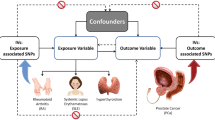
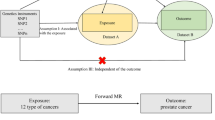
Abstract
It is unclear if the association between rheumatoid arthritis (RA) and a higher risk of prostate cancer (Pca) reflects a causal relationship. We conducted a meta-analysis and used the Mendelian randomization method (MR) to evaluate the association between RA and Pca risk. A meta-analysis and subgroup analysis of the incidence of Pca in patients with RA was conducted. To determine whether genetically elevated RA levels were causally linked to Pca, two MR samples were employed. To eliminate gender-related bias, we conducted a stratified analysis of the GWAS data for RA by gender, specifically including 140,254 males. Additional MR analysis was also performed to determine potential confounding factors influencing the association between genetically susceptible RA and Pca. In total, 409,950 participants were enrolled in 20 trials to investigate the Pca risk in patients with RA. The meta-analysis suggested that RA was unrelated to the Pca risk (SIR = 1.072, 95% CI, 0.883–1.261). However, a subgroup analysis showed that low smoking rates might increase the Pca risk in patients with RA by 24%. The MR analysis showed that increased genetic susceptibility to RA was related to a high Pca risk (OR = 36.20, 95%CI = 1.24–1053.12, P = 0.037). The causality estimation of MR-Egger, Weighted mode, Simple mode, and Weighted median method were similar in direction and magnitude. Although our meta-analysis found no correlation between RA and Pca risk, MR analyses supported a causal relationship between genetic susceptibility to RA and increased prostate risk. Early attention to Pca risk in patients with RA may be important for improving prognosis and mortality in such patients. Further research is needed to determine the etiology of RA attributed to Pca and its underlying mechanisms.





Similar content being viewed by others
Availability of data and materials
The data of RA in males can be obtained from the Wget command https://broad-ukb-sumstats-us-east-1.s3.amazonaws.com/round2/additive-tsvs/20002_1464.gwas.imputed_v3.male.tsv.bgz or AWS file https://broad-ukb-sumstats-us-east-1.s3.amazonaws.com/round2/additive-tsvs/20002_1464.gwas.imputed_v3.male.tsv.bgz. The datasets GWAS for this study can be found in the GWAS Catalog [https://www.ebi.ac.uk/gwas/]. GWAS ID details can be found in methods or Supplementary Material.
Abbreviations
- RA:
-
Rheumatoid arthritis
- Pca:
-
Prostate cancer
- MR:
-
Mendelian randomization
- GWASs:
-
Genome-wide association studies
- MOOSE:
-
Meta-analysis of observational studies in epidemiology
- MeSH:
-
Medical subject headings
- QATSO:
-
Quality assessment tool for systematic reviews of observational studies
- RCTs:
-
Randomized controlled trials
- ACR:
-
Criteria: American College of Rheumatology criteria
- ARA:
-
Criteria: American Rheumatism Association criteria
- SIR:
-
Standardized incidence rate
- OR:
-
Odds ratio
- HR:
-
Hazard ratio
- RR:
-
Relative risk
- CI:
-
Confidence interval
- SNP:
-
Single nucleotide polymorphism
- LD:
-
Linkage disequilibrium
- PRACTICAL:
-
Prostate Cancer Association Group to Investigate Cancer Associated Alterations in the Genome
- GIANT:
-
Genetic Investigation of Anthropometric Traits consortium
- MRC-IEU:
-
MRC Integrative Epidemiology Unit
- IVs:
-
Instrumental variables
- IVW:
-
Inverse variance-weighted
- PRESSO:
-
Pleiotropy Residual Sum and Outlier
References
Aletaha D, Neogi T, Silman AJ, et al. 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010;62(9):2569–81. https://doi.org/10.1002/art.27584.
Seror R, Mariette X. Malignancy and the risks of biologic therapies: current status. Rheum Dis Clin North Am. 2017;43(1):43–64. https://doi.org/10.1016/j.rdc.2016.09.006.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre A, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018. https://doi.org/10.3322/caac.21492.
Barry MJ, Simmons LH. Prevention of prostate cancer morbidity and mortality: primary prevention and early detection. Med Clin North Am. 2017;101(4):787–806. https://doi.org/10.1016/j.mcna.2017.03.009.
Thomas E, Brewster DH, Black RJ, Macfarlane GJ. Risk of malignancy among patients with rheumatic conditions. Int J Cancer. 2000;88(3):497–502. https://doi.org/10.1002/1097-0215(20001101)88:3%3c497::AID-IJC27%3e3.0.CO;2-J.
Hellgren K, Smedby KE, Feltelius N, Baecklund E, Askling J. Do rheumatoid arthritis and lymphoma share risk factors?: A comparison of lymphoma and cancer risks before and after diagnosis of rheumatoid arthritis. Arthritis Rheum. 2010;62(5):1252–8. https://doi.org/10.1002/art.27402.
Yamada T, Nakajima A, Inoue E, et al. Incidence of malignancy in Japanese patients with rheumatoid arthritis. Rheumatol Int. 2011;31(11):1487–92. https://doi.org/10.1007/s00296-010-1524-0.
Hashimoto A, Chiba N, Tsuno H, et al. Incidence of malignancy and the risk of lymphoma in Japanese patients with rheumatoid arthritis compared to the general population. J Rheumatol. 2015;42(4):564–71. https://doi.org/10.3899/jrheum.140533.
Buchbinder R, VanDoornum S, Staples M, Lassere M, March L. Malignancy risk in Australian rheumatoid arthritis patients treated with anti-tumour necrosis factor therapy: analysis of the Australian Rheumatology Association Database (ARAD) prospective cohort study. BMC Musculoskelet Disord. 2015. https://doi.org/10.1186/s12891-015-0772-2.
Yu KH, Kuo CF, Huang LH, Huang WK, See LC. Cancer risk in patients with inflammatory systemic autoimmune rheumatic diseases: a nationwide population-based dynamic cohort study in Taiwan. Medicine (Baltimore). 2016;95(18):e3540. https://doi.org/10.1097/MD.0000000000003540.
Pernar CH, Ebot EM, Wilson KM, Mucci LA. The epidemiology of prostate cancer. Cold Spring Harb Perspect Med. 2018;8(12):a030361. https://doi.org/10.1101/cshperspect.a030361.
Emdin CA, Khera AV, Kathiresan S. Mendelian randomization. JAMA. 2017;318(19):1925–6. https://doi.org/10.1001/jama.2017.17219.
Smith GD, Ebrahim S. Mendelian randomization: prospects, potentials, and limitations. Int J Epidemiol. 2004;33(1):30–42. https://doi.org/10.1093/ije/dyh132.
Stroup DF, Berlin JA, Morton SC, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283(15):2008–12. https://doi.org/10.1001/jama.283.15.2008.
Wong WCW, Cheung CSK, Hart GJ. Development of a quality assessment tool for systematic reviews of observational studies (QATSO) of HIV prevalence in men having sex with men and associated risk behaviours. Emerg Themes Epidemiol. 2008;5:23. https://doi.org/10.1186/1742-7622-5-23.
Lin L, Chu H, Murad MH, et al. Empirical comparison of publication bias tests in meta-analysis. J Gen Intern Med. 2018;33(8):1260–7. https://doi.org/10.1007/s11606-018-4425-7.
: Data-Field 20002. Accessed May 16, 2023. https://biobank.ctsu.ox.ac.uk/crystal/field.cgi?id=20002
Fr S, Aa AO, Si B, et al. Association analyses of more than 140,000 men identify 63 new prostate cancer susceptibility loci. Nat Genet. 2018. https://doi.org/10.1038/s41588-018-0142-8.
Berndt SI, Gustafsson S, Mägi R, et al. Genome-wide meta-analysis identifies 11 new loci for anthropometric traits and provides insights into genetic architecture. Nat Genet. 2013;45(5):501–12. https://doi.org/10.1038/ng.2606.
Revez JA, Lin T, Qiao Z, et al. Genome-wide association study identifies 143 loci associated with 25 hydroxyvitamin D concentration. Nat Commun. 2020;11(1):1647. https://doi.org/10.1038/s41467-020-15421-7.
Burgess S. Sample size and power calculations in Mendelian randomization with a single instrumental variable and a binary outcome. Int J Epidemiol. 2014;43(3):922–9. https://doi.org/10.1093/ije/dyu005.
Burgess S, Dudbridge F, Thompson SG. Combining information on multiple instrumental variables in Mendelian randomization: comparison of allele score and summarized data methods. Stat Med. 2016;35(11):1880–906. https://doi.org/10.1002/sim.6835.
Verbanck M, Chen CY, Neale B, Do R. Publisher Correction: detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet. 2018;50(8):1196. https://doi.org/10.1038/s41588-018-0164-2.
Hemani G, Zheng J, Elsworth B, et al. The MR-Base platform supports systematic causal inference across the human phenome. Elife. 2018;7:e34408. https://doi.org/10.7554/eLife.34408.
Gridley G, McLaughlin JK, Ekbom A, et al. Incidence of cancer among patients with rheumatoid arthritis. J Natl Cancer Inst. 1993. https://doi.org/10.1093/jnci/85.4.307.
Mellemkjaer L, Linet MS, Gridley G, Frisch M, Møller H, Olsen JH. Rheumatoid arthritis and cancer risk. Eur J Cancer. 1996;32A(10):1753–7. https://doi.org/10.1016/0959-8049(96)00210-9.
Cibere J, Sibley J, Haga M. Rheumatoid arthritis and the risk of malignancy. Arthritis Rheum. 1997;40(9):1580–6. https://doi.org/10.1002/art.1780400906.
Askling J, Fored CM, Brandt L, et al. Risks of solid cancers in patients with rheumatoid arthritis and after treatment with tumour necrosis factor antagonists. Ann Rheum Dis. 2005. https://doi.org/10.1136/ard.2004.033993.
Abásolo L, Júdez E, Descalzo MA, et al. Cancer in rheumatoid arthritis: occurrence, mortality, and associated factors in a South European population. Semin Arthritis Rheum. 2008;37(6):388–97. https://doi.org/10.1016/j.semarthrit.2007.08.006.
Wolfe F, Michaud K. Biologic treatment of rheumatoid arthritis and the risk of malignancy: analyses from a large US observational study. Arthritis Rheum. 2007;56(9):2886–95. https://doi.org/10.1002/art.22864.
Hemminki K, Li X, Sundquist K, Sundquist J, Cancer risk in hospitalized rheumatoid arthritis patients. Rheumatology (Oxford, England). 2008;47(5). doi:https://doi.org/10.1093/rheumatology/ken130
Parikh-Patel A, White RH, Allen M, Cress R. Risk of cancer among rheumatoid arthritis patients in California. Cancer Causes Control : CCC. 2009. https://doi.org/10.1007/s10552-009-9298-y.
Chen Y-J, Chang Y-T, Wang C-B. C-Y Wu The risk of cancer in patients with rheumatoid arthritis: a nationwide cohort study in Taiwan. Arthritis Rheum. 2011. https://doi.org/10.1002/art.30134.
Dreyer L, Mellemkjær L, Andersen AR, et al. Incidences of overall and site specific cancers in TNFα inhibitor treated patients with rheumatoid arthritis and other arthritides - a follow-up study from the DANBIO Registry. Ann Rheum Dis. 2013;72(1):79–82. https://doi.org/10.1136/annrheumdis-2012-201969.
Mercer LK, Davies R, Galloway JB, et al. Risk of cancer in patients receiving non-biologic disease-modifying therapy for rheumatoid arthritis compared with the UK general population. Rheumatology. 2013;52(1):91–8. https://doi.org/10.1093/rheumatology/kes350.
Lim XR, Xiang W, Tan JWL, et al. Incidence and patterns of malignancies in a multi-ethnic cohort of rheumatoid arthritis patients. Int J Rheum Dis. 2019;22(9):1679–85. https://doi.org/10.1111/1756-185X.13655.
Lee H. The risk of malignancy in korean patients with rheumatoid arthritis. Yonsei Med J. 2019;60(2):223. https://doi.org/10.3349/ymj.2019.60.2.223.
Km K, Sj M. Prevalence, incidence, and risk factors of malignancy in patients with rheumatoid arthritis: a nationwide cohort study from Korea. Korean J Internal Med. 2021. https://doi.org/10.3904/kjim.2021.146.
Hochberg MC. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997;40(9):1725. https://doi.org/10.1002/art.1780400928.
Alarcón-Segovia D, Pérez-Vázquez ME, Villa AR, Drenkard C, Cabiedes J. Preliminary classification criteria for the antiphospholipid syndrome within systemic lupus erythematosus. Semin Arthritis Rheum. 1992;21(5):275–86. https://doi.org/10.1016/0049-0172(92)90021-5.
Tallbacka K, Pettersson T, Pukkala E. Increased incidence of cancer in systemic lupus erythematosus: a Finnish cohort study with more than 25 years of follow-up. Scand J Rheumatol. 2018;47(6):461–4. https://doi.org/10.1080/03009742.2017.1384054.
De Marzo AM, Platz EA, Sutcliffe S, et al. Inflammation in prostate carcinogenesis. Nat Rev Cancer. 2007;7(4):256–69. https://doi.org/10.1038/nrc2090.
Madaan S, Abel PD, Chaudhary KS, et al. Cytoplasmic induction and over-expression of cyclooxygenase-2 in human prostate cancer: implications for prevention and treatment. BJU Int. 2000;86(6):736–41. https://doi.org/10.1046/j.1464-410x.2000.00867.x.
Chaudry A, McClinton S, Moffat LE, Wahle KW. Essential fatty acid distribution in the plasma and tissue phospholipids of patients with benign and malignant prostatic disease. Br J Cancer. 1991;64(6):1157–60. https://doi.org/10.1038/bjc.1991.481.
Prostaglandin E2 Stimulates the beta-catenin/T cell factor-dependent transcription in colon cancer - PubMed. Accessed June 21, 2022. https://pubmed.ncbi.nlm.nih.gov/15899904/
Mahmud SM, Tanguay S, Bégin LR, Franco EL, Aprikian AG. Non-steroidal anti-inflammatory drug use and prostate cancer in a high-risk population. Eur J Cancer Prev. 2006;15(2):158–64. https://doi.org/10.1097/01.cej.0000197451.02604.25.
Stock DC, Groome PA, Siemens DR, Rohland SL, Song Z. Effects of non-selective non-steroidal anti-inflammatory drugs on the aggressiveness of prostate cancer. Prostate. 2008;68(15):1655–65. https://doi.org/10.1002/pros.20834.
Salinas CA, Kwon EM, FitzGerald LM, et al. Use of aspirin and other nonsteroidal antiinflammatory medications in relation to prostate cancer risk. Am J Epidemiol. 2010;172(5):578–90. https://doi.org/10.1093/aje/kwq175.
Heinrich PC, Behrmann I, Haan S, Hermanns HM, Müller-Newen G, Schaper F. Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem J. 2003;374(Pt 1):1–20. https://doi.org/10.1042/BJ20030407.
Katoh M, Katoh M. STAT3-induced WNT5A signaling loop in embryonic stem cells, adult normal tissues, chronic persistent inflammation, rheumatoid arthritis and cancer (Review). Int J Mol Med. 2007;19(2):273–8.
Iozzo RV, Eichstetter I, Danielson KG. Aberrant expression of the growth factor Wnt-5A in human malignancy. Cancer Res. 1995;55(16):3495–9.
Firestein GS. Invasive fibroblast-like synoviocytes in rheumatoid arthritis. Passive responders or transformed aggressors? Arthritis Rheum. 1996;39(11):1781–90. https://doi.org/10.1002/art.1780391103.
VanderWeele TJ, Tchetgen Tchetgen EJ, Cornelis M, Kraft P. Methodological challenges in mendelian randomization. Epidemiology. 2014;25(3):427–35. https://doi.org/10.1097/EDE.0000000000000081.
Rees JMB, Wood AM, Burgess S. Extending the MR-Egger method for multivariable Mendelian randomization to correct for both measured and unmeasured pleiotropy. Stat Med. 2017;36(29):4705–18. https://doi.org/10.1002/sim.7492.
Burgess S, Davies NM, Thompson SG. Bias due to participant overlap in two-sample Mendelian randomization. Genet Epidemiol. 2016;40(7):597–608. https://doi.org/10.1002/gepi.21998.
Freeman G, Cowling BJ, Schooling CM. Power and sample size calculations for Mendelian randomization studies using one genetic instrument. Int J Epidemiol. 2013;42(4):1157–63. https://doi.org/10.1093/ije/dyt110.
Acknowledgements
We would like to express our sincere gratitude to the National Natural Science Foundation of China for providing financial support for this study [Grant Numbers 82070778, 82272876, 82102975]. The authors thank for Editage (www.editage.cn) helping with the editing of the manuscript. The authors acknowledge the efforts of the genome-wide association study consortia (GWAS) for providing high-quality resources for researchers.
Funding
This work was supported by the National Natural Science Foundation of China [Grant Numbers: 82070778, 82272876, 81972381, 82102975].
Author information
Authors and Affiliations
Contributions
Conceptualization: JYO, CL, LZ. Search and evaluation: JYO, LZ. Data Analysis: JYO, YQW, YYF, QMZ, LZ. Writing—original draft and Visualization: JYO, LZ,HB. Writing—Review and Revision: CL, JYO, YQW, YYF, QMZ, MQ, HB. Supervision and Project Management: CL, LLM, HB, XJT, MQ, LZ, HB. Resources: CL, LLM, HB, XJT, MQ.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that the research was conducted without any commercial or financial relationships construed as a potential conflict of interest.
Ethical Approval
The ethical review board supported the research of all consortia, and Informed consent was waived as this was a meta-analysis of previous studies.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ou, J., Zou, L., Wu, Y. et al. Causal inference between rheumatoid arthritis and prostate cancer. Clin Exp Med 23, 4681–4694 (2023). https://doi.org/10.1007/s10238-023-01151-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-023-01151-9