi.org/10.1186/s13058-019-1244-7.
Heath AK, Muller DC, van den Brandt PA, et al. Diet-wide association study of 92 foods and nutrients and Lung cancer risk in the European prospective investigation into Cancer and Nutrition study and the Netherlands Cohort Study. Int J Cancer. 2022;151(11):1935–46. https://doi.org/10.1002/ijc.34211.
Shu XO, Li H, Yang G, et al. Cohort profile: the Shanghai men’s Health Study. Int J Epidemiol. 2015;44(3):810–8. https://doi.org/10.1093/ije/dyv013.
Villegas R, Yang G, Liu D, et al. Validity and reproducibility of the food-frequency questionnaire used in the Shanghai men’s health study. Br J Nutr. 2007;97(5):993–1000. https://doi.org/10.1017/s0007114507669189.
Yang YX, Wang GY, Pan XC. Chinese food composition tables. 2rd ed. Peking University Medical Press; 2002.
Rao C, Yang G, Hu J, et al. Validation of cause-of-death statistics in urban China. Int J Epidemiol. 2007;36(3):642–51. https://doi.org/10.1093/ije/dym003.
World Health Organization. International classification of diseases. 9th ed. Basic tabulation list with alphabetical index; 1978.
Prentice RL, Zhao S. Regression models and multivariate life tables. J Am Stat Assoc. 2021;116(535):1330–45. https://doi.org/10.1080/01621459.2020.1713792.
Willett WC, Howe GR, Kushi LH. Adjustment for total energy intake in epidemiologic studies. Am J Clin Nutr. 1997;65(4):1220–28. https://doi.org/10.1093/ajcn/65.4.1220S.
Zhou BF. Predictive values of body mass index and waist circumference for risk factors of certain related Diseases in Chinese adults-study on optimal cut-off points of body mass index and waist circumference in Chinese adults. Biomed Environ Sci. 2002;15(1):83–96.
Korthauer K, Kimes PK, Duvallet C, et al. A practical guide to methods controlling false discoveries in computational biology. Genome Biol. 2019;20(1):118. https://doi.org/10.1186/s13059-019-1716-1.
Merritt MA, Tzoulaki I, Tworoger SS, et al. Investigation of dietary factors and endometrial cancer risk using a nutrient-wide association study approach in the EPIC and nurses’ health study (NHS) and NHSII. Cancer Epidemiol Biomarkers Prev. 2015;24(2):466–71. https://doi.org/10.1158/1055-9965.epi-14-0970.
Xiaofan H, Jing X, Chenni G, et al. New risk score for predicting progression of membranous nephropathy. J Transl Med. 2019;17(1):41. https://doi.org/10.1186/s12967-019-1792-8.
Yang Y, Gao J, Li HL, et al. Dose-response association between Hepatitis B surface antigen levels and Liver cancer risk in Chinese men and women. Int J Cancer. 2016;139(2):355–62. https://doi.org/10.1002/ijc.30086.
Russell RM. The vitamin a spectrum: from deficiency to toxicity. Am J Clin Nutr. 2000;71(4):878–84. https://doi.org/10.1093/ajcn/71.4.878.
Dawson MI. The importance of vitamin A in nutrition. Curr Pharm Des. 2000;6(3):311–25. https://doi.org/10.2174/1381612003401190.
Lan QY, Zhang YJ, Liao GC, et al. The association between dietary vitamin A and carotenes and the risk of primary Liver cancer: s case-control study. Nutrients. 2016;8(10):624. https://doi.org/10.3390/nu8100624.
Yang Y, Zhang D, Feng N, et al. Increased intake of vegetables, but not fruit, reduces risk for hepatocellular carcinoma: a meta-analysis. Gastroenterology. 2014;147(5):1031–42. https://doi.org/10.1053/j.gastro.2014.08.005.
Bamia C, Lagiou P, Jenab M, et al. Fruit and vegetable consumption in relation to hepatocellular carcinoma in a multi-centre, European cohort study. Br J Cancer. 2015;112(7):1273–82. https://doi.org/10.1038/bjc.2014.654.
Liu X, Yang W, Petrick JL, et al. Higher intake of whole grains and dietary fiber are associated with lower risk of Liver cancer and chronic Liver Disease mortality. Nat Commun. 2021;12(1):6388. https://doi.org/10.1038/s41467-021-26448-9.
Yang W, Ma Y, Liu Y, et al. Association of intake of whole grains and dietary fiber with risk of hepatocellular carcinoma in US adults. JAMA Oncol. 2019;5(6):879–86. https://doi.org/10.1001/jamaoncol.2018.7159.
Knowles LM, Milner JA. Diallyl disulfide inhibits p34(cdc2) kinase activity through changes in complex formation and phosphorylation. Carcinogenesis. 2000;21(6):1129-34.
Nicastro HL, Ross SA, Milner JA. Garlic and onions: their cancer prevention properties. Cancer Prev Res (Phila). 2015;8(3):181–9. https://doi.org/10.1158/1940-6207.capr-14-0172.
Rizk M, Guilloteau A, Mouillot T, et al. Dietary components modulate the risk of hepatocellular carcinoma in cirrhotic patients. Nutr Res. 2019;61:82–94. https://doi.org/10.1016/j.nutres.2018.10.002.
Ma X, Yang Y, Li HL, et al. Dietary trace element intake and Liver cancer risk: results from two population-based cohorts in China. Int J Cancer. 2017;140(5):1050–59. https://doi.org/10.1002/ijc.30522.
Van Remmen H, Ikeno Y, Hamilton M, et al. Life-long reduction in MnSOD activity results in increased DNA damage and higher incidence of cancer but does not accelerate aging. Physiol Genomics. 2003;16(1):29–37. https://doi.org/10.1152/physiolgenomics.00122.2003.
Chen CS, Zhao Q, Wang J, et al. Enhanced anti-tumor effects achieved in a murine Tumor model using combination therapy of recombinant human manganese superoxide dismutase and adriamycin. Biochem Biophys Res Commun. 2008;370(4):663–8. https://doi.org/10.1016/j.bbrc.2008.04.022.
Skourlis N, Patsis I, Martimianaki G, et al. Changes in the dietary habits of the Greek EPIC cohort participants during a 14-year follow-up period (1997–2011). Nutrients. 2020;12(7):2148. https://doi.org/10.3390/nu12072148.
Cai H, Yang G, Xiang YB, et al. Sources of variation in nutrient intakes among men in Shanghai, China. Public Health Nutr. 2005;8(8):1293–9. https://doi.org/10.1079/phn2005748.
Yang W, Sui J, Ma Y, et al. High dietary intake of vegetable or polyunsaturated fats is associated with reduced risk of hepatocellular carcinoma. Clin Gastroenterol Hepatol. 2020;18(12):2775–83e11. https://doi.org/10.1016/j.cgh.2020.01.003.
Antwi SO, Petrick JL, Campbell PT, et al. One-carbon metabolism-related micronutrients intake and risk for hepatocellular carcinoma: a prospective cohort study. Int J Cancer. 2020;147(8):2075–90. https://doi.org/10.1002/ijc.33007.
He TT, Xiao HW, Wusiman M, et al. Dietary intake of one-carbon metabolism-related nutrients and hepatocellular carcinoma survival in the Guangdong Liver Cancer Cohort. Food Funct. 2022;13(15):8081–90. https://doi.org/10.1039/d2fo00943a.
Fedirko V, Lukanova A, Bamia C, et al. Glycemic index, glycemic load, dietary carbohydrate, and dietary fiber intake and risk of liver and biliary tract cancers in western europeans. Ann Oncol. 2013;24(2):543–53. https://doi.org/10.1093/annonc/mds434.
Acknowledgements
We thank all participants and staff from the Shanghai Men’s Health Study for their contribution to this research.
Funding
This work was supported by a grant from the National Key Project of Research and Development Program of China [2021YFC2500404 & 2021YFC2500405]; and the parent cohort was supported by a subcontract of grant from the US National Institutes of Health [UM1 CA182910]. All funders had no role in the design, analysis and writing of this article.
Author information
Authors and Affiliations
Contributions
Y-BX designed research and obtained funding; J-YT, Z-YL, Q-MS, Y-TT, H-LL, Y-BX conducted the study; J-YT, Y-BX analyzed the data and interpreted the results; J-YT and Y-BX prepared and wrote the first draft; All authors reviewed and approved the final version of the paper; and Y-BX has primary responsibility for final content.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
The cohort protocol has been approved by the Institutional Review Boards of the Shanghai Cancer Institute and Vanderbilt University. Informed consent has been obtained from all participants. The current study has been approved by the Renji Hospital Ethics Committee of Shanghai Jiao Tong University School of Medicine (KY2019-197).
Consent for publication
The current manuscript does not contain any individual person’s data.
Conflict of interest
The authors have declared no potential conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tuo, JY., Li, ZY., Shen, QM. et al. A diet-wide association study for liver cancer risk: findings from a prospective cohort study in Chinese men. Eur J Epidemiol 39, 171–178 (2024). https://doi.org/10.1007/s10654-023-01071-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10654-023-01071-8