Abstract
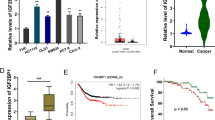
Increasing data and literature have illustrated that tumor immune escape represents a major source of tumor formation and recrudesce. Besides, novel findings also indicate that RNA N6-methyladenosine (m6A) participates in the human cancer immune escape. Here, our study investigated the functions of m6A reader YTHDF1 in prostate cancer (PCa) immune response and explored the functional mechanism. Results reported that YTHDF1 up-regulated in PCa samples and was closely correlated to poor clinical prognosis. Functionally, YTHDF1 inhibited the killing activity of CD8 + T cells to PCa cells, and moreover mitigated the ferroptosis. Mechanistically, PD-L1 acted as the target of YTHDF1, and YTHDF1 upregulated the transcriptional activity of PD-L1 mRNA. Collectively, YTHDF1 promoted functional PD-L1 partially through enhancing its transcriptional stability, which was necessary for PCa cells to evade effector T cell cytotoxicity and CD8 + T cells mediated ferroptosis. In conclusion, these findings indicate that YTHDF1 represses the CD8 + T cell-mediated antitumor immunity and ferroptosis in PCa via m6A-PD-L1 manner, which may provide novel insight for PCa immunotherapy.






Similar content being viewed by others
Data Availability
Not applicable.
References
Rizzo A, Santoni M, Mollica V, Fiorentino M, Brandi G, Massari F (2022) Microbiota and prostate cancer. Sem Cancer Biol 86:1058–1065
Sandhu S, Moore CM, Chiong E, Beltran H, Bristow RG, Williams SG (2021) Prostate cancer. Lancet (London England) 398:1075–1090
Sathianathen NJ, Konety BR, Crook J, Saad F, Lawrentschuk N (2018) Landmarks in prostate cancer. Nat Reviews Urol 15:627–642
Wasim S, Lee SY, Kim J (2022) Complexities of Prostate Cancer. International journal of molecular sciences 23
Zhao Y, Wen S, Li H et al (2023) Enhancer RNA promotes resistance to radiotherapy in bone-metastatic prostate cancer by m(6)a modification. Theranostics 13:596–610
Wang F, Wu L, Yin L, Shi H, Gu Y, Xing N (2022) Combined treatment with anti-PSMA CAR NK-92 cell and anti-PD-L1 monoclonal antibody enhances the antitumour efficacy against castration-resistant prostate cancer. Clin Translational Med 12:e901
He L, Li H, Wu A, Peng Y, Shu G, Yin G (2019) Functions of N6-methyladenosine and its role in cancer. Mol Cancer 18:176
Liu L, Li H, Hu D et al (2022) Insights into N6-methyladenosine and programmed cell death in cancer. Mol Cancer 21:32
Huang W, Chen TQ, Fang K, Zeng ZC, Ye H, Chen YQ (2021) N6-methyladenosine methyltransferases: functions, regulation, and clinical potential. J Hematol Oncol 14:117
Han J, Kong H, Wang X, Zhang XA (2022) Novel insights into the interaction between N6-methyladenosine methylation and noncoding RNAs in musculoskeletal disorders. Cell Prolif 55:e13294
Chen Y, Pan C, Wang X et al (2021) Silencing of METTL3 effectively hinders invasion and metastasis of prostate cancer cells. Theranostics 11:7640–7657
Yuan Y, Li H, Pu W et al (2022) Cancer metabolism and tumor microenvironment: fostering each other? Sci China Life Sci 65:236–279
Paskeh MDA, Entezari M, Mirzaei S et al (2022) Emerging role of exosomes in cancer progression and tumor microenvironment remodeling. J Hematol Oncol 15:83
Zhou X, Zou L, Liao H et al (2022) Abrogation of HnRNP L enhances anti-PD-1 therapy efficacy via diminishing PD-L1 and promoting CD8(+) T cell-mediated ferroptosis in castration-resistant prostate cancer. Acta Pharm Sinica B 12:692–707
Zhang Y, Zhu S, Du Y et al (2022) RelB upregulates PD-L1 and exacerbates prostate cancer immune evasion. J Experimental Clin cancer Research: CR 41:66
Zhu D, Xu R, Huang X et al (2021) Deubiquitinating enzyme OTUB1 promotes cancer cell immunosuppression via preventing ER-associated degradation of immune checkpoint protein PD-L1. Cell Death Differ 28:1773–1789
Zhang H, Hu Y, Liu D et al (2022) The histone demethylase Kdm6b regulates the maturation and cytotoxicity of TCRαβ(+)CD8αα(+) intestinal intraepithelial lymphocytes. Cell Death Differ 29:1349–1363
Liu C, Wang M, Zhang H et al (2022) Tumor microenvironment and immunotherapy of oral cancer. Eur J Med Res 27:198
Sherman MH, Beatty GL (2023) Tumor Microenvironment in Pancreatic Cancer Pathogenesis and Therapeutic Resistance. Annu Rev Pathol 18:123–148
Liu Y, Li C, Lu Y, Liu C, Yang W (2022) Tumor microenvironment-mediated immune tolerance in development and treatment of gastric cancer. Front Immunol 13:1016817
Du W, Zhu J, Zeng Y et al (2021) KPNB1-mediated nuclear translocation of PD-L1 promotes non-small cell lung cancer cell proliferation via the Gas6/MerTK signaling pathway. Cell Death Differ 28:1284–1300
Chang LS, Barroso-Sousa R, Tolaney SM, Hodi FS, Kaiser UB, Min L (2019) Endocrine toxicity of Cancer Immunotherapy Targeting Immune Checkpoints. Endocr Rev 40:17–65
Wen Y, Tang F, Tu C, Hornicek F, Duan Z, Min L (2022) Immune checkpoints in osteosarcoma: recent advances and therapeutic potential. Cancer Lett 547:215887
Jang HR, Shin SB, Kim CH et al (2021) PLK1/vimentin signaling facilitates immune escape by recruiting Smad2/3 to PD-L1 promoter in metastatic lung adenocarcinoma. Cell Death Differ 28:2745–2764
Zhou X, Ren T, Zan H, Hua C, Guo X (2022) Novel Immune Checkpoints in Esophageal Cancer: from biomarkers to therapeutic targets. Front Immunol 13:864202
Wang Y, Wang Y, Ren Y, Zhang Q, Yi P, Cheng C (2022) Metabolic modulation of immune checkpoints and novel therapeutic strategies in cancer. Sem Cancer Biol 86:542–565
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Yibing Wang, Peng Jin performed the assays and wrote the main manuscript and prepared Figs. 1, 2, 3, 4, 5 and 6. Xia Wang was responsible for the funding. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
All authors declare no conflicts of interest.
Ethics approval and consent to participate
Written informed consent was obtained from each patient, and the study had been approved by the Ethical Committee of Shengjing Hospital of China Medical University.
Consent for publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Jin, P. & Wang, X. N6-methyladenosine regulator YTHDF1 represses the CD8 + T cell-mediated antitumor immunity and ferroptosis in prostate cancer via m6A/PD-L1 manner. Apoptosis 29, 142–153 (2024). https://doi.org/10.1007/s10495-023-01885-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-023-01885-7