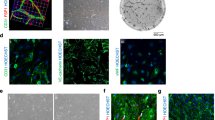
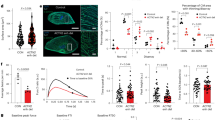
Abstract
Understanding how macrophages promote myocardial repair can help create new therapies for infarct repair. We aimed to determine what mechanisms underlie the reparative properties of macrophages. Cytokine arrays revealed that neonatal cardiac macrophages from the injured neonatal heart secreted high amounts of osteopontin (OPN). In vitro, recombinant OPN stimulated cardiac cell outgrowth, cardiomyocyte (CM) cell-cycle re-entry, and CM migration. In addition, OPN induced nuclear translocation of the cytoplasmatic yes-associated protein 1 (YAP1) and upregulated transcriptional factors and cell-cycle genes. Significantly, by blocking the OPN receptor CD44, we eliminated the effects of OPN on CMs. OPN also activated the proliferation and migration of non-CM cells: endothelial cells and cardiac mesenchymal stromal cells in vitro. Notably, the significant role of OPN in myocardial healing was demonstrated by impaired healing in OPN-deficient neonatal hearts. Finally, in the adult mice, a single injection of OPN into the border of the ischemic zone induced CM cell-cycle re-entry, improved scar formation, local and global cardiac function, and LV remodelling 30 days after MI. In summary, we have shown, for the first time, that recombinant OPN activates cell-cycle re-entry in CMs. In addition, recombinant OPN stimulates multiple cardiac cells and improves scar formation, LV remodelling, and regional and global function after MI. Therefore, we propose OPN as a new cell-free therapy to optimize infarct repair.








Similar content being viewed by others
Availability of data and material
A detailed description of the methods is provided in the Data Supplement Materials. All data that support the findings are available within the article, in the Data Supplement, or upon reasonable request from the corresponding author.
References
Albertsson AM, Zhang X, Leavenworth J, Bi D, Nair S, Qiao L, Hagberg H, Mallard C, Cantor H, Wang X (2014) The effect of osteopontin and osteopontin-derived peptides on preterm brain injury. J Neuroinflammation 11:197. https://doi.org/10.1186/s12974-014-0197-0
Aurora AB, Porrello ER, Tan W, Mahmoud AI, Hill JA, Bassel-Duby R, Sadek HA, Olson EN (2014) Macrophages are required for neonatal heart regeneration. J Clin Invest 124:1382–1392. https://doi.org/10.1172/JCI72181
Baehr A, Umansky KB, Bassat E, Jurisch V, Klett K, Bozoglu T, Hornaschewitz N, Solyanik O, Kain D, Ferraro B, Cohen-Rabi R, Krane M, Cyran C, Soehnlein O, Laugwitz KL, Hinkel R, Kupatt C, Tzahor E (2020) Agrin promotes coordinated therapeutic processes leading to improved cardiac repair in pigs. Circulation 142:868–881. https://doi.org/10.1161/CIRCULATIONAHA.119.045116
Bassat E, Mutlak YE, Genzelinakh A, Shadrin IY, Baruch Umansky K, Yifa O, Kain D, Rajchman D, Leach J, Riabov Bassat D, Udi Y, Sarig R, Sagi I, Martin JF, Bursac N, Cohen S, Tzahor E (2017) The extracellular matrix protein agrin promotes heart regeneration in mice. Nature 547:179–184. https://doi.org/10.1038/nature22978
Bauer M, Cheng S, Jain M, Ngoy S, Theodoropoulos C, Trujillo A, Lin FC, Liao R (2011) Echocardiographic speckle-tracking based strain imaging for rapid cardiovascular phenotyping in mice. Circ Res 108:908–916. https://doi.org/10.1161/CIRCRESAHA.110.239574
Ben-Mordechai T, Holbova R, Landa-Rouben N, Harel-Adar T, Feinberg MS, Abd Elrahman I, Blum G, Epstein FH, Silman Z, Cohen S, Leor J (2013) Macrophage subpopulations are essential for infarct repair with and without stem cell therapy. J Am Coll Cardiol 62:1890–1901. https://doi.org/10.1016/j.jacc.2013.07.057
Bergmann O, Zdunek S, Felker A, Salehpour M, Alkass K, Bernard S, Sjostrom SL, Szewczykowska M, Jackowska T, Dos Remedios C, Malm T, Andra M, Jashari R, Nyengaard JR, Possnert G, Jovinge S, Druid H, Frisen J (2015) Dynamics of cell generation and turnover in the human heart. Cell 161:1566–1575. https://doi.org/10.1016/j.cell.2015.05.026
Blewett CJ, Cilley RE, Ehrlich HP, Blackburn JH 2nd, Dillon PW, Krummel TM (1997) Regenerative healing of incisional wounds in midgestational murine hearts in organ culture. J Thorac Cardiovasc Surg 113:880–885. https://doi.org/10.1016/S0022-5223(97)70260-5
Chen W, Ma Q, Suzuki H, Hartman R, Tang J, Zhang JH (2011) Osteopontin reduced hypoxia-ischemia neonatal brain injury by suppression of apoptosis in a rat pup model. Stroke 42:764–769. https://doi.org/10.1161/STROKEAHA.110.599118
Dai J, Peng L, Fan K, Wang H, Wei R, Ji G, Cai J, Lu B, Li B, Zhang D, Kang Y, Tan M, Qian W, Guo Y (2009) Osteopontin induces angiogenesis through activation of PI3K/AKT and ERK1/2 in endothelial cells. Oncogene 28:3412–3422. https://doi.org/10.1038/onc.2009.189
Deng L, Zhou JF, Sellers RS, Li JF, Nguyen AV, Wang Y, Orlofsky A, Liu Q, Hume DA, Pollard JW, Augenlicht L, Lin EY (2010) A novel mouse model of inflammatory bowel disease links mammalian target of rapamycin-dependent hyperproliferation of colonic epithelium to inflammation-associated tumorigenesis. Am J Pathol 176:952–967. https://doi.org/10.2353/ajpath.2010.090622
Ehler E, Moore-Morris T, Lange S (2013) Isolation and culture of neonatal mouse cardiomyocytes. J Vis Exp JoVE 79: e50154. https://doi.org/10.3791/50154
Epelman S, Lavine KJ, Beaudin AE, Sojka DK, Carrero JA, Calderon B, Brija T, Gautier EL, Ivanov S, Satpathy AT, Schilling JD, Schwendener R, Sergin I, Razani B, Forsberg EC, Yokoyama WM, Unanue ER, Colonna M, Randolph GJ, Mann DL (2014) Embryonic and adult-derived resident cardiac macrophages are maintained through distinct mechanisms at steady state and during inflammation. Immunity 40:91–104. https://doi.org/10.1016/j.immuni.2013.11.019
Eschenhagen T, Bolli R, Braun T, Field LJ, Fleischmann BK, Frisen J, Giacca M, Hare JM, Houser S, Lee RT, Marban E, Martin JF, Molkentin JD, Murry CE, Riley PR, Ruiz-Lozano P, Sadek HA, Sussman MA, Hill JA (2017) Cardiomyocyte regeneration: a consensus statement. Circulation 136:680–686. https://doi.org/10.1161/CIRCULATIONAHA.117.029343
Frangogiannis NG (2019) The extracellular matrix in ischemic and nonischemic heart failure. Circ Res 125:117–146. https://doi.org/10.1161/CIRCRESAHA.119.311148
Graf K, Do YS, Ashizawa N, Meehan WP, Giachelli CM, Marboe CC, Fleck E, Hsueh WA (1997) Myocardial osteopontin expression is associated with left ventricular hypertrophy. Circulation 96:3063–3071. https://doi.org/10.1161/01.cir.96.9.3063
Haubner BJ, Schuetz T, Penninger JM (2016) A reproducible protocol for neonatal ischemic injury and cardiac regeneration in neonatal mice. Basic Res Cardiol 111:64. https://doi.org/10.1007/s00395-016-0580-3
He L, Nguyen NB, Ardehali R, Zhou B (2020) Heart regeneration by endogenous stem cells and cardiomyocyte proliferation: controversy, fallacy, and progress. Circulation 142:275–291. https://doi.org/10.1161/CIRCULATIONAHA.119.045566
Hsu KH, Tsai HW, Lin PW, Hsu YS, Shan YS, Lu PJ (2010) Clinical implication and mitotic effect of CD44 cleavage in relation to osteopontin/CD44 interaction and dysregulated cell cycle protein in gastrointestinal stromal tumor. Ann Surg Oncol 17:2199–2212. https://doi.org/10.1245/s10434-010-0927-1
Itou J, Oishi I, Kawakami H, Glass TJ, Richter J, Johnson A, Lund TC, Kawakami Y (2012) Migration of cardiomyocytes is essential for heart regeneration in zebrafish. Development 139:4133–4142. https://doi.org/10.1242/dev.079756
Koliaraki V, Prados A, Armaka M, Kollias G (2020) The mesenchymal context in inflammation, immunity and cancer. Nat Immunol 21:974–982. https://doi.org/10.1038/s41590-020-0741-2
Konfino T, Landa N, Ben-Mordechai T, Leor J (2015) The type of injury dictates the mode of repair in neonatal and adult heart. J Am Heart Assoc 4:e001320. https://doi.org/10.1161/JAHA.114.001320
Kuhn B, del Monte F, Hajjar RJ, Chang YS, Lebeche D, Arab S, Keating MT (2007) Periostin induces proliferation of differentiated cardiomyocytes and promotes cardiac repair. Nat Med 13:962–969. https://doi.org/10.1038/nm1619
Kuraoka M, Kimura E, Nagata T, Okada T, Aoki Y, Tachimori H, Yonemoto N, Imamura M, Si T (2016) Serum osteopontin as a novel biomarker for muscle regeneration in duchenne muscular dystrophy. Am J Pathol 186:1302–1312. https://doi.org/10.1016/j.ajpath.2016.01.002
Landa N, Miller L, Feinberg MS, Holbova R, Shachar M, Freeman I, Cohen S, Leor J (2008) Effect of injectable alginate implant on cardiac remodeling and function after recent and old infarcts in rat. Circulation 117:1388–1396. https://doi.org/10.1161/CIRCULATIONAHA.107.727420
Lavine KJ, Epelman S, Uchida K, Weber KJ, Nichols CG, Schilling JD, Ornitz DM, Randolph GJ, Mann DL (2014) Distinct macrophage lineages contribute to disparate patterns of cardiac recovery and remodeling in the neonatal and adult heart. Proc Natl Acad Sci U S A 111:16029–16034. https://doi.org/10.1073/pnas.1406508111
Leone M, Magadum A, Engel FB (2015) Cardiomyocyte proliferation in cardiac development and regeneration: a guide to methodologies and interpretations. Am J Physiol Heart Circ Physiol 309:H1237-1250. https://doi.org/10.1152/ajpheart.00559.2015
Liang CC, Park AY, Guan JL (2007) In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc 2:329–333. https://doi.org/10.1038/nprot.2007.30
Lok ZSY, Lyle AN (2019) Osteopontin in vascular disease. Arterioscler Thromb Vasc Biol 39:613–622. https://doi.org/10.1161/ATVBAHA.118.311577
Mahmoud AI, Porrello ER, Kimura W, Olson EN, Sadek HA (2014) Surgical models for cardiac regeneration in neonatal mice. Nat Protoc 9:305–311. https://doi.org/10.1038/nprot.2014.021
Millard SM, Heng O, Opperman KS, Sehgal A, Irvine KM, Kaur S, Sandrock CJ, Wu AC, Magor GW, Batoon L, Perkins AC, Noll JE, Zannettino ACW, Sester DP, Levesque JP, Hume DA, Raggatt LJ, Summers KM, Pettit AR (2021) Fragmentation of tissue-resident macrophages during isolation confounds analysis of single-cell preparations from mouse hematopoietic tissues. Cell Rep 37:110058. https://doi.org/10.1016/j.celrep.2021.110058
Mohamed TMA, Ang YS, Radzinsky E, Zhou P, Huang Y, Elfenbein A, Foley A, Magnitsky S, Srivastava D (2018) Regulation of cell cycle to stimulate adult cardiomyocyte proliferation and cardiac regeneration. Cell 173(104–116):e112. https://doi.org/10.1016/j.cell.2018.02.014
Murry CE, Giachelli CM, Schwartz SM, Vracko R (1994) Macrophages express osteopontin during repair of myocardial necrosis. Am J Pathol 145:1450–1462
Muzumdar MD, Tasic B, Miyamichi K, Li L, Luo L (2007) A global double-fluorescent Cre reporter mouse. Genesis 45:593–605. https://doi.org/10.1002/dvg.20335
Naftali-Shani N, Levin-Kotler LP, Palevski D, Amit U, Kain D, Landa N, Hochhauser E, Leor J (2017) Left ventricular dysfunction switches mesenchymal stromal cells toward an inflammatory phenotype and impairs their reparative properties via toll-like receptor-4. Circulation 135:2271–2287. https://doi.org/10.1161/CIRCULATIONAHA.116.023527
Ogawa K, Tsurutani M, Hashimoto A, Soeda M (2019) Simple propagation method for resident macrophages by co-culture and subculture, and their isolation from various organs. BMC Immunol 20:34. https://doi.org/10.1186/s12865-019-0314-z
Palevski D, Levin-Kotler LP, Kain D, Naftali-Shani N, Landa N, Ben-Mordechai T, Konfino T, Holbova R, Molotski N, Rosin-Arbesfeld R, Lang RA, Leor J (2017) Loss of macrophage wnt secretion improves remodeling and function after myocardial infarction in mice. J Am Heart Assoc. https://doi.org/10.1161/JAHA.116.004387
Paliwal P, Pishesha N, Wijaya D, Conboy IM (2012) Age dependent increase in the levels of osteopontin inhibits skeletal muscle regeneration. Aging (Albany NY) 4:553–566. https://doi.org/10.18632/aging.100477
Pfister G, Toor SM, Sasidharan Nair V, Elkord E (2020) An evaluation of sorter induced cell stress (SICS) on peripheral blood mononuclear cells (PBMCs) after different sort conditions—are your sorted cells getting SICS? J Immunol Methods 487:112902. https://doi.org/10.1016/j.jim.2020.112902
Phifer CB, Terry LM (1986) Use of hypothermia for general anesthesia in preweanling rodents. Physiol Behav 38:887–890
Plouffe BD, Murthy SK, Lewis LH (2015) Fundamentals and application of magnetic particles in cell isolation and enrichment: a review. Rep Prog Phys 78:016601. https://doi.org/10.1088/0034-4885/78/1/016601
Porrello ER, Mahmoud AI, Simpson E, Hill JA, Richardson JA, Olson EN, Sadek HA (2011) Transient regenerative potential of the neonatal mouse heart. Science 331:1078–1080. https://doi.org/10.1126/science.1200708
Povsic TJ, Sanz-Ruiz R, Climent AM, Bolli R, Taylor DA, Gersh BJ, Menasche P, Perin EC, Pompilio G, Atsma DE, Badimon L, DeMaria AN, Hare JM, Henry TD, Janssens S, Kastrup J, Torella D, Traverse JH, Willerson JT, Fernandez-Aviles F (2021) Reparative cell therapy for the heart: critical internal appraisal of the field in response to recent controversies. ESC Heart Fail. https://doi.org/10.1002/ehf2.13256
Sadek H, Olson EN (2020) Toward the goal of human heart regeneration. Cell Stem Cell 26:7–16. https://doi.org/10.1016/j.stem.2019.12.004
Sansonetti M, Waleczek FJG, Jung M, Thum T, Perbellini F (2020) Resident cardiac macrophages: crucial modulators of cardiac (patho)physiology. Basic Res Cardiol 115:77. https://doi.org/10.1007/s00395-020-00836-6
Sawaki D, Czibik G, Pini M, Ternacle J, Suffee N, Mercedes R, Marcelin G, Surenaud M, Marcos E, Gual P, Clement K, Hue S, Adnot S, Hatem SN, Tsuchimochi I, Yoshimitsu T, Henegar C, Derumeaux G (2018) Visceral adipose tissue drives cardiac aging through modulation of fibroblast senescence by osteopontin production. Circulation 138:809–822. https://doi.org/10.1161/CIRCULATIONAHA.117.031358
Sayers JR, Riley PR (2021) Heart regeneration: beyond new muscle and vessels. Cardiovasc Res 117:727–742. https://doi.org/10.1093/cvr/cvaa320
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682. https://doi.org/10.1038/nmeth.2019
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Sharon Y, Raz Y, Cohen N, Ben-Shmuel A, Schwartz H, Geiger T, Erez N (2015) Tumor-derived osteopontin reprograms normal mammary fibroblasts to promote inflammation and tumor growth in breast cancer. Cancer Res 75:963–973. https://doi.org/10.1158/0008-5472.Can-14-1990
Shirakawa K, Endo J, Kataoka M, Katsumata Y, Anzai A, Moriyama H, Kitakata H, Hiraide T, Ko S, Goto S, Ichihara G, Fukuda K, Minamino T, Sano M (2020) MerTK expression and ERK activation are essential for the functional maturation of osteopontin-producing reparative macrophages after myocardial infarction. J Am Heart Assoc 9:e017071. https://doi.org/10.1161/JAHA.120.017071
Shirakawa K, Endo J, Kataoka M, Katsumata Y, Yoshida N, Yamamoto T, Isobe S, Moriyama H, Goto S, Kitakata H, Hiraide T, Fukuda K, Sano M (2018) IL (Interleukin)-10-STAT3-galectin-3 axis is essential for osteopontin-producing reparative macrophage polarization after myocardial infarction. Circulation 138:2021–2035. https://doi.org/10.1161/CIRCULATIONAHA.118.035047
Shirakawa K, Sano M (2021) Osteopontin in cardiovascular diseases. Biomolecules. https://doi.org/10.3390/biom11071047
Singh K, Sirokman G, Communal C, Robinson KG, Conrad CH, Brooks WW, Bing OH, Colucci WS (1999) Myocardial osteopontin expression coincides with the development of heart failure. Hypertension 33:663–670
Singh M, Foster CR, Dalal S, Singh K (2010) Osteopontin: role in extracellular matrix deposition and myocardial remodeling post-MI. J Mol Cell Cardiol 48:538–543. https://doi.org/10.1016/j.yjmcc.2009.06.015
Suzuki H, Ayer R, Sugawara T, Chen W, Sozen T, Hasegawa Y, Kanamaru K, Zhang JH (2010) Protective effects of recombinant osteopontin on early brain injury after subarachnoid hemorrhage in rats. Crit Care Med 38:612–618. https://doi.org/10.1097/CCM.0b013e3181c027ae
Trueblood NA, Xie Z, Communal C, Sam F, Ngoy S, Liaw L, Jenkins AW, Wang J, Sawyer DB, Bing OH, Apstein CS, Colucci WS, Singh K (2001) Exaggerated left ventricular dilation and reduced collagen deposition after myocardial infarction in mice lacking osteopontin. Circ Res 88:1080–1087. https://doi.org/10.1161/hh1001.090842
Tzahor E, Poss KD (2017) Cardiac regeneration strategies: staying young at heart. Science 356:1035–1039. https://doi.org/10.1126/science.aam5894
Wasgewatte Wijesinghe DK, Mackie EJ, Pagel CN (2019) Normal inflammation and regeneration of muscle following injury require osteopontin from both muscle and non-muscle cells. Skelet Muscle 9:6. https://doi.org/10.1186/s13395-019-0190-5
Watanabe S, Alexander M, Misharin AV, Budinger GRS (2019) The role of macrophages in the resolution of inflammation. J Clin Invest 129:2619–2628. https://doi.org/10.1172/jci124615
Weber A, Büttner AL, Rellecke P, Petrov G, Albert A, Sixt SU, Lichtenberg A, Akhyari P (2020) Osteopontin as novel biomarker for reversibility of pressure overload induced left ventricular hypertrophy. Biomark Med 14:513–523. https://doi.org/10.2217/bmm-2019-0410
Wodsedalek DJ, Paddock SJ, Wan TC, Auchampach JA, Kenarsary A, Tsaih SW, Flister MJ, O’Meara CC (2019) IL-13 promotes in vivo neonatal cardiomyocyte cell cycle activity and heart regeneration. Am J Physiol Heart Circ Physiol 316:H24–H34. https://doi.org/10.1152/ajpheart.00521.2018
Wu CC, Jeratsch S, Graumann J, Stainier DYR (2020) Modulation of mammalian cardiomyocyte cytokinesis by the extracellular matrix. Circ Res 127:896–907. https://doi.org/10.1161/CIRCRESAHA.119.316303
Xin M, Kim Y, Sutherland LB, Murakami M, Qi X, McAnally J, Porrello ER, Mahmoud AI, Tan W, Shelton JM, Richardson JA, Sadek HA, Bassel-Duby R, Olson EN (2013) Hippo pathway effector Yap promotes cardiac regeneration. Proc Natl Acad Sci U S A 110:13839–13844. https://doi.org/10.1073/pnas.1313192110
Yousefi K, Irion CI, Takeuchi LM, Ding W, Lambert G, Eisenberg T, Sukkar S, Granzier HL, Methawasin M, Lee DI, Hahn VS, Kass DA, Hatzistergos KE, Hare JM, Webster KA, Shehadeh LA (2019) Osteopontin promotes left ventricular diastolic dysfunction through a mitochondrial pathway. J Am Coll Cardiol 73:2705–2718. https://doi.org/10.1016/j.jacc.2019.02.074
Zhang Y, Xia H, Ge X, Chen Q, Yuan D, Chen Q, Leng W, Chen L, Tang Q, Bi F (2014) CD44 acts through RhoA to regulate YAP signaling. Cell Signal 26:2504–2513. https://doi.org/10.1016/j.cellsig.2014.07.031
Zhao B, Tumaneng K, Guan KL (2011) The Hippo pathway in organ size control, tissue regeneration and stem cell self-renewal. Nat Cell Biol 13:877–883. https://doi.org/10.1038/ncb2303
Zhou Q, Li L, Zhao B, Guan KL (2015) The hippo pathway in heart development, regeneration, and diseases. Circ Res 116:1431–1447. https://doi.org/10.1161/CIRCRESAHA.116.303311
Acknowledgements
We thank Mrs. Noam Ziv for her skillful English-language editing. We thank Prof. Eldad Tzahor for his critical comments. This work was performed in partial fulfillment of requirements for the Ph.D. degree of Itai Rotem and Tal Konfino, Sackler Faculty of Medicine, Tel Aviv University, Israel.
Funding
We gratefully acknowledge support for this project provided by grants from the Seymour Fefer Foundation and the Israel Science Foundation (ISF). Itai Rotem was supported by a Ph.D. scholarship from Mrs. Tuna Gursoy.
Author information
Authors and Affiliations
Contributions
The authors confirm contributions to the paper as follows: study conception and design: IR, TK, and JL; data collection: IR, TK, TC, YS, OS-T, NL, NN-S, DL, DP, and JL; analysis and interpretation of results: IR, TK, TC, YS, OS-T, NL, and JL; draft manuscript preparation: IR, TK, TC, YS, OS-T, NL, NN-S, DL, DP, and JL. All authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no competing interests.
Ethics approval
All experimental protocols were approved by the Ethical Committees of the Sheba Medical Center.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (AVI 56911 KB)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rotem, I., Konfino, T., Caller, T. et al. Osteopontin promotes infarct repair. Basic Res Cardiol 117, 51 (2022). https://doi.org/10.1007/s00395-022-00957-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-022-00957-0